Advanced Manufacturing of Difluprednate: A Cost-Effective 9-Step Synthetic Route
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value corticosteroids, and patent CN103509075A presents a transformative approach to manufacturing difluprednate. This specific intellectual property details a novel nine-step synthesis that fundamentally shifts the production paradigm from scarce, complex precursors to widely available, cost-effective starting materials. By utilizing hydrocortisone-21-acetate as the foundational building block, the process circumvents the significant supply chain bottlenecks associated with traditional routes that rely on difficult-to-source intermediates like anecortave acetate. The technical breakthrough lies not merely in the step count but in the strategic re-engineering of critical functionalization steps, particularly the C17 esterification and the dual fluorination sequence. This innovation addresses long-standing challenges in steroid chemistry regarding regioselectivity and reagent toxicity, offering a pathway that is both economically superior and environmentally compliant. For global supply chain leaders, this represents a viable strategy to secure long-term availability of this potent ophthalmic anti-inflammatory agent while mitigating the risks associated with hazardous chemical handling.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
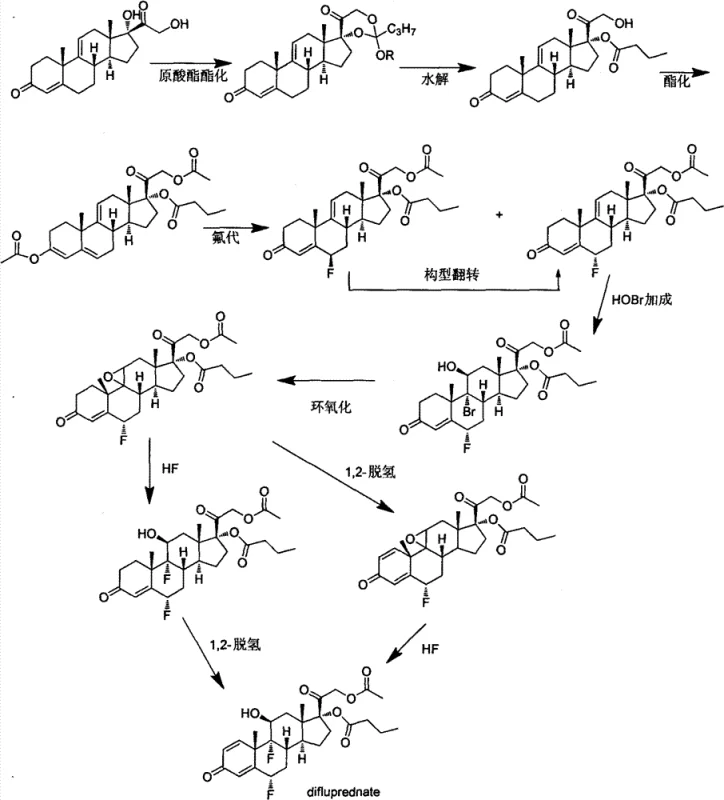
Historically, the synthesis of difluprednate has been plagued by reliance on obscure starting materials and hazardous reaction conditions that hinder scalability. As illustrated in the legacy pathways, earlier methods often commenced with anecortave acetate, a compound that is not commercially ubiquitous and requires a cumbersome seven-step synthesis itself. Even when attempting to derive this precursor from hydrocortisone acetate, the instability of the 17,21-dihydroxy structure during hydrolysis leads to significant rearrangement and the formation of unwanted 17-keto byproducts, drastically reducing overall throughput. Furthermore, conventional protocols necessitated the use of perchloryl fluoride and anhydrous hydrogen fluoride gas for fluorination steps. These reagents are not only extremely toxic and corrosive but also demand specialized, high-cost containment infrastructure, rendering the process economically unfeasible for standard multipurpose pharmaceutical plants. The multi-step protection and deprotection sequences required to install the C17 butyrate and C21 acetate groups further compounded these issues, introducing multiple points of failure and yield loss.
The Novel Approach
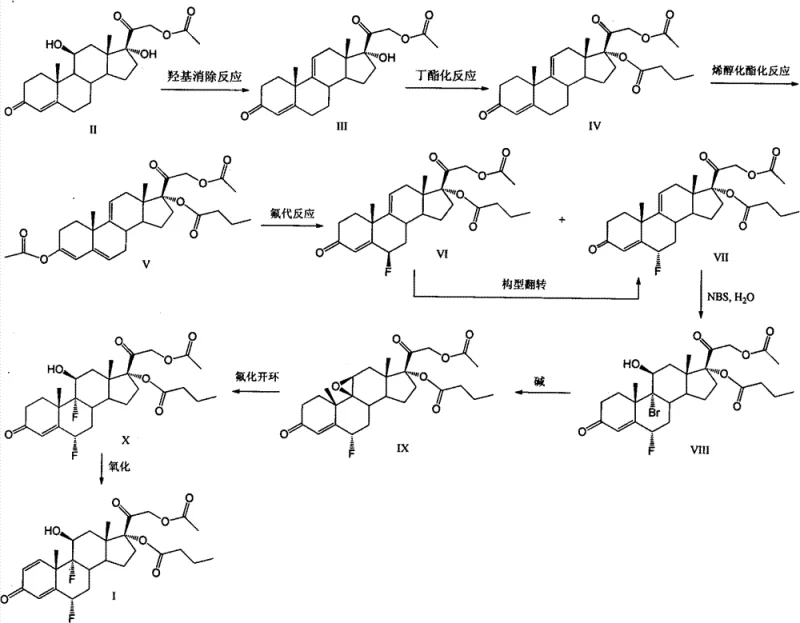
In stark contrast, the methodology disclosed in CN103509075A streamlines the entire operation by leveraging the inherent reactivity of the steroid nucleus more effectively. The new route initiates with the dehydration of hydrocortisone-21-acetate to form a 9,11-diene system, which serves as a stable platform for subsequent modifications. A pivotal innovation is the direct butyrylation at the C17 position, which proceeds with quantitative efficiency, completely eliminating the need for the迂回 (detour) orthoesterification and hydrolysis steps that characterized prior art. This simplification not only shortens the timeline but also removes the generation of difficult-to-separate isomers. Moreover, the substitution of gaseous fluorinating agents with solid, manageable reagents like Selectfluor and Olah's reagent marks a significant safety advancement. This modernized workflow ensures that the critical fluorine atoms are installed with high precision under mild conditions, facilitating a smoother transition from laboratory bench to commercial reactor without the need for exotic equipment.
Mechanistic Insights into Selective Fluorination and Esterification
The core chemical ingenuity of this process resides in the precise control of stereochemistry and regioselectivity during the fluorination and esterification stages. The synthesis employs an enolization-acetylation strategy to shift the double bond in the A-ring, effectively activating the C6 position for electrophilic attack. This activation allows for the use of Selectfluor, a stable electrophilic fluorinating agent, to introduce the first fluorine atom with high specificity. Following this, the configuration is inverted to ensure the correct alpha-orientation. The second fluorination at the C9 position is achieved through a bromohydrin intermediate formed via N-bromosuccinimide addition, followed by epoxidation and ring-opening with Olah's reagent. This sequence is mechanistically superior because Olah's reagent (hydrogen fluoride-pyridine complex) provides a controlled source of nucleophilic fluoride that opens the epoxide ring with inversion, installing the 9-alpha-fluorine without the violent exotherms associated with anhydrous HF gas. This controlled mechanism minimizes side reactions and degradation of the sensitive steroid backbone.
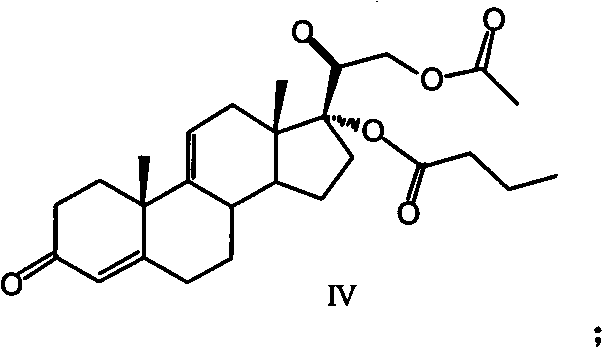
Furthermore, the establishment of the C17 butyrate ester is achieved through a direct acylation mechanism that exploits the steric environment of the 9,11-diene intermediate. In traditional saturated steroids, differentiating between the C17 and C21 hydroxyl groups is notoriously difficult, often requiring bulky protecting groups. However, the specific conformational properties of the 9,11-diene system in Compound III allow the C17 hydroxyl to react preferentially with butyric anhydride in the presence of a catalyst like DMAP. This results in the formation of Compound IV, a key intermediate that possesses the correct ester pattern early in the synthesis. By locking in this structural feature at step two, the process avoids the scrambling of ester groups that typically occurs during later-stage manipulations. This mechanistic foresight ensures that the final product, difluprednate, is obtained with a cleaner impurity profile, reducing the burden on downstream purification processes.
How to Synthesize Difluprednate Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly temperature control during the fluorination and dehydration steps to maintain stereochemical integrity. The process is designed to be telescoped where possible, minimizing the isolation of unstable intermediates and reducing solvent consumption. Operators should prioritize the quality of the Selectfluor reagent and ensure strict anhydrous conditions during the initial enolization to prevent hydrolysis of the acetate groups. The final dehydrogenation step using DDQ must be monitored closely via TLC to prevent over-oxidation of the steroid nucleus. Detailed standard operating procedures regarding reagent stoichiometry, quenching protocols, and crystallization conditions are essential for reproducibility. For a comprehensive breakdown of the specific reaction conditions, solvent systems, and workup procedures for each of the nine steps, please refer to the standardized guide below.
- Dehydrate hydrocortisone-21-acetate using methanesulfonyl chloride to form the 9,11-diene intermediate.
- Perform direct 17-position butyrylation using butyric anhydride and DMAP to quantitatively obtain the key intermediate Compound IV.
- Execute sequential fluorination at positions 6 and 9 using Selectfluor and Olah's reagent, followed by DDQ dehydrogenation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial advantages that directly impact the bottom line and supply security for pharmaceutical manufacturers. The shift to hydrocortisone-21-acetate as the starting material leverages a commodity chemical that is produced globally in massive quantities, ensuring a stable and competitive pricing structure compared to the bespoke precursors required by older methods. The elimination of toxic gas handling infrastructure significantly lowers the capital expenditure required for plant setup and reduces ongoing operational costs related to safety compliance and waste disposal. By simplifying the synthetic sequence and improving yields at critical junctions, the overall material throughput is enhanced, allowing for greater production capacity within existing facilities. These factors combine to create a manufacturing profile that is resilient against raw material volatility and regulatory pressure.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the drastic simplification of the C17 esterification step. By achieving quantitative yield in a single direct reaction, the process eliminates the material losses and solvent costs associated with the multi-step orthoester protection and deprotection cycles found in legacy patents. Additionally, the replacement of expensive and hazardous gaseous fluorinating agents with solid, shelf-stable reagents like Selectfluor reduces procurement complexity and storage costs. The higher overall yield means less raw material is required per kilogram of final API, leading to significant variable cost savings. Furthermore, the reduced generation of hazardous waste lowers the environmental compliance costs, contributing to a leaner and more profitable production model.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of hydrocortisone-21-acetate, a widely available bulk steroid intermediate with a mature global supply network. Unlike the obscure anecortave acetate or the undefined 6-alpha-fluoro-isofluprednisolone used in previous methods, this starting material is not subject to single-source bottlenecks. The robustness of the reaction conditions, which tolerate standard industrial solvents and do not require cryogenic temperatures for most steps, ensures that production can be maintained across diverse geographic locations without specialized infrastructure. This flexibility allows procurement teams to diversify their manufacturing base, reducing the risk of disruption due to regional logistical issues or facility maintenance.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing reagents and conditions that are compatible with standard stainless steel reactors. The avoidance of anhydrous hydrogen fluoride gas removes a major safety barrier to large-scale production, as it eliminates the need for specialized nickel-lined or monel equipment. The use of Olah's reagent and Selectfluor allows for safer handling and easier quenching of reaction mixtures, simplifying the waste treatment process. This alignment with green chemistry principles not only facilitates regulatory approval in stringent markets but also future-proofs the manufacturing site against increasingly tight environmental regulations regarding volatile organic compounds and hazardous emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this difluprednate synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of the supply source. The answers reflect the specific improvements in safety, yield, and operational simplicity that distinguish this method from historical precedents.
Q: What is the primary advantage of this new difluprednate synthesis route?
A: The primary advantage is the use of readily available hydrocortisone-21-acetate as a starting material and the elimination of toxic gases like perchloryl fluoride, replaced by safer solid reagents like Selectfluor.
Q: How does this process improve yield at the C17 position?
A: It achieves quantitative yield at the C17 position by using direct butyrylation on the 9,11-diene intermediate, avoiding the low-yield orthoesterification and hydrolysis detours found in legacy methods.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions, common solvents, and avoids extremely hazardous anhydrous hydrogen fluoride gas, making it significantly safer and more scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluprednate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient API manufacturing processes like the one described in CN103509075A. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel route are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of difluprednate meets the highest international pharmacopeial standards. Our commitment to process optimization allows us to deliver high-quality intermediates and APIs that support the development of next-generation ophthalmic therapies.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this greener, more efficient method. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to be your long-term, reliable partner in the production of complex steroid pharmaceuticals.
