Advanced Manufacturing of Terbutaline Sulfate: A Safer, Scalable Route for Global Pharma Supply Chains
Advanced Manufacturing of Terbutaline Sulfate: A Safer, Scalable Route for Global Pharma Supply Chains
The pharmaceutical industry constantly seeks robust, scalable, and safe synthetic pathways for critical active pharmaceutical ingredients (APIs), and the preparation of Terbutaline Sulfate stands as a prime example of process innovation. As detailed in the recent patent CN110950765A, a novel four-step synthesis method has been developed that fundamentally addresses the safety and efficiency bottlenecks plaguing traditional manufacturing routes. This technical breakthrough utilizes 3,5-dihydroxy acetophenone as a cost-effective starting material, employing a strategic sequence of benzyl protection, copper-mediated bromination, DMSO oxidation, and reductive amination. For R&D directors and supply chain leaders, this patent represents a significant shift away from hazardous reagents like selenium dioxide and methyllithium, offering a pathway that is not only chemically elegant but also commercially viable for high-purity API intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Terbutaline Sulfate has been fraught with significant technical and safety challenges that hinder efficient commercial scale-up. Prior art methods, such as those reported by early developers, often relied on the use of 3,5-dihydroxybenzoic acid, necessitating a lengthy sequence of esterification, protection, hydrolysis, and acyl chlorination before even reaching the ketone stage. More critically, established routes frequently employed highly toxic oxidants like selenium dioxide or dangerously flammable metal reagents such as methyllithium to construct the necessary carbon framework. These reagents pose severe occupational health risks, require specialized containment infrastructure, and generate complex waste streams that are costly to treat. Furthermore, direct oxidation methods using DMSO without proper activation often suffered from uncontrolled side reactions, leading to poor product purity and difficult downstream purification processes that eroded overall process economics.
The Novel Approach
The methodology disclosed in patent CN110950765A introduces a streamlined and inherently safer strategy that bypasses these historical pitfalls by optimizing the reaction sequence and reagent selection. By starting directly with 3,5-dihydroxy acetophenone, the process eliminates several upstream steps associated with carboxylic acid manipulation, thereby shortening the overall timeline and reducing material loss. The core innovation lies in the controlled bromination using copper bromide followed by a mild oxidation with dimethyl sulfoxide (DMSO) catalyzed by alkali, which effectively converts the ketone to the crucial aldehyde intermediate without the toxicity of selenium compounds. This approach ensures that the reaction conditions remain mild, typically operating between 30°C and 80°C, avoiding the need for high-pressure or cryogenic environments. Consequently, this novel route offers a reliable pharmaceutical intermediates supplier with a distinct competitive advantage in terms of operational safety and regulatory compliance.
Mechanistic Insights into Copper-Mediated Bromination and DMSO Oxidation
The chemical elegance of this synthesis is anchored in the precise mechanistic control exerted during the transformation of 3,5-dibenzyloxy acetophenone into the corresponding phenylacetaldehyde derivative. The process initiates with a regioselective alpha-bromination utilizing copper(II) bromide (CuBr2) in a mixed solvent system of ethyl acetate and chloroform. This step generates the alpha-bromo ketone intermediate with high fidelity, setting the stage for the subsequent oxidation. Unlike harsh halogenation methods that can lead to poly-halogenated byproducts, the copper-mediated protocol ensures clean conversion. Following filtration to remove copper salts, the crude bromo-ketone undergoes oxidation in the presence of DMSO and a mild base such as sodium bicarbonate. This Kornblum-type oxidation mechanism facilitates the displacement of the bromide ion by the sulfoxide oxygen, followed by elimination to yield the aldehyde functionality essential for the subsequent amine coupling.
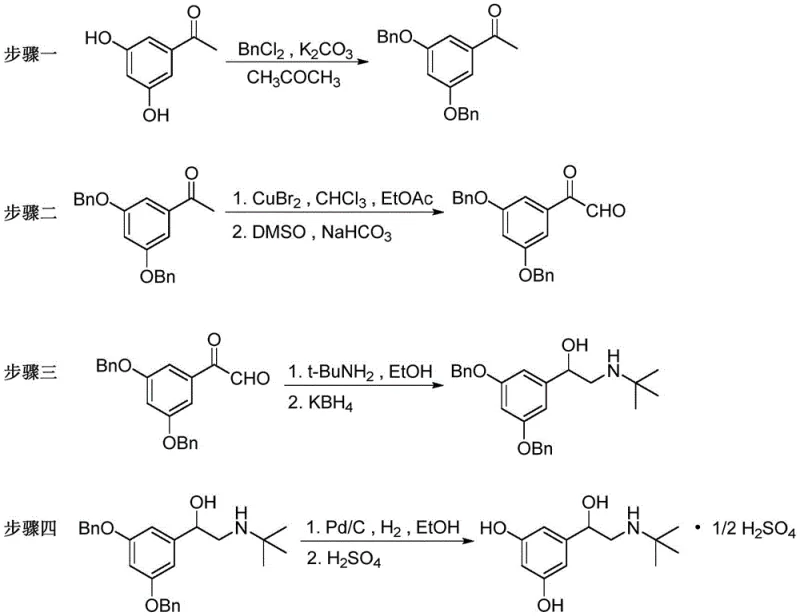
Following the formation of the aldehyde, the synthesis proceeds through a reductive amination with tert-butylamine, a critical step that installs the bulky amine group required for beta-2 adrenergic receptor selectivity. The use of potassium borohydride (KBH4) as the reducing agent is particularly advantageous for impurity control; it is selective enough to reduce the imine intermediate without affecting other sensitive functional groups, ensuring a clean profile for the resulting amino-alcohol. The final deprotection via catalytic hydrogenation over Pd/C removes the benzyl protecting groups simultaneously, revealing the free phenolic hydroxyl groups while preserving the stereochemical integrity of the chiral center formed during reduction. This mechanistic pathway minimizes the generation of difficult-to-remove impurities, directly addressing the purity concerns that often plague generic API manufacturing.
How to Synthesize Terbutaline Sulfate Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and safety. The process begins with the protection of the phenolic hydroxyl groups using benzyl chloride and potassium carbonate in acetone at elevated temperatures, followed by the critical copper-bromide mediated functionalization. The subsequent reductive amination must be carefully monitored to prevent over-reduction or incomplete condensation. For process chemists looking to replicate this high-efficiency route, the detailed standardized operating procedures are critical for success.
- Perform benzyl protection on 3,5-dihydroxy acetophenone using benzyl chloride and potassium carbonate in acetone at 50-80°C to obtain 3,5-dibenzyloxy acetophenone.
- Conduct bromination with copper bromide followed by oxidation using DMSO and sodium bicarbonate to generate the key aldehyde intermediate.
- Execute reductive amination with tert-butylamine and potassium borohydride in ethanol to form the amino-alcohol backbone.
- Finalize the process via catalytic hydrogenation using Pd/C to remove benzyl groups, followed by salt formation with sulfuric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible strategic benefits that extend far beyond simple chemical yield. By eliminating the reliance on selenium dioxide and methyllithium, manufacturers can drastically simplify their environmental, health, and safety (EHS) protocols, removing the need for expensive hazardous waste disposal services and specialized storage facilities for pyrophoric materials. This shift not only reduces the direct cost of goods sold (COGS) but also mitigates the risk of production stoppages due to regulatory inspections or safety incidents. Furthermore, the use of commodity chemicals like copper bromide and DMSO ensures a stable and resilient supply chain, insulating production schedules from the volatility often seen with specialized organometallic reagents.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of expensive and hazardous reagents with inexpensive, bulk-available alternatives. The elimination of selenium-based oxidants removes a significant cost center associated with toxic waste treatment and regulatory compliance fees. Additionally, the shortened reaction sequence reduces solvent consumption and energy usage, as the reactions proceed efficiently at moderate temperatures without the need for energy-intensive heating or cooling cycles. This cumulative effect leads to substantial cost savings in pharmaceutical intermediates manufacturing, allowing for more competitive pricing in the global market.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for API production, and this route enhances reliability by utilizing raw materials that are widely sourced and commercially mature. Unlike processes dependent on custom-synthesized or import-restricted reagents, the inputs for this method—such as acetone, ethanol, and common inorganic salts—are readily available from multiple vendors globally. This diversification of the supply base reduces the risk of single-source bottlenecks and ensures that production timelines can be met consistently, even during periods of global logistical disruption.
- Scalability and Environmental Compliance: From an environmental standpoint, the process is designed for green chemistry principles, generating fewer hazardous byproducts and utilizing solvents that are easier to recover and recycle. The mild reaction conditions facilitate easier scale-up from pilot plant to multi-ton commercial production without the engineering complexities associated with high-pressure hydrogenation or cryogenic reactions. This scalability ensures that the manufacturing capacity can be rapidly expanded to meet market demand while maintaining strict adherence to increasingly stringent environmental regulations regarding heavy metal discharge and volatile organic compound (VOC) emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical application of this method for industrial partners.
Q: Why is this new synthesis route considered safer than traditional methods?
A: Traditional methods often utilize highly toxic oxidants like selenium dioxide or dangerous metal reagents like methyllithium. This patented process replaces them with copper bromide and DMSO, significantly reducing toxicity risks and eliminating the need for extreme temperature or pressure conditions.
Q: What are the typical yield improvements in this manufacturing process?
A: The process demonstrates robust yields across all stages, with the initial protection step achieving approximately 80-82% yield and the final hydrogenation and salification step reaching yields around 80-83%, ensuring high material efficiency for commercial production.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method is specifically designed for scalability. It utilizes cheap, easily available raw materials, avoids high-risk reagents, and operates under mild reaction conditions (30-80°C), making it highly suitable for cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terbutaline Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires more than just chemical knowledge; it demands deep engineering expertise and a commitment to quality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel route are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of Terbutaline Sulfate meets the highest international standards, providing our partners with the confidence needed to navigate complex regulatory filings.
We invite global pharmaceutical companies to collaborate with us to leverage this advanced manufacturing technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your project moves forward with the most efficient and economically sound strategy available in the market.
