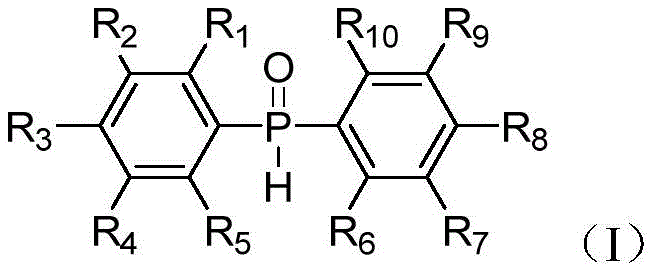
Advanced Synthesis of Substituted Diarylphosphine Oxides for High-Performance Polymer Additives
Advanced Synthesis of Substituted Diarylphosphine Oxides for High-Performance Polymer Additives
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the demand for greener, more versatile synthetic routes for high-value intermediates. A pivotal advancement in this field is documented in Chinese Patent CN110872321B, which discloses a novel preparation method for diarylphosphine oxide compounds containing various substituent groups. This technology represents a paradigm shift from traditional high-energy disproportionation reactions to a mild, catalytic electrophilic substitution pathway. For R&D directors and procurement strategists in the polymer and specialty chemical sectors, this patent offers a robust solution for producing critical monomers used in epoxy resin flame retardancy and acylphosphine photoinitiators. The ability to synthesize these compounds under温和 (mild) conditions not only enhances safety profiles but also drastically reduces the environmental footprint associated with solid waste treatment, positioning this method as a cornerstone for sustainable chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of diphenyl phosphine oxide (DPO) and its derivatives relied heavily on the high-temperature disproportionation of phenyl phosphine dichloride. As illustrated in the legacy process flow, this conventional approach necessitates reaction temperatures exceeding 200°C, leading to substantial energy consumption and operational hazards. Furthermore, the process generates a stoichiometric amount of aluminum chloride complexes that require aggressive decomplexing agents to liberate the product. This step produces massive quantities of difficult-to-treat solid waste, creating a severe environmental burden and inflating disposal costs. Crucially, the disproportionation mechanism inherently limits structural diversity, making it virtually impossible to introduce different substituent groups onto the benzene rings, thereby restricting the application scope of the resulting materials in advanced polymer formulations.

The Novel Approach
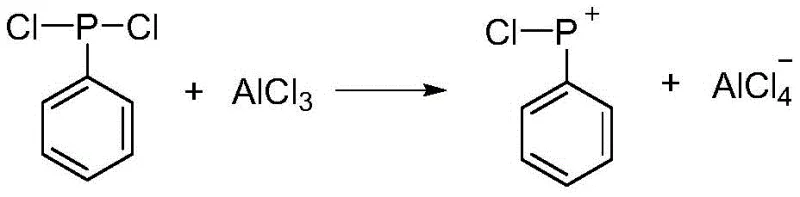
In stark contrast, the methodology outlined in CN110872321B utilizes a direct coupling strategy between aryl phosphine dichloride and aromatic hydrocarbons catalyzed by Lewis acids such as anhydrous aluminum chloride. This innovative route operates at significantly lower temperatures, typically between 60°C and 100°C, which dramatically lowers the thermal load on reactor systems. The process bypasses the need for separate decomplexing agents; instead, the intermediate complex is directly hydrolyzed in an ice-water mixture to yield the target phosphine oxide. This simplification not only streamlines the workflow but also enables the precise engineering of molecular structures by allowing the use of substituted benzenes, thus unlocking the potential for tailored flame retardants and photoinitiators with enhanced performance characteristics.
Mechanistic Insights into AlCl3-Catalyzed Electrophilic Substitution
The core of this technological breakthrough lies in the efficient generation of electrophilic phosphorus species under mild conditions. The reaction initiates when the aryl phosphine dichloride interacts with the aluminum chloride catalyst, facilitating the departure of a chloride ion to generate a highly reactive phosphonium cation (P+). This electrophilic center then attacks the electron-rich pi-system of the aromatic hydrocarbon substrate, forming a sigma-complex intermediate. Unlike the chaotic radical processes often seen at high temperatures, this controlled electrophilic substitution ensures high regioselectivity and minimizes the formation of tar-like by-products. The subsequent elimination of hydrogen chloride, mediated by the tetrachloroaluminate anion, stabilizes the diaryl phosphine chloride-catalyst complex, setting the stage for the final conversion.
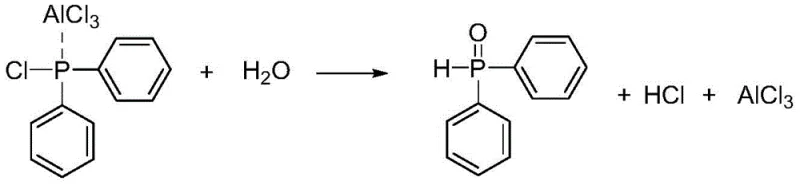
The final transformation involves a carefully controlled hydrolysis step that distinguishes this process from prior art. Instead of harsh chemical treatments, the reaction mixture is quenched in ice water, where the aluminum complex dissociates, and the phosphorus-chlorine bond is cleaved to form the phosphorus-oxygen double bond. This hydrolysis step is exothermic but manageable due to the low temperature of the quench medium, ensuring the integrity of sensitive functional groups on the aromatic rings. The result is a high-purity diarylphosphine oxide with minimal inorganic contamination, as the aluminum salts remain in the aqueous phase, simplifying downstream purification and reducing the need for extensive chromatographic separation.
How to Synthesize Substituted Diarylphosphine Oxide Efficiently
Implementing this synthesis requires precise control over stoichiometry and temperature gradients to maximize yield and purity. The process begins with the rigorous exclusion of moisture to prevent premature catalyst deactivation, followed by the sequential addition of reagents to manage the exotherm. While the general protocol is straightforward, optimizing the molar ratios of the aromatic substrate to the phosphine dichloride is critical for suppressing poly-substitution side reactions. For detailed operational parameters, safety guidelines, and specific workup procedures tailored to your facility's capabilities, please refer to the standardized synthesis protocol below.
- Combine aryl phosphine dichloride, aromatic hydrocarbon derivative, and anhydrous aluminum chloride catalyst in a reactor under nitrogen.
- Heat the mixture to 60-100°C and maintain reaction for 10-15 hours to form the diaryl phosphine chloride-catalyst complex.
- Cool the complex to 0-20°C, hydrolyze by dropping into ice-water, extract with organic solvent, and purify to obtain the final oxide.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this mild catalytic process offers profound strategic benefits beyond mere technical feasibility. By eliminating the high-temperature disproportionation step, manufacturers can significantly reduce energy utility costs and extend the lifespan of reactor vessels subjected to less thermal stress. The removal of the decomplexing agent requirement translates directly into a reduction in raw material procurement costs and a drastic decrease in hazardous solid waste generation, aligning production with increasingly stringent global environmental regulations. Furthermore, the versatility of the feedstock allows procurement teams to source a wider range of commercially available substituted benzenes, mitigating supply risks associated with specialized intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive decomplexing agents and the reduction in energy consumption due to lower operating temperatures lead to substantial operational expenditure savings. The simplified workup procedure reduces labor hours and solvent usage, further driving down the cost per kilogram of the final active ingredient, making it a highly competitive option for bulk polymer additive production.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as benzene, toluene, and xylenes, coupled with common Lewis acid catalysts, ensures a stable and resilient supply chain. Unlike processes dependent on exotic or single-source reagents, this method leverages widely available industrial feedstocks, minimizing the risk of production stoppages due to raw material shortages and ensuring consistent delivery schedules for downstream clients.
- Scalability and Environmental Compliance: The aqueous workup and absence of difficult-to-treat solid by-products make this process inherently scalable from pilot plant to multi-ton commercial production. The reduced environmental burden facilitates easier permitting and compliance with green chemistry initiatives, enhancing the corporate sustainability profile of manufacturers who adopt this technology for producing flame retardants and photoinitiators.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters and partners.
Q: What are the primary advantages of this new synthesis method over traditional disproportionation?
A: The new method operates at significantly lower temperatures (60-100°C vs >200°C), eliminates the need for complex decomplexing agents, and allows for the introduction of diverse substituents on the phenyl rings, which was impossible with the old disproportionation route.
Q: Can this process be scaled for industrial production of flame retardants?
A: Yes, the process uses common Lewis acid catalysts like aluminum chloride and standard aromatic feedstocks. The mild conditions and simple aqueous workup make it highly suitable for large-scale commercial manufacturing with reduced energy consumption.
Q: What is the typical purity and yield achievable with this technology?
A: Experimental data indicates HPLC purity levels exceeding 98% with isolated yields ranging from 75% to over 95%, depending on the specific substituents used on the aromatic rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diarylphosphine Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance phosphine oxides play in the next generation of flame-retardant materials and UV-curing systems. Our technical team has extensively analyzed the pathway described in CN110872321B and possesses the expertise to optimize this chemistry for your specific application needs. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our stringent purity specifications are met consistently through our rigorous QC labs and state-of-the-art analytical infrastructure.
We invite you to collaborate with us to leverage this advanced synthesis technology for your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and product quality.
