Advanced Chiral Phosphorus Ligands for High-Efficiency Asymmetric Hydrogenation and Commercial Scale-Up
The landscape of asymmetric synthesis is continually evolving, driven by the demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. Patent CN1599743A introduces a significant advancement in this field through the disclosure of novel chiral phosphorus compounds, specifically derivatives of 5H-dibenzo[a,d]cycloheptene, often referred to as TROPP ligands. These compounds are not merely incremental improvements but represent a structural breakthrough that allows for unprecedented tunability of steric and electronic environments around the catalytic center. For R&D directors and process chemists, this patent offers a robust toolkit for developing highly selective catalytic systems that can overcome the limitations of traditional phosphine ligands. The ability to modify the substitution pattern on the rigid dibenzo[a,d]cycloheptene backbone means that catalysts can be tailored for specific challenging substrates, ensuring high conversion rates and exceptional enantiomeric excess.
For procurement managers and supply chain heads, the implications of this technology extend beyond the laboratory bench. The synthesis pathways described rely on commercially available starting materials and standard chemical transformations, suggesting a viable route for cost-effective manufacturing at scale. By adopting these advanced ligands, manufacturers can potentially reduce the number of purification steps required to achieve desired purity levels, thereby streamlining the overall production process. This aligns perfectly with the industry's push towards more sustainable and efficient manufacturing practices, where reducing waste and energy consumption is paramount. As a reliable pharmaceutical intermediates supplier, understanding the nuances of such patented technologies is crucial for maintaining a competitive edge in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for asymmetric hydrogenation often rely on well-known phosphine ligands that, while effective for certain substrates, frequently struggle with broader applicability and tunability. A significant limitation lies in the difficulty of predicting catalytic activity and selectivity for new substrates, as the steric and electronic requirements vary drastically depending on the reaction type. Conventional ligands may lack the necessary rigidity or the specific spatial arrangement required to induce high levels of stereocontrol, leading to lower enantiomeric excess and necessitating costly downstream purification processes. Furthermore, many existing catalytic systems require high metal loadings or extreme reaction conditions to achieve acceptable conversion rates, which increases operational costs and complicates waste management. The inability to easily modify the ligand structure without synthesizing entirely new scaffolds limits the speed at which new catalytic solutions can be developed for emerging chemical challenges.
The Novel Approach
The novel approach presented in patent CN1599743A addresses these shortcomings by introducing a versatile family of phosphorus compounds based on the 5H-dibenzo[a,d]cycloheptene framework. This scaffold provides a rigid chiral environment that effectively transfers stereochemical information to the substrate during catalysis. Unlike conventional ligands, these novel compounds allow for independent variation of substituents at multiple positions, enabling precise tuning of the catalyst's electronic and steric properties to match specific reaction requirements. 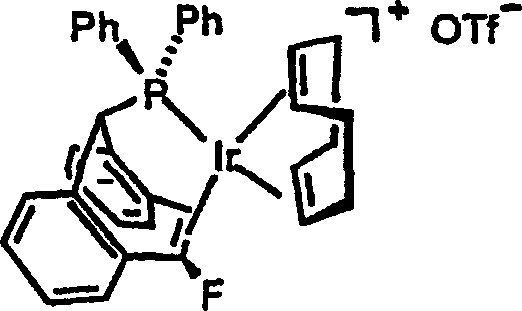 This structural flexibility facilitates the development of highly active catalysts capable of operating under milder conditions with lower metal loadings. The patent demonstrates that these ligands, particularly when complexed with Iridium, exhibit superior performance in the hydrogenation of challenging substrates like enamines and imines, achieving high conversions and stereoselectivity that were previously difficult to attain. This represents a paradigm shift towards more adaptable and efficient catalytic systems.
This structural flexibility facilitates the development of highly active catalysts capable of operating under milder conditions with lower metal loadings. The patent demonstrates that these ligands, particularly when complexed with Iridium, exhibit superior performance in the hydrogenation of challenging substrates like enamines and imines, achieving high conversions and stereoselectivity that were previously difficult to attain. This represents a paradigm shift towards more adaptable and efficient catalytic systems.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The mechanistic superiority of these novel ligands stems from their unique ability to form stable yet reactive transition metal complexes. When coordinated with Iridium, the phosphorus atoms of the TROPP ligands create a well-defined chiral pocket that guides the approach of the substrate. The rigid dibenzo[a,d]cycloheptene backbone minimizes conformational freedom, ensuring that the chiral information is consistently transmitted during the catalytic cycle. This rigidity is crucial for maintaining high enantioselectivity, as it prevents the formation of non-selective catalytic species that could lead to racemic mixtures. The electronic properties of the phosphorus center can be further modulated by the choice of substituents, allowing chemists to optimize the electron density at the metal center for specific bond activation steps. 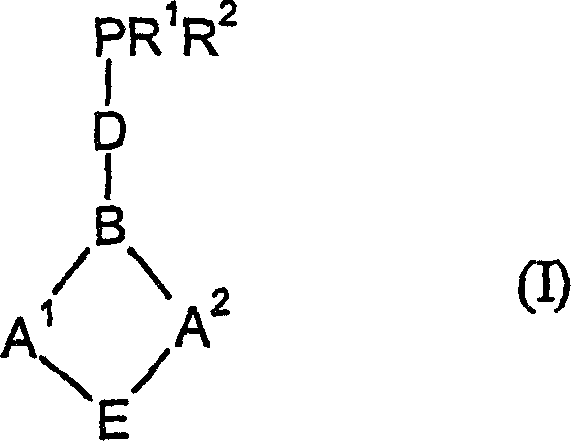 This level of control is essential for achieving the high turnover numbers and selectivity required in industrial applications, where even minor improvements in efficiency can translate to significant cost savings.
This level of control is essential for achieving the high turnover numbers and selectivity required in industrial applications, where even minor improvements in efficiency can translate to significant cost savings.
Furthermore, the impurity profile of reactions utilizing these ligands is typically cleaner due to the high specificity of the catalytic transformation. The well-defined coordination geometry reduces side reactions such as isomerization or over-reduction, which are common pitfalls in less selective systems. This results in a product stream that is easier to purify, reducing the burden on downstream processing units. For quality control teams, this means more consistent batch-to-batch reproducibility and a lower risk of failing stringent purity specifications. The ability to predict and control the outcome of the reaction through ligand design empowers process developers to scale up reactions with greater confidence, knowing that the fundamental chemistry is robust and reliable.
How to Synthesize 5H-Dibenzo[a,d]cycloheptene Derivatives Efficiently
The synthesis of these high-value ligands follows a logical and scalable sequence that begins with readily available ketone precursors. The process involves the reduction of substituted 5H-dibenzo[a,d]cyclohepten-5-ones to their corresponding alcohols, followed by conversion to reactive halides or sulfonates. These intermediates are then coupled with secondary phosphines to install the crucial phosphorus functionality. This modular approach allows for the introduction of diverse functional groups at various stages, facilitating the rapid generation of ligand libraries for screening. The detailed standardized synthesis steps see the guide below.
- Reduce substituted 5H-dibenzo[a,d]cyclohepten-5-ones to corresponding alcohols using borohydrides.
- Convert the resulting alcohols into halides or sulfonates using thionyl chloride or similar agents.
- React the halides with secondary phosphines to form the final chiral phosphorus ligand structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of these novel ligands offers substantial advantages for procurement and supply chain operations. The synthesis routes described in the patent utilize common reagents and standard unit operations, which simplifies the sourcing of raw materials and reduces dependency on exotic or scarce chemicals. This accessibility enhances supply chain reliability, ensuring that production schedules are not disrupted by material shortages. Additionally, the high efficiency of the resulting catalysts means that less metal is required per unit of product, directly lowering the cost of goods sold. The reduction in metal loading also simplifies the removal of residual metals from the final product, a critical step in pharmaceutical manufacturing that often requires expensive scavenging resins or additional purification stages.
- Cost Reduction in Manufacturing: The implementation of these highly active catalysts leads to significant cost reductions by minimizing the amount of precious metal required for each batch. Since the ligands enable high turnover numbers, the overall consumption of expensive Iridium or Rhodium salts is drastically lowered. Furthermore, the improved selectivity reduces the formation of by-products, which means less raw material is wasted and fewer resources are spent on separating impurities. This efficiency translates into a leaner manufacturing process with lower operational expenditures, making the production of complex chiral intermediates more economically viable without compromising on quality standards.
- Enhanced Supply Chain Reliability: The synthetic pathways for these ligands are robust and rely on established chemical transformations, which mitigates the risk of supply chain disruptions. Because the starting materials are commercially available or easily synthesized from bulk chemicals, manufacturers can secure long-term supply contracts with multiple vendors. This diversification of the supply base ensures continuity of supply even in volatile market conditions. Moreover, the scalability of the synthesis allows for rapid ramp-up of production capacity to meet surging demand, providing a strategic advantage in fulfilling large-scale orders for key pharmaceutical intermediates without lengthy lead times.
- Scalability and Environmental Compliance: Scaling these catalytic processes to industrial levels is facilitated by the mild reaction conditions and the stability of the catalyst systems. The ability to run reactions at lower temperatures and pressures reduces energy consumption and enhances safety profiles in large reactors. From an environmental standpoint, the high atom economy and reduced waste generation align with green chemistry principles, helping companies meet increasingly stringent regulatory requirements. The minimized use of hazardous solvents and the potential for solvent recycling further contribute to a smaller environmental footprint, positioning manufacturers as responsible stewards of sustainability in the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these novel ligand systems. They are derived from the specific technical details and beneficial effects outlined in the patent documentation, providing clarity for stakeholders evaluating this technology for their own processes. Understanding these aspects is vital for making informed decisions about integrating these advanced catalytic solutions into existing manufacturing workflows.
Q: What are the primary applications of these novel phosphorus ligands?
A: These ligands are primarily designed for asymmetric catalytic processes, specifically demonstrating high efficacy in the hydrogenation of enamines, enamides, and imines when complexed with transition metals like Iridium.
Q: How does the substitution pattern affect catalyst performance?
A: The patent highlights that the substitution pattern on the dibenzo[a,d]cycloheptene backbone allows for easy variation of steric and electronic properties, enabling fine-tuning for specific substrate conversions and improved stereoselectivity.
Q: Are these ligands suitable for large-scale industrial manufacturing?
A: Yes, the synthesis routes described utilize standard reagents and conditions such as reduction and nucleophilic substitution, which are amenable to scale-up from laboratory to commercial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phosphorus Ligands Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies disclosed in patent CN1599743A for the production of high-value chiral intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to full-scale manufacturing is seamless. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of ligand or intermediate meets the highest international standards. We understand that consistency and quality are non-negotiable in the pharmaceutical supply chain, and our infrastructure is designed to deliver exactly that.
We invite you to collaborate with us to leverage these advanced catalytic technologies for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs, demonstrating how these ligands can optimize your current processes. Please contact us to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with efficient, scalable, and cost-effective chemical solutions.
