Advanced Reactive Distillation for High-Purity Beta-Isophorone: A Commercial Scale-Up Breakthrough
Advanced Reactive Distillation for High-Purity Beta-Isophorone: A Commercial Scale-Up Breakthrough
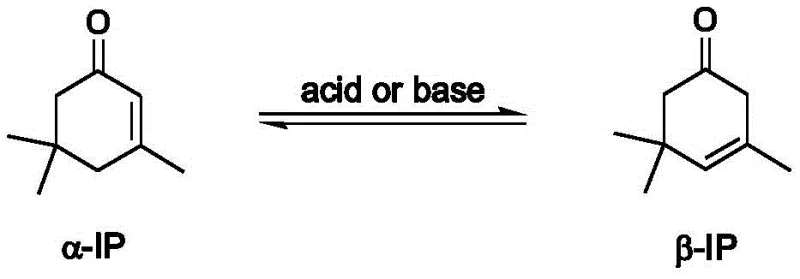
The global demand for high-purity fine chemical intermediates, particularly those serving the vitamin and fragrance sectors, necessitates manufacturing processes that balance efficiency with environmental sustainability. Patent CN107216244B introduces a transformative methodology for the preparation of 3,5,5-trimethyl-3-cyclohexen-1-one, commonly known as beta-isophorone (β-IP). This compound serves as a critical precursor in the synthesis of Vitamin E, carotenoids, astaxanthin, and various high-value fragrances. The disclosed technology utilizes a novel pre-nitrogen phosphazene catalyst within a reactive distillation framework, effectively addressing the longstanding challenges of low conversion rates, equipment corrosion, and byproduct accumulation that have plagued traditional isomerization routes. By shifting the reaction equilibrium through continuous product removal, this process achieves exceptional selectivity levels between 99.2% and 99.9%, establishing a new benchmark for industrial feasibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isomerization of alpha-isophorone (α-IP) to its beta-isomer has been fraught with significant technical and economic inefficiencies. Traditional methods relying on inorganic alkali catalysts, such as sodium hydroxide or sodium carbonate, suffer from severe operational drawbacks including the precipitation of salts which leads to clogging and necessitates complex filtration steps. Furthermore, these strong bases are highly corrosive to reactor vessels, increasing maintenance costs and safety risks. Alternative approaches utilizing transition metal catalysts like iron or cobalt acetylacetonates introduce heavy metal contamination concerns, requiring expensive purification steps to meet pharmaceutical grade standards. Additionally, acid-catalyzed routes often exhibit poor conversion rates and generate substantial amounts of self-condensation byproducts, drastically reducing the overall space-time yield and creating a heavy burden on waste treatment facilities.
The Novel Approach
The innovative process described in the patent overcomes these barriers by employing a homogeneous organocatalytic system based on pre-nitrogen phosphazene bases. Unlike heterogeneous or inorganic systems, these catalysts are fully soluble in the alpha-isophorone feedstock, eliminating the need for additional solvents and preventing salt precipitation entirely. The integration of reactive distillation technology allows for the continuous removal of the product, which has a lower boiling point (190°C) compared to the raw material (215°C). This physical separation drives the chemical equilibrium forward, ensuring high conversion without the need for excessive catalyst loading. The result is a streamlined, continuous operation that minimizes downstream processing requirements and maximizes resource utilization.
Mechanistic Insights into Phosphazene-Catalyzed Isomerization
The core of this technological advancement lies in the unique structural properties of the phosphazene catalyst, specifically the pre-nitrogen phosphazene derivatives defined by Formula (1). These molecules possess a highly basic nitrogen center shielded by significant steric bulk provided by the cyclic amine framework and variable alkyl substituents (R and R' groups). This steric hindrance is mechanistically critical; it physically blocks the approach of the isophorone anion intermediate to other isophorone molecules, thereby suppressing the unwanted self-condensation reactions that typically degrade yield in conventional base-catalyzed processes. By stabilizing the transition state for proton transfer while inhibiting nucleophilic attack on the carbonyl carbon of neighboring molecules, the catalyst ensures that the reaction pathway remains strictly focused on the desired double-bond migration.
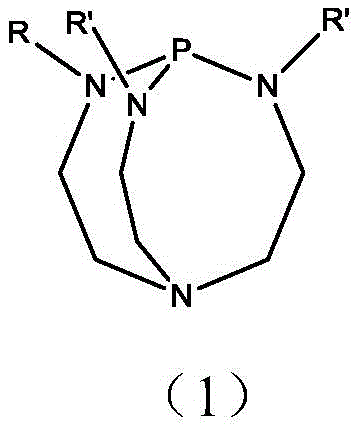
Furthermore, the patent highlights the strategic use of mixed catalyst systems, combining fully substituted and double-substituted phosphazenes in specific molar ratios. This combination allows for the precise fine-tuning of the system's basicity, optimizing the rate of isomerization while maintaining the integrity of the product against degradation. The homogeneous nature of the catalysis ensures uniform heat and mass transfer throughout the reactive distillation column, preventing local hot spots that could lead to polymerization. Consequently, the impurity profile of the crude product is significantly cleaner, with the reaction selectivity consistently exceeding 99.2%, which simplifies the subsequent vacuum rectification steps required to achieve the final specification of 99.5% to 99.8% purity.
How to Synthesize Beta-Isophorone Efficiently
The synthesis protocol outlined in the patent provides a robust framework for scaling this isomerization reaction from laboratory benchtop to full commercial production. The process begins with the preparation or procurement of the specific phosphazene catalyst variants, followed by a continuous reactive distillation setup that integrates reaction and separation into a single unit operation. This approach not only reduces capital expenditure on equipment but also lowers energy consumption by utilizing the heat of reaction for separation. For detailed operational parameters regarding catalyst loading, column specifications, and temperature gradients, please refer to the standardized synthesis guide below.
- Mix alpha-isophorone raw material with a catalytic amount (0.0001-1 wt%) of pre-nitrogen phosphazene catalyst at room temperature.
- Feed the mixture into a tower reactor heated to 150-230°C with a theoretical plate count of 25-50 and a reflux ratio of 2: 1 to 10:1.
- Continuously distill the lower-boiling beta-isophorone product from the top of the column while maintaining absolute pressure between 0.2-2 Bar to shift equilibrium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this phosphazene-catalyzed reactive distillation process offers compelling strategic advantages that extend beyond simple yield improvements. The elimination of inorganic salts and transition metals removes the need for extensive washing, filtration, and heavy metal scavenging steps, which translates directly into reduced utility consumption and shorter cycle times. The continuous nature of the reactive distillation column ensures a steady, predictable output of high-purity material, mitigating the risks associated with batch-to-batch variability. This reliability is crucial for maintaining uninterrupted supply chains for downstream customers in the vitamin and fragrance industries, where consistency is paramount.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings primarily through the drastic reduction in catalyst loading, which can be as low as 0.0001 wt% to 1 wt% relative to the substrate. Because the catalyst is soluble and highly selective, there is no need for expensive solvents or complex neutralization procedures that generate large volumes of saline wastewater. The ability to operate without added solvents reduces raw material costs and eliminates the energy-intensive step of solvent recovery. Additionally, the high selectivity minimizes the loss of valuable raw material to byproducts, effectively lowering the cost of goods sold (COGS) by maximizing the yield of the target molecule per unit of feedstock.
- Enhanced Supply Chain Reliability: The robustness of the phosphazene catalyst system contributes to enhanced supply continuity by reducing equipment downtime associated with cleaning and maintenance. Traditional processes involving corrosive acids or bases often require frequent reactor refurbishment, whereas the milder conditions of this organocatalytic method extend the lifespan of production assets. The simplified downstream processing, requiring only a single vacuum distillation step to reach >99.5% purity, accelerates the time from reaction completion to finished goods inventory. This agility allows manufacturers to respond more rapidly to market fluctuations and urgent customer orders without compromising on quality standards.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this technology aligns perfectly with modern green chemistry principles. The absence of heavy metals and the reduction in hazardous waste streams simplify compliance with increasingly stringent environmental regulations. The process is inherently scalable; the principles of reactive distillation function efficiently from pilot scale to multi-tonnage annual production, ensuring that quality remains consistent regardless of volume. The reduction in 'three wastes' (waste gas, liquid, and solid) not only lowers disposal costs but also enhances the corporate sustainability profile, a key factor for multinational clients evaluating their supplier base.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this advanced isomerization technology. These answers are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on how this method outperforms legacy systems in terms of purity, efficiency, and operational safety.
Q: Why is phosphazene catalyst superior to traditional inorganic bases for beta-isophorone production?
A: Unlike inorganic bases like NaOH which cause salt precipitation and severe equipment corrosion, phosphazene catalysts are soluble in the reaction medium, eliminating salt-out issues. Furthermore, their bulky steric structure inhibits self-condensation byproducts, resulting in significantly higher selectivity (up to 99.9%) and reduced waste generation.
Q: How does the reactive distillation process improve yield compared to batch isomerization?
A: The process leverages the boiling point difference between the原料 alpha-isophorone (215°C) and the product beta-isophorone (190°C). By continuously removing the lower-boiling product from the top of the column, the chemical equilibrium is constantly shifted towards product formation, overcoming the thermodynamic limitations of static batch reactions.
Q: What represents the primary purity specification achievable with this technology?
A: Through the combination of highly selective phosphazene catalysis and subsequent vacuum rectification, the technology achieves a final product purity ranging from 99.5 wt% to 99.8 wt%, which meets the stringent requirements for downstream synthesis of Vitamin E and carotenoid intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Isophorone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic processes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the phosphazene-catalyzed route are fully realized in practice. Our facilities are equipped with state-of-the-art reactive distillation columns and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of beta-isophorone meets the exacting standards required for Vitamin E and carotenoid synthesis.
We invite potential partners to engage with our technical team to explore how this innovative process can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the specific economic benefits relevant to your operation. We encourage you to contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments, ensuring a seamless integration of this high-performance intermediate into your manufacturing portfolio.
