Advanced Chiral Phosphorus Ligands for Scalable Asymmetric Catalysis Manufacturing
The chemical industry continuously seeks advancements in asymmetric catalysis to produce high-purity chiral intermediates essential for modern pharmaceuticals and agrochemicals. Patent CN100349905C introduces a significant breakthrough in this domain by disclosing novel phosphorus compounds, specifically ligands derived from the 5H-dibenzo[a,d]suberene scaffold, often referred to as Tropp ligands. These compounds address the persistent challenge of achieving high stereoselectivity and catalytic activity in transition metal-catalyzed reactions. Unlike traditional ligands that may suffer from rigidity or limited electronic tunability, the structures described in this patent offer a versatile platform for optimizing catalytic performance. The invention encompasses not only the ligands themselves but also their preparation methods and their use in forming active transition metal complexes, particularly with iridium and rhodium. For R&D directors and technical procurement specialists, understanding the underlying chemical architecture is crucial for evaluating the feasibility of integrating these materials into existing synthetic pathways for API intermediates and fine chemicals.
The commercial implications of this technology extend beyond mere academic interest, offering tangible benefits for supply chain stability and cost management in fine chemical manufacturing. By enabling more efficient asymmetric hydrogenation processes, these ligands can potentially reduce the reliance on expensive resolution steps or less efficient catalytic systems. The patent details a wide range of substituents that can be incorporated into the ligand framework, allowing for precise customization to match specific substrate requirements. This adaptability is vital for process chemists who need to tailor catalysts for complex molecule synthesis. Furthermore, the ability to form stable complexes with various transition metals ensures that the technology can be applied across a broad spectrum of chemical transformations. As a reliable supplier of advanced catalytic materials, recognizing the potential of such patented technologies allows companies to stay ahead in the competitive landscape of specialty chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional asymmetric catalysis often relies on well-established ligand classes such as BINAP or DuPhos, which, while effective, possess inherent structural limitations that can restrict their utility in certain challenging transformations. The rigid binaphthyl backbone of BINAP, for instance, provides a defined chiral environment but lacks the conformational flexibility needed to accommodate bulky or electronically diverse substrates efficiently. This rigidity can lead to suboptimal binding geometries with the metal center, resulting in lower turnover frequencies or reduced enantiomeric excess in difficult hydrogenation reactions. Additionally, the synthesis of some conventional chiral ligands can be costly and involve multiple steps with low overall yields, impacting the economic viability of large-scale processes. Process chemists frequently encounter scenarios where standard ligands fail to provide the necessary selectivity for specific imine or enamine substrates, necessitating time-consuming screening campaigns or the development of entirely new synthetic routes. These inefficiencies translate directly into increased production costs and extended lead times for bringing new drug candidates to market.
The Novel Approach
The novel approach presented in patent CN100349905C utilizes a dibenzo[a,d]suberene backbone which offers a unique combination of steric bulk and conformational adaptability that overcomes many drawbacks of conventional systems. The seven-membered ring system in the Tropp scaffold allows for a degree of flexibility that enables the ligand to adjust its geometry around the metal center, optimizing the chiral pocket for a wider variety of substrates. This structural feature is particularly advantageous for the asymmetric hydrogenation of functionally diverse groups such as imines and alkene acid amides, where precise spatial arrangement is critical for high stereoselectivity. Furthermore, the synthetic accessibility of the dibenzo[a,d]suberene core allows for extensive derivatization, enabling chemists to fine-tune the electronic properties of the phosphorus atoms through the introduction of various aryl or alkyl substituents. This tunability means that a single ligand family can be adapted to solve multiple catalytic challenges, reducing the need for entirely different catalyst systems for different steps in a synthesis sequence. The result is a more streamlined and robust catalytic process that enhances overall manufacturing efficiency.
Mechanistic Insights into Tropp Ligand Catalytic Cycles
Understanding the mechanistic behavior of these novel phosphorus ligands is essential for maximizing their performance in industrial applications. The core structure, as illustrated in the general formula, features a phosphorus atom attached to a chiral or pro-chiral dibenzo[a,d]suberene framework, which dictates the stereochemical outcome of the reaction. ![General Formula I of novel phosphorus compounds showing the dibenzo[a,d]suberene backbone and phosphorus substituents](/insights/img/chiral-phosphorus-ligands-asymmetric-catalysis-supplier-20260306190720-01.webp) . When coordinated with transition metals like iridium or rhodium, the ligand creates a specific chiral environment that directs the approach of the substrate to the metal center. The steric bulk provided by the fused aromatic rings prevents unwanted side reactions and stabilizes the active catalytic species against decomposition. Electronic modulation is achieved through the substituents on the phosphorus atom, which can be adjusted to influence the electron density at the metal center, thereby affecting the rate of oxidative addition and reductive elimination steps in the catalytic cycle. This precise control over the metal-ligand interaction is what allows for the high enantiomeric excesses observed in the hydrogenation of prochiral imines and enamines.
. When coordinated with transition metals like iridium or rhodium, the ligand creates a specific chiral environment that directs the approach of the substrate to the metal center. The steric bulk provided by the fused aromatic rings prevents unwanted side reactions and stabilizes the active catalytic species against decomposition. Electronic modulation is achieved through the substituents on the phosphorus atom, which can be adjusted to influence the electron density at the metal center, thereby affecting the rate of oxidative addition and reductive elimination steps in the catalytic cycle. This precise control over the metal-ligand interaction is what allows for the high enantiomeric excesses observed in the hydrogenation of prochiral imines and enamines.
Impurity control is another critical aspect where the mechanistic understanding of these ligands provides significant value. In asymmetric catalysis, the formation of the wrong enantiomer or byproducts often stems from non-selective background reactions or the presence of inactive catalyst isomers. The Tropp ligands described in the patent are designed to form well-defined complexes that minimize the presence of such inactive species. The rigidity of the phosphorus-binding site combined with the flexibility of the backbone ensures that the substrate binds in a highly specific orientation, reducing the likelihood of competing reaction pathways. Moreover, the stability of the resulting metal complexes under reaction conditions means that catalyst degradation is minimized, leading to cleaner reaction profiles and easier downstream purification. For manufacturing teams, this translates to higher purity final products with reduced levels of chiral impurities, which is a stringent requirement for regulatory compliance in pharmaceutical production. The ability to predict and control these mechanistic factors is a key advantage of adopting this advanced ligand technology.
How to Synthesize Tropp Ligands Efficiently
The synthesis of these high-performance ligands follows a logical sequence of organic transformations that are well-suited for scale-up. The process typically begins with the preparation of the dibenzo[a,d]suberene ketone precursor, which is then reduced to the corresponding alcohol using standard reducing agents. This alcohol is subsequently converted into a leaving group, such as a chloride, before reacting with a secondary phosphine to install the catalytic center. 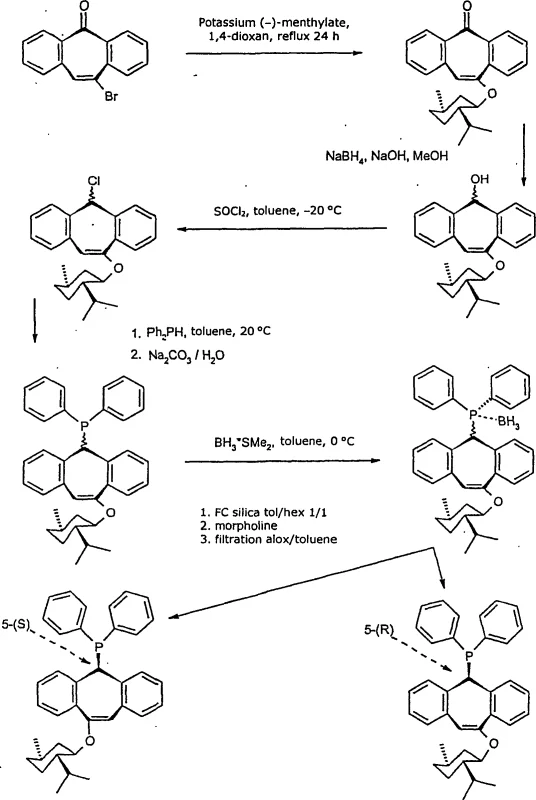 . The detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating the process.
. The detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating the process.
- Reduction of substituted 5H-dibenzo[a,d]suberene-5-ketone precursors using sodium borohydride or aluminum isopropoxide to form the corresponding alcohol intermediates.
- Conversion of the alcohol intermediate into a leaving group, typically a chloride or sulfonate, using thionyl chloride or similar halogenating agents under controlled temperatures.
- Nucleophilic substitution reaction with secondary phosphines or lithium phosphides to install the phosphorus functionality, followed by purification and optional metal complexation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ligand technology presents several strategic advantages that align with goals of cost reduction and supply reliability. The synthetic route for these ligands avoids the use of extremely rare or prohibitively expensive starting materials, relying instead on commercially available aromatic precursors and standard phosphorus reagents. This accessibility ensures that the supply chain for the ligands themselves is robust and less susceptible to geopolitical or market fluctuations that often affect specialized fine chemicals. Furthermore, the high efficiency of the catalysts formed with these ligands means that lower catalyst loadings can often be employed to achieve the same conversion rates as less active systems. This reduction in metal usage directly correlates to lower raw material costs and simplified waste management, as there is less heavy metal residue to remove from the final product. The overall process intensification enabled by these catalysts supports a more sustainable and economically viable manufacturing model.
- Cost Reduction in Manufacturing: The implementation of these novel ligands facilitates significant cost savings by enhancing the efficiency of asymmetric hydrogenation steps, which are often bottlenecks in synthesis. By achieving higher turnover numbers and better selectivity, the need for costly chromatographic separations or recrystallization steps to remove unwanted isomers is drastically reduced. The elimination of inefficient resolution processes further contributes to substantial cost savings, as the entire batch of product can be utilized rather than discarding the unwanted enantiomer. Additionally, the stability of the catalysts allows for longer operational lifetimes, reducing the frequency of catalyst replenishment and associated downtime. These factors combine to lower the overall cost of goods sold for complex chiral intermediates, providing a competitive edge in pricing strategies for downstream pharmaceutical clients.
- Enhanced Supply Chain Reliability: Securing a stable supply of high-quality chiral ligands is critical for maintaining continuous production schedules. The synthetic methodology described in the patent utilizes robust chemical transformations that are less sensitive to minor variations in reaction conditions, ensuring consistent batch-to-batch quality. This reliability reduces the risk of production delays caused by out-of-specification materials. Moreover, the versatility of the ligand scaffold means that a single manufacturing line can potentially produce a variety of ligand derivatives by simply changing the phosphine substituent, offering flexibility in responding to changing customer demands. This adaptability strengthens the supply chain by reducing dependency on multiple specialized suppliers and consolidating sourcing strategies. Consequently, procurement teams can negotiate better terms and ensure long-term availability of critical catalytic materials.
- Scalability and Environmental Compliance: Scaling asymmetric catalytic processes from the laboratory to commercial production often poses significant challenges, but the chemistry underlying these ligands is inherently scalable. The reactions proceed under moderate conditions and do not require extreme pressures or temperatures that would necessitate specialized high-cost equipment. From an environmental perspective, the high selectivity of the catalysts minimizes the generation of chemical waste, aligning with green chemistry principles and reducing the burden on waste treatment facilities. The reduced use of heavy metals also simplifies compliance with increasingly stringent environmental regulations regarding metal residues in pharmaceutical products. This alignment with sustainability goals not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturing operation, appealing to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ligand technology in industrial settings. These answers are derived from the specific technical disclosures and experimental data found within the patent documentation, providing a factual basis for decision-making. Understanding these details helps bridge the gap between theoretical potential and practical application, ensuring that all stakeholders have a clear view of the technology's capabilities and limitations. It is important to consult with technical experts when adapting these general principles to specific production environments.
Q: What are the primary advantages of Tropp ligands over conventional BINAP ligands?
A: Tropp ligands based on the dibenzo[a,d]suberene scaffold offer greater conformational flexibility and tunable electronic properties compared to the rigid binaphthyl backbone of BINAP, allowing for optimized stereoselectivity in challenging hydrogenation reactions.
Q: Can these ligands be scaled for industrial production?
A: Yes, the synthetic routes described involve standard organic transformations such as reduction, chlorination, and phosphination which are amenable to large-scale manufacturing with appropriate safety controls for phosphine handling.
Q: What types of substrates are compatible with these catalytic systems?
A: These iridium and rhodium complexes are particularly effective for the asymmetric hydrogenation of imines, enamine, and alkene acid amides, which are critical intermediates in pharmaceutical and agrochemical synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tropp Ligands Supplier
NINGBO INNO PHARMCHEM stands ready to support your transition to advanced catalytic technologies with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team of expert chemists understands the nuances of handling sensitive phosphorus compounds and transition metal complexes, ensuring that every batch meets stringent purity specifications required for GMP manufacturing. We operate rigorous QC labs equipped with state-of-the-art analytical instruments to verify the enantiomeric excess and chemical purity of our ligands and catalysts. Our commitment to quality assurance means that you can rely on us for consistent supply without compromising on the performance of your critical synthetic steps. We view ourselves as a strategic partner in your R&D and manufacturing success, offering more than just a product but a comprehensive solution for your chiral synthesis needs.
We invite you to contact our technical procurement team to discuss how these novel ligands can be integrated into your specific processes. We are prepared to provide a Customized Cost-Saving Analysis that evaluates the potential economic impact of switching to this technology for your existing product lines. Please reach out to request specific COA data and route feasibility assessments tailored to your target molecules. Our goal is to demonstrate clearly how partnering with us can drive efficiency and innovation in your supply chain. Let us help you unlock the full potential of asymmetric catalysis for your next generation of pharmaceutical and agrochemical products.
