Advanced Chiral Ligand Technology for Scalable Asymmetric Catalysis and Commercial Production
The landscape of asymmetric synthesis is continuously evolving, driven by the demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. Patent CN1791607A introduces a significant breakthrough in this domain by disclosing a novel class of chiral nitrogen-phosphorus compounds and their corresponding transition metal complexes. These ligands are specifically engineered to overcome the limitations of traditional systems, offering enhanced steric and electronic properties that are crucial for inducing high enantioselectivity. For R&D directors and process chemists, this technology represents a pivotal shift towards more efficient catalytic systems that can operate under milder conditions while delivering superior stereocontrol. The ability to tune the ligand environment around the metal center allows for precise optimization of reaction pathways, directly impacting the purity profiles of complex active pharmaceutical ingredients (APIs).
From a supply chain perspective, the commercial viability of these ligands is underpinned by their synthetic accessibility. Unlike many specialized chiral ligands that require lengthy, multi-step resolutions or expensive chiral pool starting materials, the methodology outlined in this patent leverages abundant bicyclic scaffolds such as camphor derivatives. This foundational advantage translates into a more robust and reliable supply chain for high-purity pharmaceutical intermediates. By utilizing straightforward coupling reactions and standard reduction techniques, manufacturers can mitigate the risks associated with raw material scarcity. Consequently, procurement managers can anticipate a stabilization of costs and lead times, ensuring continuous production flows for critical downstream applications in drug discovery and development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of effective chiral ligands for asymmetric catalysis has been plagued by significant structural and economic challenges. Conventional ligand systems often suffer from conformational flexibility, which leads to a lack of rigidity in the catalyst-substrate complex. This flexibility can result in poor stereoinduction, necessitating extensive screening of reaction conditions to achieve acceptable enantiomeric excess levels. Furthermore, many established ligands require complex synthetic routes involving multiple protection and deprotection steps, which drastically increase the overall cost of goods sold (COGS). The reliance on scarce chiral building blocks or difficult resolution processes further exacerbates supply chain vulnerabilities, making it difficult for manufacturers to guarantee consistent quality and availability for large-scale commercial production.
The Novel Approach
The innovative strategy presented in CN1791607A addresses these deficiencies by introducing a rigid bicyclic framework that locks the nitrogen and phosphorus donor atoms into a specific spatial arrangement. This structural rigidity minimizes conformational freedom, thereby creating a well-defined chiral pocket around the transition metal center. The result is a catalyst system that exhibits remarkable consistency in enantioselectivity across a broad range of substrates. Additionally, the synthetic route is streamlined, utilizing efficient palladium-catalyzed cross-coupling reactions to assemble the core structure. This approach not only simplifies the manufacturing process but also enhances the scalability of the ligand production. By reducing the number of synthetic steps and avoiding cumbersome purification protocols, this novel method offers a clear pathway for cost reduction in pharmaceutical intermediates manufacturing.
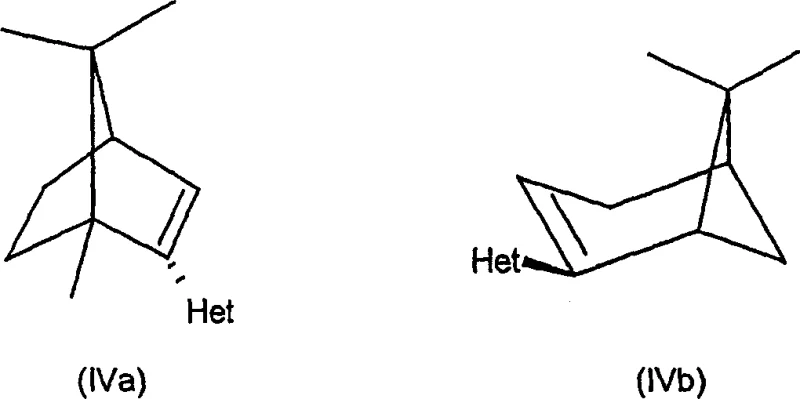
Mechanistic Insights into Nitrogen-Phosphorus Ligand Coordination
The efficacy of these chiral ligands stems from their unique ability to coordinate with transition metals such as Iridium and Palladium in a bidentate fashion. The nitrogen atom, typically part of a pyridyl or quinolyl moiety, and the phosphorus atom work in concert to stabilize the metal center while imposing a specific chiral environment. The bicyclic backbone, derived from structures like pinene or camphor, provides substantial steric bulk that effectively shields one face of the metal complex. This steric hindrance forces the incoming substrate to approach from a specific trajectory, thereby dictating the stereochemical outcome of the reaction. For R&D teams, understanding this mechanistic nuance is critical for optimizing catalyst loading and reaction parameters to maximize turnover numbers and minimize waste generation during process development.
Furthermore, the electronic properties of the ligand can be finely tuned by modifying the substituents on the phosphorus atom or the heteroaryl ring. This tunability allows chemists to adjust the electron density at the metal center, which is particularly important for reactions involving oxidative addition or reductive elimination steps. In asymmetric hydrogenation, for instance, the electron-rich nature of certain phosphine variants can accelerate the activation of molecular hydrogen, leading to faster reaction rates without compromising selectivity. This level of control over the catalytic cycle ensures that impurities arising from side reactions are minimized, resulting in a cleaner crude product profile that simplifies downstream processing and purification efforts significantly.
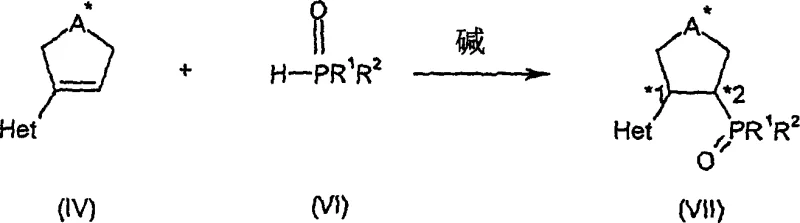
How to Synthesize Chiral Nitrogen-Phosphorus Ligands Efficiently
The synthesis of these high-value ligands follows a logical and scalable sequence that begins with the functionalization of readily available bicyclic ketones. The initial step involves the conversion of the ketone into a vinyl triflate, which serves as an excellent electrophile for subsequent cross-coupling reactions. This is followed by a palladium-catalyzed coupling with a heteroaryl zinc reagent to install the nitrogen-containing arm of the ligand. The final stages involve the introduction of the phosphorus moiety through a nucleophilic substitution or addition-elimination sequence, followed by reduction to the active phosphine state. Detailed standardized synthesis steps are provided in the guide below to assist process chemists in replicating these results.
- Convert bicyclic ketones (e.g., camphor) to vinyl triflates using LDA and N-phenyltrifluoromethanesulfonimide at low temperatures.
- Perform palladium-catalyzed cross-coupling with heteroaryl zinc reagents to form the key alkene intermediate.
- React the intermediate with secondary phosphine oxides using potassium tert-butoxide in DMSO, followed by silane reduction to yield the final ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ligand technology offers tangible strategic benefits beyond mere technical performance. The primary advantage lies in the simplification of the supply chain for chiral catalysts. Because the ligands are synthesized from abundant natural products like camphor, the risk of raw material bottlenecks is significantly reduced compared to ligands relying on synthetic chiral pools. This abundance ensures a stable supply base, allowing for long-term contracting and better inventory planning. Moreover, the robustness of the synthetic route means that production can be scaled up rapidly to meet fluctuating market demands without the need for specialized equipment or hazardous reagents that often complicate regulatory compliance.
- Cost Reduction in Manufacturing: The streamlined synthetic pathway eliminates several costly steps associated with traditional ligand synthesis, such as extensive chromatographic purifications or cryogenic reactions. By utilizing standard coupling conditions and common solvents like DMSO and toluene, the operational expenditure for manufacturing is substantially lowered. Additionally, the high catalytic efficiency of the resulting metal complexes means that lower catalyst loadings can be used to achieve full conversion, further reducing the consumption of precious metals like Iridium and Palladium. This cumulative effect leads to significant cost savings in the overall production of chiral intermediates.
- Enhanced Supply Chain Reliability: The use of commercially available starting materials and reagents ensures that the supply chain is resilient to disruptions. Unlike proprietary ligands that may be sourced from a single vendor, the generic nature of the building blocks allows for multi-sourcing strategies, enhancing negotiation power and security of supply. The stability of the ligand precursors also facilitates easier storage and transportation, reducing the logistical complexities and costs associated with temperature-controlled shipping. This reliability is crucial for maintaining continuous manufacturing operations in the fast-paced pharmaceutical industry.
- Scalability and Environmental Compliance: The process described in the patent is inherently green, utilizing fewer steps and generating less waste compared to conventional methods. The avoidance of heavy metal contaminants in the ligand synthesis phase simplifies the purification of the final API, aligning with stringent regulatory guidelines regarding residual metals. Furthermore, the high atom economy of the coupling reactions contributes to a more sustainable manufacturing footprint. This environmental compatibility not only reduces waste disposal costs but also supports corporate sustainability goals, making the technology attractive for companies aiming to reduce their ecological impact while maintaining high production standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these aspects helps stakeholders evaluate the feasibility of integrating this chemistry into their existing workflows.
Q: What are the primary advantages of these chiral ligands over conventional systems?
A: The ligands described in CN1791607A offer superior steric and electronic tunability due to their rigid bicyclic backbone. This results in higher enantioselectivity and conversion rates in asymmetric hydrogenation and allylic substitution compared to flexible acyclic ligands.
Q: Can these ligands be scaled for industrial manufacturing?
A: Yes, the synthesis utilizes readily available starting materials like camphor and standard coupling reagents. The process avoids exotic catalysts in the ligand synthesis step, making it highly suitable for commercial scale-up from kilograms to metric tons.
Q: Which transition metals are most compatible with these ligands?
A: The patent highlights exceptional performance with Iridium (Ir) for asymmetric hydrogenation and hydroboration, and Palladium (Pd) for allylic substitution reactions, achieving enantiomeric excess values exceeding 95% in optimized conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ligands Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in accelerating drug development and commercialization. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. We are committed to delivering products with stringent purity specifications and supporting our clients with rigorous QC labs that validate every batch against the highest industry standards. Our infrastructure is designed to handle the complexities of chiral synthesis, providing a secure and efficient partner for your long-term supply needs.
We invite you to collaborate with us to leverage these cutting-edge ligand systems for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific process requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your production efficiency and reduce time-to-market for your critical pharmaceutical intermediates.
