Advanced Manufacturing of Prilocaine Hydrochloride: A Scalable Route for Global API Production
The pharmaceutical industry continuously seeks robust, scalable, and environmentally compliant synthetic routes for critical active pharmaceutical ingredients (APIs) and their intermediates. Patent CN102093248A presents a significant technological advancement in the preparation of N-(2-Methylphenyl)-2-(propylamino)propanamide, commonly known as Prilocaine Hydrochloride or Xylanest. This local anesthetic is vital for various surgical procedures, including epidural and infiltration anesthesia, offering a superior safety profile compared to older agents like procaine. The disclosed methodology addresses longstanding challenges in the synthesis of this compound by eliminating harsh reaction conditions such as high temperature and high pressure, which traditionally pose safety risks and increase capital expenditure for manufacturing facilities. By leveraging a mild, three-step sequence involving amidation, amination, and salt formation, this patent outlines a pathway that not only enhances operational safety but also dramatically improves the overall yield and purity of the final product, making it a cornerstone technology for modern pharmaceutical intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Prilocaine and its hydrochloride salt has been plagued by the use of hazardous reagents and inefficient catalytic systems that hinder large-scale industrial adoption. Traditional protocols often relied on benzene or toluene as primary solvents, both of which are classified as carcinogenic or toxic, necessitating expensive containment systems and rigorous waste treatment protocols to meet environmental regulations. Furthermore, conventional methods frequently utilized pyridine as a catalyst or solvent, a substance known for its strong, unpleasant odor and toxicity, which complicates operator safety and requires specialized ventilation infrastructure. Another significant bottleneck in prior art was the use of sodium carbonate (yellow soda ash) as a base, which demonstrated relatively low catalytic efficiency, leading to incomplete reactions and the formation of difficult-to-remove impurities. These factors combined to create a manufacturing process with suboptimal yields, higher production costs due to waste disposal, and increased lead times, ultimately affecting the reliability of the pharmaceutical intermediate supplier network.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent introduces a greener, more efficient synthetic strategy that prioritizes both economic viability and operator safety. The core innovation lies in the substitution of toxic solvents with acetone and water, coupled with the use of potassium carbonate as a highly effective catalyst for the initial amidation step. This shift allows the reaction to proceed smoothly at mild temperatures ranging from 20°C to 30°C, eliminating the energy-intensive requirements of high-heat processes. The new route ensures that raw materials are readily available and inexpensive, while the simplified operational steps—such as straightforward filtration and washing—drastically reduce processing time. By avoiding the use of benzene and pyridine, the process inherently lowers the burden on environmental compliance teams and reduces the total cost of ownership for manufacturing plants. This represents a paradigm shift in cost reduction in API manufacturing, transforming a complex, hazardous operation into a streamlined, high-yield procedure suitable for global supply chains.
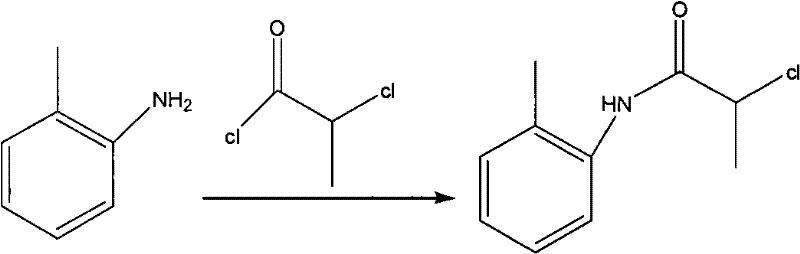
Mechanistic Insights into Potassium Carbonate-Catalyzed Amidation
The first critical stage of this synthesis involves the amidation of o-toluidine with 2-chloropropionyl chloride, a reaction that sets the foundation for the entire molecular architecture. In this mechanism, potassium carbonate acts not merely as a base but as a crucial acid scavenger that neutralizes the hydrochloric acid byproduct generated during the nucleophilic attack of the amine on the acid chloride. The use of acetone as a solvent under ice-water bath conditions ensures that the exothermic nature of the reaction is carefully controlled, preventing thermal runaway and the formation of degradation byproducts. The molar ratio of o-toluidine to 2-chloropropionyl chloride is optimized between 1:1 and 1:1.5, ensuring that the limiting reagent is fully consumed while minimizing excess waste. The precipitation of the intermediate solid during the reaction serves as a driving force, pushing the equilibrium towards product formation according to Le Chatelier's principle. This precise control over stoichiometry and temperature is what allows the process to achieve exceptional yields, often exceeding 90%, which is a critical metric for high-purity pharmaceutical intermediates.
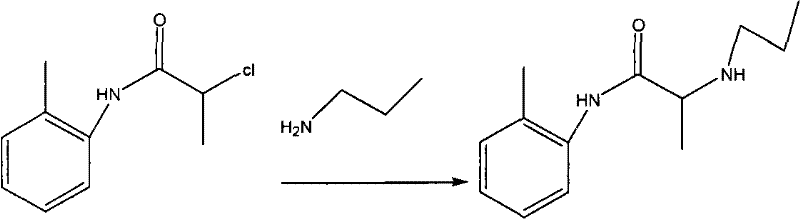
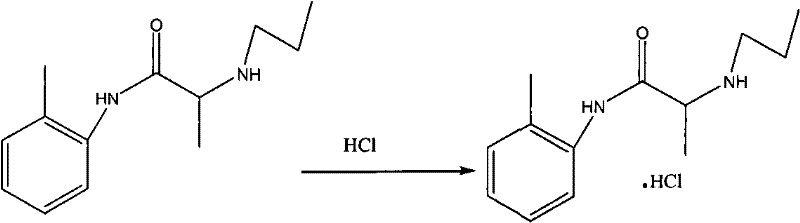
Following the initial amidation, the second step involves a nucleophilic substitution where the chloro-group of the intermediate is displaced by n-propylamine. This transformation is facilitated by heating the mixture in acetone or toluene to reflux temperatures between 70°C and 110°C for a duration of 12 to 14 hours. The extended reaction time ensures complete conversion of the intermediate, which is verified via Thin Layer Chromatography (TLC) to detect the disappearance of the starting material spot. The choice of solvent here is flexible, allowing manufacturers to choose based on availability and boiling point preferences, adding to the process's robustness. Finally, the crude base is converted into the stable hydrochloride salt by dissolving it in ethyl acetate or acetone and adjusting the pH to between 1 and 3 using concentrated hydrochloric acid. This acidification step not only stabilizes the molecule for storage but also facilitates purification through recrystallization, ensuring the final product meets stringent quality specifications with a melting point of 167-168°C and content greater than 99.0%.
How to Synthesize Prilocaine Hydrochloride Efficiently
Implementing this synthesis route requires careful attention to temperature control and stoichiometric ratios to maximize the benefits of the patented method. The process is divided into three distinct operational units: the initial amidation to form the chloro-intermediate, the subsequent amination to introduce the propyl group, and the final salification to produce the stable hydrochloride salt. Each step has been optimized to minimize impurity profiles and maximize throughput, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates. Operators must ensure that the addition of 2-chloropropionyl chloride is performed slowly to maintain the internal temperature below 30°C, as this prevents side reactions that could compromise the purity of the final anesthetic agent. The workup procedures, involving simple water washes and filtration, are designed to be easily automated or scaled in standard stainless steel reactors without the need for exotic materials of construction.
- Perform amidation of o-toluidine with 2-chloropropionyl chloride using potassium carbonate in acetone at 20-30°C.
- React the resulting intermediate with n-propylamine in acetone or toluene under reflux conditions (70-110°C) for 12-14 hours.
- Dissolve the crude product in ethyl acetate or acetone and adjust pH to 1-3 with concentrated hydrochloric acid to form the final salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic advantages that extend beyond simple chemical yield. The elimination of toxic solvents like benzene and catalysts like pyridine removes a significant layer of regulatory complexity and cost associated with hazardous material handling, storage, and disposal. This simplification of the chemical bill of materials (BOM) directly translates to cost reduction in pharmaceutical intermediate manufacturing by lowering the overhead required for environmental health and safety (EHS) compliance. Furthermore, the use of commodity chemicals such as acetone, potassium carbonate, and hydrochloric acid ensures that the supply chain is resilient against market volatility, as these reagents are globally sourced and unlikely to face shortages. The high yield reported in the patent examples, particularly the 93% yield in the first step, means that less raw material is wasted per kilogram of finished product, effectively reducing the cost of goods sold (COGS) and improving margin potential for downstream API manufacturers.
- Cost Reduction in Manufacturing: The process achieves significant cost savings by replacing expensive and hazardous reagents with inexpensive, commodity-grade chemicals. The high catalytic efficiency of potassium carbonate reduces the amount of base required compared to sodium carbonate, while the high conversion rates minimize the loss of valuable starting materials like o-toluidine. Additionally, the simplified workup procedure eliminates the need for complex extraction sequences or chromatographic purifications, reducing solvent consumption and energy usage during the drying phases. These cumulative efficiencies result in a leaner manufacturing process that is highly competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: By relying on widely available raw materials and avoiding specialty reagents that may have long lead times, this method ensures a consistent and reliable supply of the intermediate. The robustness of the reaction conditions, which tolerate slight variations in temperature and mixing without significant yield loss, makes the process forgiving and stable for contract manufacturing organizations (CMOs). This stability is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing buyers to plan their inventory with greater confidence and avoid production stoppages caused by raw material delays or batch failures.
- Scalability and Environmental Compliance: The absence of high-pressure requirements and the use of standard solvents make this process inherently scalable from pilot plant to multi-ton production. The removal of carcinogenic benzene and toxic pyridine aligns the manufacturing process with modern green chemistry principles and strict international environmental regulations. This compliance reduces the risk of regulatory shutdowns and simplifies the permitting process for new production lines. The ability to recycle solvents like acetone and ethyl acetate further enhances the sustainability profile of the operation, appealing to stakeholders focused on corporate social responsibility and long-term environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. They are derived from the specific improvements and data points highlighted in the patent documentation, providing clarity on how this technology compares to existing standards. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement teams assessing the quality and consistency of suppliers utilizing this method.
Q: What are the key advantages of this synthesis method over conventional routes?
A: This method eliminates the use of toxic solvents like benzene and hazardous catalysts like pyridine. It operates under mild conditions (20-30°C for the first step) and utilizes readily available raw materials, significantly improving safety and environmental compliance for industrial production.
Q: How does the new catalytic system improve yield?
A: By replacing sodium carbonate with potassium carbonate and optimizing the solvent system to acetone/water, the reaction achieves higher conversion rates. The patent reports yields up to 93% in the initial amidation step, compared to lower efficiencies in traditional methods.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is specifically designed for industrial scalability. It avoids high-pressure equipment, uses simple workup procedures like filtration and washing, and ensures high purity (99.0%) through recrystallization, making it ideal for reliable supply chain integration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prilocaine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the supply of high-quality anesthetic intermediates. Our technical team has extensively analyzed the route described in CN102093248A and possesses the expertise to execute this chemistry with precision and consistency. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require material for clinical trials or full-scale commercial launch, our capacity can meet your demands. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of Prilocaine Hydrochloride meets the rigorous standards required for pharmaceutical applications, including content assays and impurity profiling.
We invite global partners to collaborate with us to leverage this advanced synthetic technology for their supply chains. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage interested parties to contact our technical procurement team to request specific COA data and comprehensive route feasibility assessments. Let us help you optimize your production costs and secure a stable supply of this vital pharmaceutical intermediate.
