Advanced Asymmetric Hydrogenation Technology for High-Purity Pharmaceutical Intermediates
Advanced Asymmetric Hydrogenation Technology for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust methodologies for synthesizing chiral intermediates, particularly for antihypertensive agents like renin inhibitors. Patent CN101090770B introduces a groundbreaking transition metal-catalyzed asymmetric hydrogenation process that addresses critical challenges in producing acrylic acid derivatives. This technology leverages a novel catalyst system comprising a transition metal from the ruthenium, rhodium, or iridium group, paired with a specific combination of chiral and achiral phosphorus ligands. By optimizing the coordination sphere of the metal center, this method achieves theoretical yields up to 100% and optical purities exceeding 99% ee, representing a significant leap forward for reliable pharmaceutical intermediate supplier capabilities. The ability to convert alpha-substituted cinnamic acid derivatives into corresponding chiral acids or esters with such high fidelity ensures that downstream drug synthesis maintains stringent quality standards required for global regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of unsaturated carboxylic acids has relied heavily on homogeneous catalysts utilizing chiral bis-tertiary diphosphine ligands. While literature such as WO 02/02500 describes the use of ferrocenyl-based structures like Walphos ligands to achieve optical yields around 95% ee, these systems suffer from inherent economic and logistical drawbacks. The synthesis of Walphos ligands is notoriously complex and costly, creating a bottleneck for cost reduction in API manufacturing. Furthermore, alternative approaches using monodentate ligands have demonstrated insufficient activity towards cinnamic acid derivatives, often necessitating prolonged hydrogenation times and resulting in poor enantiomeric excess. These inefficiencies translate directly into higher production costs and extended lead times, complicating the supply chain for high-purity pharmaceutical intermediates. Additionally, some prior art mixtures of monophosphorus compounds have shown significantly worse results in optical purity compared to optimized bidentate systems, leaving a gap for a more efficient solution.
The Novel Approach
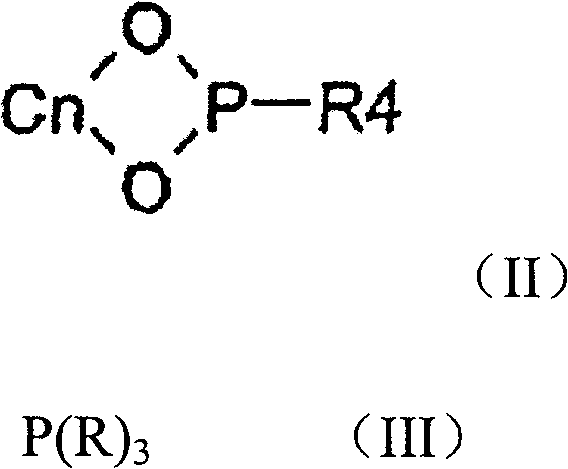
The innovation disclosed in CN101090770B circumvents these issues by employing a dual-ligand strategy that balances steric and electronic properties effectively. Instead of relying solely on expensive, complex chiral diphosphines, the process utilizes a mixture of a chiral phosphorus ligand of formula (II) and an achiral phosphine ligand of formula (III).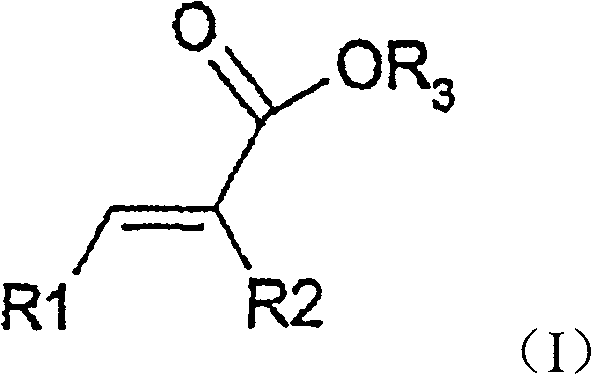
 This combination allows for fine-tuning the catalyst's active site, enabling the hydrogenation of compounds of formula (I) to formula (IV) with exceptional stereocontrol. The method is versatile, accommodating various substituents on the aromatic rings and alkyl chains, making it suitable for the commercial scale-up of complex polymer additives and specialty chemicals beyond just pharma. By avoiding the intricate synthesis of ferrocenyl backbones, this approach offers a streamlined pathway that significantly lowers the barrier to entry for producing high-value chiral building blocks.
This combination allows for fine-tuning the catalyst's active site, enabling the hydrogenation of compounds of formula (I) to formula (IV) with exceptional stereocontrol. The method is versatile, accommodating various substituents on the aromatic rings and alkyl chains, making it suitable for the commercial scale-up of complex polymer additives and specialty chemicals beyond just pharma. By avoiding the intricate synthesis of ferrocenyl backbones, this approach offers a streamlined pathway that significantly lowers the barrier to entry for producing high-value chiral building blocks.
Mechanistic Insights into Rh-Catalyzed Asymmetric Hydrogenation
The core of this technology lies in the synergistic interaction between the transition metal and the mixed ligand environment. When rhodium, preferably in the form of precursors like [Rh(COD)2]BF4, coordinates with both the chiral phosphorus ligand and the achiral phosphine, it forms a highly active catalytic species. The chiral ligand, often featuring a binaphthyl or similar backbone with phosphorus-oxygen bonds, dictates the primary stereochemical outcome by creating a chiral pocket. Simultaneously, the achiral phosphine, such as triphenylphosphine or substituted variants, modulates the electron density and steric bulk around the metal center. This modulation is critical for facilitating the oxidative addition of hydrogen and the subsequent migratory insertion into the carbon-carbon double bond of the substrate. The precise ratio of chiral to achiral ligand, typically ranging from 2.5:1 to 1.2:1, is essential for maximizing turnover frequency and enantioselectivity, ensuring that the reaction proceeds rapidly without compromising optical purity.
Impurity control is another vital aspect of this mechanistic design. In conventional single-ligand systems, competing non-selective pathways often lead to racemic byproducts or incomplete conversion, necessitating costly purification steps like recrystallization or chiral chromatography. However, the dual-ligand system described here suppresses these non-selective pathways effectively. Comparative data indicates that using chiral ligands alone can result in enantiomeric excess as low as 0-16%, whereas the addition of the achiral co-ligand boosts this dramatically to over 80% in test cases and up to 99% in optimized examples. This drastic improvement in selectivity minimizes the formation of unwanted diastereomers and racemates, simplifying the downstream isolation process. Consequently, the final product meets rigorous purity specifications with minimal waste generation, aligning with green chemistry principles and environmental compliance standards.
How to Synthesize Chiral Acrylic Acid Derivatives Efficiently
Implementing this hydrogenation protocol requires careful attention to reaction parameters to fully realize its potential benefits. The process is designed to be operationally simple, utilizing standard high-pressure equipment commonly found in fine chemical facilities. Operators must ensure the exclusion of oxygen to prevent catalyst deactivation, typically achieved through multiple purge cycles with inert gases like nitrogen. The choice of solvent plays a pivotal role, with mixtures of protic solvents like isopropanol and water proving particularly effective in enhancing enantiomeric purity. Detailed standardized synthesis steps see the guide below.
- Dissolve the acrylic acid derivative substrate, chiral phosphorus ligand, achiral phosphine ligand, and transition metal precursor in a solvent mixture such as isopropanol and water.
- Purge the reaction vessel with inert gas to remove oxygen, then heat the mixture to the desired reaction temperature between 20°C and 65°C.
- Pressurize the system with hydrogen gas to 10-150 bar, maintain stirring overnight, and isolate the final chiral product after cooling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalyst system presents compelling economic and operational advantages. The primary driver for cost optimization is the elimination of expensive, hard-to-source ferrocenyl ligands. By substituting these with more accessible chiral phosphorus ligands and commodity achiral phosphines, the raw material cost profile of the catalyst system is significantly improved. This reduction in input costs translates directly to a lower cost of goods sold (COGS) for the final API intermediate, allowing pharmaceutical companies to maintain healthier margins or offer more competitive pricing in tender processes. Furthermore, the high conversion rates observed, often reaching 100%, mean that there is minimal loss of valuable starting materials, maximizing atom economy and reducing the volume of waste that requires disposal.
- Cost Reduction in Manufacturing: The simplified ligand architecture removes the need for complex multi-step organic synthesis associated with traditional high-performance ligands. This structural simplicity not only lowers the purchase price of the catalyst components but also reduces the risk of supply disruptions caused by specialized manufacturing bottlenecks. Additionally, the high activity of the catalyst allows for lower catalyst loading ratios, potentially ranging from 1:1000 to 1:10000 relative to the substrate, which further drives down the cost per kilogram of product. The ability to operate at moderate temperatures and pressures also reduces energy consumption compared to more forcing conditions required by less active systems.
- Enhanced Supply Chain Reliability: Sourcing complex chiral ligands can often be a single point of failure in a supply chain. By utilizing a system based on more generic chemical building blocks, the risk of supply interruption is drastically minimized. The robustness of the reaction conditions, tolerating a range of substrates and solvent mixtures, ensures consistent production output even if minor variations in raw material quality occur. This reliability is crucial for maintaining continuous manufacturing schedules and meeting the just-in-time delivery expectations of major pharmaceutical clients. It effectively de-risks the procurement strategy for critical intermediates used in life-saving medications.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in autoclave setups that mimic industrial reactors. The use of isopropanol and water as solvents is particularly advantageous from an environmental, health, and safety (EHS) perspective, as these are less toxic and easier to recover than chlorinated or aromatic solvents. High enantioselectivity reduces the need for resource-intensive purification steps, thereby lowering the overall environmental footprint of the manufacturing process. This alignment with sustainability goals is increasingly important for multinational corporations aiming to reduce their carbon footprint and adhere to strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into existing production lines.
Q: How does the dual-ligand system improve enantioselectivity compared to single ligands?
A: The combination of a chiral phosphorus ligand with an achiral phosphine ligand creates a unique coordination environment around the transition metal center. This synergistic effect significantly enhances stereocontrol during the hydrogenation process, achieving optical purities up to 99% ee, whereas single ligand systems often result in poor enantiomeric excess or low conversion rates.
Q: What are the cost advantages of this catalyst system over ferrocenyl-based ligands?
A: Traditional ferrocenyl-based ligands like Walphos require complex multi-step syntheses, driving up raw material costs. The novel catalyst system utilizes simpler chiral phosphorus ligands combined with readily available achiral phosphines, drastically simplifying the supply chain and reducing the overall cost of goods for large-scale API intermediate production.
Q: Is this hydrogenation process scalable for industrial manufacturing?
A: Yes, the process operates under robust conditions using standard hydrogenation equipment like autoclaves. With conversion rates reaching 100% and the ability to use common solvents like isopropanol and water, the method is highly amenable to commercial scale-up from kilogram to multi-ton production without requiring exotic reagents or extreme conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acrylic Acid Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic and patent research into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in laboratory settings are faithfully reproduced on an industrial scale. We understand that consistency is key in the pharmaceutical sector; therefore, our stringent purity specifications and rigorous QC labs guarantee that every batch of chiral intermediates meets the highest international standards. Our infrastructure is designed to handle sensitive transition metal catalysis safely and efficiently, mitigating the risks associated with high-pressure hydrogenation.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and route feasibility assessments to demonstrate how this dual-ligand hydrogenation process can optimize your supply chain and enhance your product's market competitiveness. Let us be your partner in delivering high-quality, cost-effective solutions for the global healthcare market.
