Advanced Liquid-Phase Isomerization of Alpha-Isophorone to Beta-Isophorone for Industrial Vitamin E Synthesis
The chemical industry continuously seeks robust methodologies for producing high-value intermediates, particularly those serving as critical precursors for vitamins and fine chemicals. Patent CN1235954A introduces a transformative approach for the preparation of 3,5,5-trimethylcyclohexa-3-en-1-one, commonly known as beta-isophorone, through the liquid-phase isomerization of its alpha-isomer. This innovation addresses long-standing inefficiencies in the synthesis of ketoisophorone and trimethylhydroquinone, which are essential building blocks for Vitamin E (tocopherol) production. By leveraging catalytic quantities of readily available alkali-metal salts or alkaline-earth salts, the disclosed method achieves a space-time yield suitable for large-scale industrial application without the need for complex auxiliary substances. This represents a significant paradigm shift from traditional gas-phase or stoichiometric organometallic routes, offering a cleaner, more economically viable pathway for reliable beta-isophorone supplier networks to meet the growing global demand for antioxidant intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isomerization of alpha-isophorone to beta-isophorone has been plagued by thermodynamic and kinetic challenges that hinder efficient commercial production. Traditional gas-phase methods, often utilizing heterogeneous catalysts like magnesium or aluminum oxides at temperatures ranging from 300°C to 800°C, suffer from severe selectivity issues. As illustrated in the reaction network below, the alpha-isomer can undergo multiple competing parallel reactions, leading to undesirable byproducts such as trimethylcyclohexadiene, 3,5-xylenol, and heavy coking residues. These side reactions not only consume valuable feedstock but also necessitate frequent catalyst regeneration and complex downstream purification steps. Furthermore, prior art involving stoichiometric Grignard reagents or homogeneous acid catalysts presents significant safety hazards and waste disposal challenges, with space-time yields often too low to justify the capital expenditure required for commercial scale-up of complex fragrance intermediates.
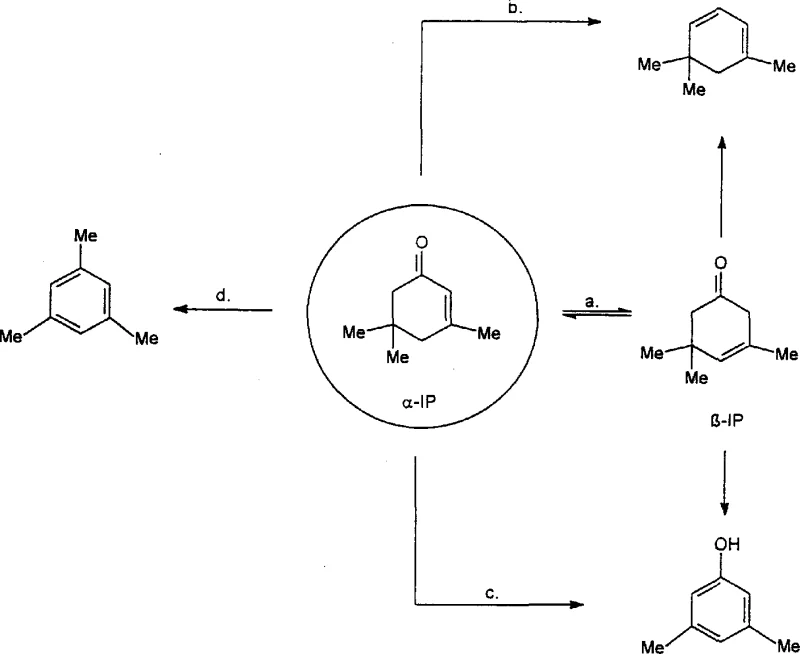
The Novel Approach
In stark contrast to these legacy techniques, the novel liquid-phase process described in CN1235954A utilizes simple salt-like catalysts to drive the equilibrium towards the desired beta-isomer with exceptional precision. By operating in the liquid phase at moderate temperatures between 100°C and 260°C, the method effectively suppresses the thermal decomposition pathways that dominate in gas-phase reactors. The use of alkali metal carbonates or hydroxides allows for a continuous operation mode where the reaction mixture is subjected to vacuum distillation, continuously removing the beta-isophorone product to prevent reverse isomerization. This strategic removal of product shifts the thermodynamic equilibrium, enabling conversion rates and selectivities that far exceed those of previous methods. Consequently, this approach facilitates cost reduction in vitamin E intermediate manufacturing by minimizing raw material loss and simplifying the isolation of high-purity beta-isophorone suitable for sensitive pharmaceutical applications.
Mechanistic Insights into Alkali Salt-Catalyzed Isomerization
The core mechanism of this improved process relies on the basicity of the alkali metal or alkaline earth metal salts to facilitate a proton transfer mechanism that rearranges the double bond within the cyclohexenone ring. Unlike acidic catalysts which may promote polymerization or dehydration, the basic environment provided by catalysts such as sodium carbonate (Na2CO3) or potassium hydroxide (KOH) selectively targets the allylic protons of the alpha-isophorone. This specific interaction promotes the migration of the double bond from the 2-position to the 3-position without inducing significant degradation of the carbon skeleton. Experimental data from the patent indicates that selectivity for beta-isophorone can exceed 99% when optimal catalyst concentrations, typically ranging from 0.001 to 5 mole percent, are employed. This high selectivity is crucial for maintaining the integrity of the product stream, ensuring that downstream oxidation to ketoisophorone proceeds without interference from isomeric impurities that could compromise the quality of the final vitamin or fragrance product.
Furthermore, the mechanistic advantage extends to the suppression of high-boiling byproducts (HB), which are often the bane of continuous chemical processes. In conventional systems, the accumulation of heavy ends leads to reactor fouling and reduced heat transfer efficiency over time. However, the salt-catalyzed liquid phase system demonstrates remarkable stability, with the formation rate of high-boiling materials remaining negligible even during extended operation. The catalyst itself remains active throughout the process, and because it is non-volatile, it stays in the reactor bottoms along with the unreacted alpha-isophorone. This allows for the seamless recycling of both the catalyst and the starting material, creating a closed-loop system that maximizes atom economy. For R&D teams focusing on process intensification, this mechanism offers a robust platform for reducing lead time for high-purity beta-isophorone batches by eliminating the need for frequent shutdowns and catalyst replacement cycles.
How to Synthesize Beta-Isophorone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a pilot or production setting. The process begins with the preparation of a suspension of the chosen alkali salt catalyst in industrial-grade alpha-isophorone, which is then fed into a heated isomerization reactor equipped with a distillation column. The reaction is conducted under conditions that allow for the continuous removal of the beta-isomer overhead while retaining the catalyst and unreacted alpha-isomer in the pot. Detailed standardized synthetic steps for replicating this high-efficiency isomerization are provided in the guide below.
- Prepare the reaction mixture by adding industrial grade alpha-isophorone and a catalytic amount of alkali metal salt (e.g., sodium carbonate or potassium hydroxide) into the isomerization reactor.
- Heat the suspension to a temperature range of 100-260°C under normal pressure or slight vacuum to initiate the isomerization reaction while maintaining constant stirring.
- Continuously distill the reaction mixture under vacuum to separate the formed beta-isophorone from the unreacted alpha-isophorone, recycling the bottoms back to the reactor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this liquid-phase isomerization technology translates directly into enhanced operational resilience and margin protection. The shift from expensive, specialized organometallic catalysts or complex heterogeneous fixed-bed systems to commodity alkali salts drastically simplifies the raw material sourcing strategy. Sodium carbonate and potassium hydroxide are globally available, low-cost commodities with stable pricing, insulating the production process from the volatility often associated with transition metal catalysts. This fundamental change in input materials ensures a more predictable cost structure, allowing for better long-term budgeting and contract negotiation with downstream customers in the pharmaceutical and agrochemical sectors who demand consistent supply reliability.
- Cost Reduction in Manufacturing: The elimination of stoichiometric reagents and the ability to recycle the catalyst indefinitely result in substantial cost savings per kilogram of product. By avoiding the use of precious metals or complex organic bases, the process removes the need for expensive metal scavenging steps and hazardous waste treatment associated with heavy metal residues. Additionally, the high space-time yield means that smaller reactor volumes can produce the same output as larger, less efficient gas-phase units, reducing the capital intensity and energy consumption per unit of production. This efficiency gain is critical for maintaining competitiveness in the global market for fine chemical intermediates where margin compression is a constant challenge.
- Enhanced Supply Chain Reliability: The robustness of the salt-catalyzed system significantly reduces the risk of unplanned production stoppages. Traditional gas-phase processes are prone to catalyst deactivation via coking, which necessitates frequent regeneration or replacement cycles that disrupt supply continuity. In contrast, the liquid-phase method described maintains consistent catalytic activity over prolonged periods, ensuring a steady flow of product to meet Just-In-Time delivery requirements. The simplicity of the equipment setup, which does not require specialized high-temperature alloy reactors or complex gas handling infrastructure, further enhances the reliability of the supply chain by reducing mechanical failure points and maintenance downtime.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process offers a cleaner profile that aligns with increasingly stringent global emission standards. The reduction in high-boiling byproducts and the absence of volatile organic solvents or corrosive acids minimize the burden on wastewater treatment facilities and off-gas scrubbing systems. The ability to operate at lower temperatures also contributes to a lower overall carbon footprint for the manufacturing site. For supply chain heads tasked with sustainability goals, adopting this technology demonstrates a commitment to green chemistry principles while simultaneously achieving the scalability needed to ramp up production from pilot kilograms to multi-tonne annual capacities without encountering the bottlenecks typical of older technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this isomerization technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: What catalysts are preferred for the isomerization of alpha-isophorone?
A: The patent specifies alkali metal salts such as sodium carbonate, potassium carbonate, sodium hydroxide, and lithium hydroxide as highly effective catalysts. These salts offer superior selectivity (>99%) compared to traditional heterogeneous gas-phase catalysts.
Q: How does this process improve space-time yield compared to gas-phase methods?
A: By operating in the liquid phase at moderate temperatures (150-250°C) rather than high-temperature gas phase (300-800°C), the process significantly reduces byproduct formation like trimethylbenzene and cyclohexadienes, thereby increasing the space-time yield of beta-isophorone.
Q: Can unreacted alpha-isophorone be recycled in this process?
A: Yes, the process is designed for continuous operation where the bottom product from the vacuum distillation column, containing unreacted alpha-isophorone and the catalyst, is circulated back to the isomerization unit, maximizing raw material efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Isophorone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the synthesis of life-saving vitamins and high-performance materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory concept to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of beta-isophorone performs consistently in your downstream oxidation and coupling reactions. Our facility is equipped to handle the specific thermal and distillation requirements of this liquid-phase process, optimizing yield and minimizing impurity profiles to support your R&D and manufacturing goals.
We invite you to collaborate with us to optimize your supply chain for vitamin precursors and fragrance intermediates. By leveraging our expertise in process chemistry, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project timelines and performance expectations. Let us be your strategic partner in navigating the complexities of fine chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
