Industrial Scale Synthesis of Prilocaine Hydrochloride: A Green Chemistry Breakthrough for API Manufacturing
The pharmaceutical industry is constantly evolving towards greener, safer, and more efficient manufacturing processes, particularly for critical local anesthetics like Prilocaine Hydrochloride. Patent CN102093248B introduces a transformative method for preparing N-(2-Methylphenyl)-2-(propylamino)propanamide, commonly known as Prilocaine or Xylanest, which addresses significant historical limitations in synthetic chemistry. This innovation shifts the paradigm from hazardous, high-energy protocols to a streamlined, ambient-pressure workflow that utilizes benign solvents and highly effective catalysts. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding the technical nuances of this patent is crucial, as it represents a viable pathway for cost reduction in API manufacturing while adhering to strict environmental regulations. The method eliminates the need for toxic benzene and pyridine, replacing them with acetone and ethyl acetate, thereby creating a safer working environment and reducing the burden on waste treatment facilities. Furthermore, the optimization of reaction conditions ensures that the process is not only chemically robust but also economically superior, offering a compelling value proposition for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Prilocaine has been plagued by the use of archaic and hazardous chemical reagents that pose severe risks to both operator safety and environmental compliance. Traditional protocols frequently relied on benzene or toluene as primary solvents, substances now heavily restricted due to their carcinogenic properties and stringent regulatory caps on residual limits in final drug products. Additionally, the use of pyridine as a catalyst or solvent introduced strong, irritating odors and toxicity issues that complicated large-scale handling and required expensive containment systems. Conventional methods also often employed sodium carbonate as a catalyst, which, while inexpensive, demonstrated suboptimal catalytic efficiency leading to incomplete reactions and lower overall yields. These inefficiencies necessitated complex purification steps to remove unreacted starting materials and by-products, driving up production costs and extending lead times. The reliance on harsh reaction conditions, including potential high-temperature requirements in older variations, further increased energy consumption and equipment wear, making the commercial scale-up of complex pharmaceutical intermediates challenging and risky for manufacturers aiming for consistent quality.
The Novel Approach
The methodology outlined in Patent CN102093248B presents a sophisticated solution to these legacy problems by engineering a reaction pathway that prioritizes safety, efficiency, and purity. By substituting toxic aromatic solvents with acetone and ethyl acetate, the new process drastically reduces the toxicity profile of the entire manufacturing line, facilitating easier solvent recovery and recycling. The strategic replacement of sodium carbonate with potassium carbonate serves as a critical upgrade, as potassium salts generally exhibit better solubility and reactivity in organic media, acting as a more potent acid scavenger during the amidation phase. This catalytic improvement drives the reaction to completion more effectively, minimizing the formation of side products and simplifying the downstream purification workload. Moreover, the process operates under remarkably mild conditions, with the initial amidation occurring at a controlled 20-30°C, eliminating the need for energy-intensive heating or high-pressure vessels. This gentle approach not only preserves the integrity of the sensitive amine functionalities but also allows for the use of standard glass-lined or stainless steel reactors, significantly lowering capital expenditure barriers for high-purity pharmaceutical intermediates production.
Mechanistic Insights into Potassium Carbonate-Catalyzed Amidation and Amination
The core of this synthetic breakthrough lies in the precise control of nucleophilic substitution reactions, beginning with the formation of the key chloro-amide intermediate. In the first step, o-toluidine acts as a nucleophile, attacking the electrophilic carbonyl carbon of 2-chloropropionyl chloride. Potassium carbonate plays a dual role here: it acts as a base to deprotonate the aniline nitrogen, increasing its nucleophilicity, and simultaneously neutralizes the hydrochloric acid by-product generated during the acylation. This acid scavenging is vital; without it, the accumulation of HCl would protonate the remaining o-toluidine, rendering it non-nucleophilic and stalling the reaction. The use of an ice-water bath during the addition of the acid chloride is a critical process parameter, as the reaction is exothermic. Maintaining the temperature between 20-30°C prevents thermal degradation of the reagents and controls the rate of gas evolution, ensuring a smooth reaction profile. The precipitation of the intermediate solid during the reaction is a beneficial phenomenon, as it drives the equilibrium forward according to Le Chatelier's principle and facilitates easy isolation via filtration.
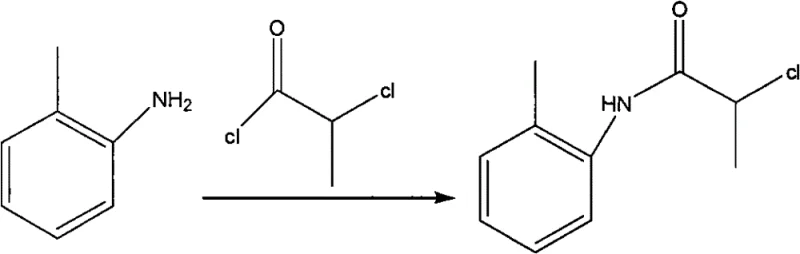
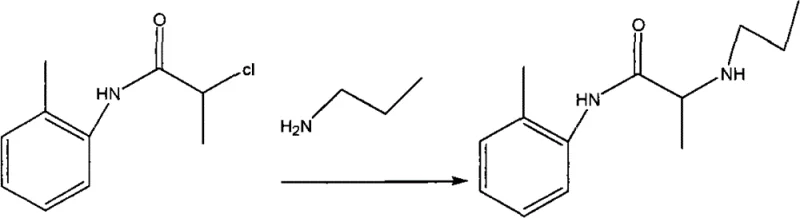
Following the isolation of the intermediate, the second mechanistic phase involves a nucleophilic substitution where the chlorine atom on the alpha-carbon is displaced by propylamine. This step transforms the chloro-amide into the final Prilocaine base. The reaction requires elevated temperatures, typically refluxing between 70-110°C in acetone or toluene, to overcome the activation energy barrier for the displacement of the chloride ion. Propylamine, being a strong nucleophile, attacks the chiral center, and the presence of excess amine helps drive the reaction to completion while also acting as a base to mop up any generated acid. The patent specifies a molar ratio of intermediate to propylamine of 1:2 to 1:4, ensuring that the concentration of the nucleophile remains high throughout the 12 to 14-hour reaction window. This extended reaction time is necessary to ensure full conversion, which is monitored via Thin Layer Chromatography (TLC) to detect the disappearance of the starting material spot. The final step involves the conversion of the free base into its hydrochloride salt by adjusting the pH to 1-3 with concentrated hydrochloric acid, a process that exploits the difference in solubility between the free base and the salt to achieve high-purity crystallization.
How to Synthesize Prilocaine Hydrochloride Efficiently
Executing this synthesis requires strict adherence to the optimized parameters defined in the patent to maximize yield and minimize impurities. The process is divided into three distinct operational units: the low-temperature amidation, the high-temperature amination, and the final salification and purification. Operators must pay close attention to the temperature control during the acid chloride addition and the stoichiometric ratios of the catalysts to ensure reproducibility. The following guide outlines the standardized workflow derived from the patent examples, serving as a foundational protocol for process engineers aiming to implement this technology.
- Perform amidation of o-toluidine with 2-chloropropionyl chloride using potassium carbonate in acetone at 20-30°C to form the chloro-amide intermediate.
- React the intermediate with propylamine in acetone or toluene under reflux (70-110°C) to substitute the chlorine atom and form the prilocaine base.
- Dissolve the crude base in ethyl acetate or acetone and adjust pH to 1-3 with concentrated hydrochloric acid to precipitate pure Prilocaine Hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers profound strategic advantages that extend beyond simple chemical yield. The elimination of benzene and pyridine from the supply chain removes the logistical and regulatory burdens associated with handling Schedule 1 or highly restricted hazardous chemicals. This shift significantly simplifies the procurement process, as acetone and ethyl acetate are commodity chemicals with stable pricing and abundant global availability. Furthermore, the removal of toxic solvents drastically reduces the cost of waste disposal and environmental compliance, as the effluent stream is far less hazardous and easier to treat. The mild reaction conditions also translate to lower energy consumption, as the process does not require cryogenic cooling below 0°C or high-pressure autoclaves, allowing for the use of standard multipurpose reactors found in most CDMO facilities. These factors combine to create a manufacturing process that is inherently more resilient to supply shocks and regulatory changes, ensuring a steady flow of high-purity pharmaceutical intermediates to downstream API manufacturers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the optimization of raw material utilization and the simplification of unit operations. By utilizing potassium carbonate instead of sodium carbonate, the reaction efficiency is markedly improved, leading to higher conversion rates and less waste of expensive starting materials like o-toluidine and propylamine. The high yield reported in the patent examples means that less raw material is required per kilogram of final product, directly lowering the Cost of Goods Sold (COGS). Additionally, the simplified work-up procedure, which relies on straightforward filtration and water washing rather than complex extractions or distillations, reduces labor hours and utility costs. The ability to recycle solvents like acetone and ethyl acetate further enhances the economic viability, creating a closed-loop system that minimizes material loss and maximizes profitability for the reliable pharmaceutical intermediates supplier.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of universally available reagents that are not subject to the same geopolitical or regulatory restrictions as benzene or pyridine. The robustness of the reaction conditions means that production is less susceptible to interruptions caused by equipment failure or utility fluctuations, as the process tolerances are wider and more forgiving. The high purity of the crude product obtained after the amination step reduces the dependency on complex chromatographic purification, which can often be a bottleneck in manufacturing schedules. This streamlined workflow ensures faster batch turnover times, allowing suppliers to respond more agilely to market demand spikes. Consequently, partners can rely on a more predictable delivery schedule, reducing lead time for high-purity pharmaceutical intermediates and securing their own production timelines against upstream volatility.
- Scalability and Environmental Compliance: From a scalability perspective, the absence of high-pressure steps and the use of common solvents make this process exceptionally easy to transfer from pilot plant to full commercial scale. The exothermic nature of the first step is manageable with standard jacketed reactors, removing the need for specialized cryogenic infrastructure. Environmentally, the process aligns with Green Chemistry principles by reducing the use of hazardous substances and generating less toxic waste, which is increasingly important for maintaining social license to operate and meeting corporate sustainability goals. The final product's high purity (over 99% in examples) minimizes the risk of regulatory rejection due to impurity profiles, safeguarding the supply chain from costly recalls or batch rejections. This combination of operational ease and environmental stewardship makes the technology a future-proof investment for long-term cost reduction in API manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and technical specifications provided in Patent CN102093248B, offering clarity on yield expectations, solvent choices, and purification strategies. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific production needs.
Q: How does this new method improve upon traditional Prilocaine synthesis?
A: Traditional methods often utilize toxic solvents like benzene and pyridine, along with less efficient catalysts like sodium carbonate. This patented process replaces hazardous solvents with acetone and ethyl acetate, uses potassium carbonate for higher catalytic efficiency, and operates under milder conditions, significantly reducing environmental impact and operational risks.
Q: What are the typical yields achieved in this synthetic route?
A: According to the experimental data in Patent CN102093248B, the process demonstrates high efficiency. The initial amidation step can achieve yields around 93%, the subsequent amination step reaches approximately 87%, and the final salification and recrystallization yield about 85%, resulting in a high-purity final product with a melting point of 167-168°C.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is specifically designed for industrial scalability. It avoids harsh conditions such as high pressure and extreme temperatures, utilizes readily available raw materials, and simplifies post-treatment through straightforward filtration and recrystallization, making it ideal for reliable pharmaceutical intermediates supplier operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prilocaine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is critical for the future of the pharmaceutical industry. Our team of expert chemists has thoroughly analyzed Patent CN102093248B and possesses the technical capability to execute this advanced Prilocaine Hydrochloride synthesis with precision and consistency. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this laboratory-optimized process are fully realized at an industrial level. Our facilities are equipped with state-of-the-art reaction vessels capable of maintaining the strict temperature controls required for the amidation step, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards.
We invite global partners to collaborate with us to leverage this innovative technology for their supply chains. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this green chemistry approach can optimize your bottom line. We encourage interested parties to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance. Let us be your partner in delivering high-quality, cost-effective, and sustainable pharmaceutical intermediates to the global market.
