Advanced Organocatalytic Strategy for Scalable Chiral Spirooxindole Pharmaceutical Intermediates
Advanced Organocatalytic Strategy for Scalable Chiral Spirooxindole Pharmaceutical Intermediates
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for greener, more efficient, and cost-effective methodologies. A significant breakthrough in this domain is detailed in Chinese Patent CN108409746B, which discloses a novel preparation method for chiral oxo spiro indole compounds containing pyrrole or indoloazepine structures. These spirooxindole scaffolds are critical structural motifs found in numerous bioactive natural products and synthetic drugs, particularly those exhibiting potent antitumor activities. The patent introduces a streamlined approach that utilizes chiral phosphoric acid as an organocatalyst to facilitate a direct [m+1] cycloaddition reaction. This innovation represents a paradigm shift from traditional multi-step syntheses, offering a robust pathway for producing high-purity pharmaceutical intermediates with excellent stereocontrol.
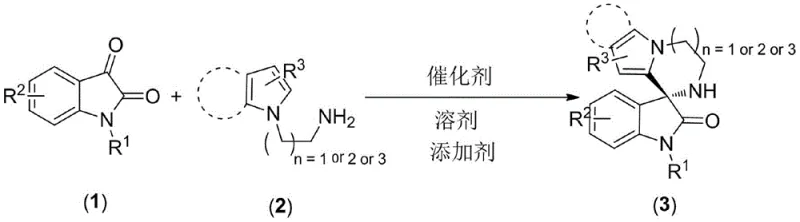
For R&D directors and process chemists, the ability to construct complex spirocyclic cores in a single step is highly desirable. The disclosed method employs N-substituted isatin or benzene ring-substituted isatin reacting with N-alkylamino-substituted pyrrole or indole derivatives. By leveraging the hydrogen-bonding activation capability of chiral phosphoric acids, the reaction proceeds through a [5+1], [6+1], or [7+1] cycloaddition pathway. This versatility allows for the generation of diverse ring systems, including the previously difficult-to-access 3',4'-dihydro-2'H-spiro[indoline-3,1'-pyrrolo[1,2-a]pyrazine]-2-one skeletons. The strategic design of this catalytic system not only enhances reaction efficiency but also provides a reliable platform for exploring structure-activity relationships in early-stage drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral oxo spiro indole compounds has been fraught with challenges that hinder large-scale application. Traditional strategies often rely on the initial construction of a carbon-carbon double bond at the 3-position of the isatin moiety, typically via Knoevenagel condensation or Wittig reactions. These preliminary steps add complexity to the synthetic route, increasing both time and material costs. Furthermore, subsequent cycloaddition reactions frequently require harsh conditions or specific substrate modifications that limit scope. For instance, prior art involving transition metal catalysis, such as nickel or rhodium complexes, often necessitates rigorous exclusion of moisture and oxygen, requiring specialized equipment and inert gas protection. Additionally, the use of phosphorus ylides in Wittig reactions generates stoichiometric amounts of triphenylphosphine oxide byproducts, which are notoriously difficult to remove and can interfere with downstream purification, thereby reducing overall yield and purity.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the technology described in CN108409746B offers a direct, one-pot solution that bypasses the need for pre-functionalized olefinic intermediates. By utilizing unmodified isatin derivatives directly in a catalytic [m+1] cycloaddition, the process drastically reduces the number of unit operations required. The use of chiral phosphoric acid catalysts eliminates the dependency on expensive and potentially toxic transition metals, addressing both economic and environmental concerns. This organocatalytic approach operates under mild conditions, typically between 0°C and 50°C, and tolerates a broader range of functional groups. The ability to achieve high enantioselectivity without the need for chiral ligands associated with metal centers simplifies the supply chain for catalysts and reduces the risk of metal contamination in the final active pharmaceutical ingredient (API).
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cycloaddition
The success of this transformation lies in the unique activation mode of the chiral phosphoric acid catalyst. These small organic molecules function as Brønsted acids, activating the electrophilic carbonyl group of the isatin substrate through a dual hydrogen-bonding network. Simultaneously, the basic nitrogen of the phosphate can interact with the nucleophilic amine component, organizing the transition state within a well-defined chiral pocket. This bifunctional activation ensures that the cycloaddition proceeds with high facial selectivity, leading to the preferential formation of one enantiomer over the other. The steric bulk of the substituents on the phosphoric acid backbone, such as the binaphthyl or spirocyclic frameworks shown in the patent, plays a crucial role in discriminating between the prochiral faces of the reacting species.
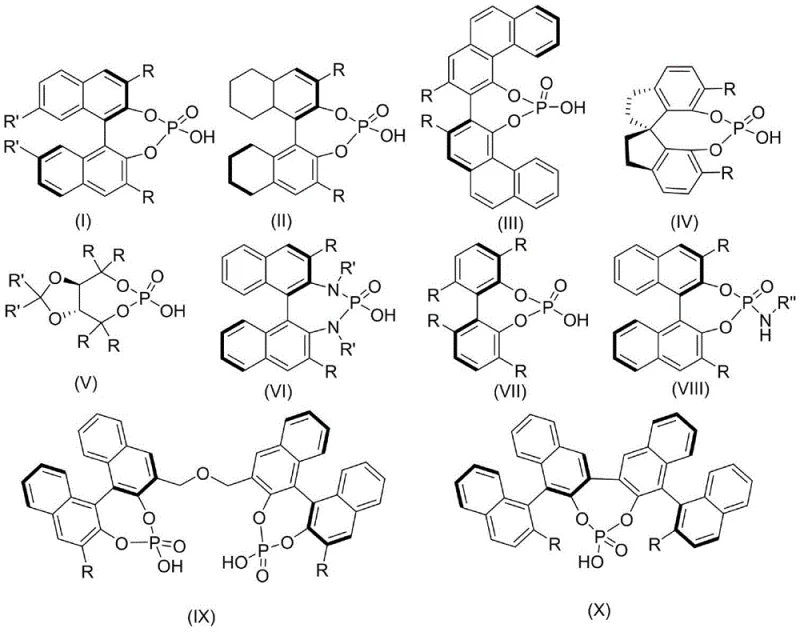
From an impurity control perspective, this mechanism offers distinct advantages. Since the reaction does not involve radical intermediates or highly reactive organometallic species, the formation of non-specific byproducts is minimized. The primary side reactions are typically limited to unreacted starting materials or simple hydrolysis products, which are easily separated during workup. The absence of metal catalysts also means there is no risk of metal-induced decomposition or complexation with the product, which can often complicate the impurity profile in metal-catalyzed processes. This clean reaction profile is essential for meeting the stringent purity specifications required for clinical-grade pharmaceutical intermediates, ensuring that the final material is free from genotoxic impurities often associated with heavy metal residues.
How to Synthesize Chiral Oxo Spiro Indole Compounds Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and pilot plant equipment. The general procedure involves dissolving the isatin and amino-pyrrole substrates in a common organic solvent such as dichloromethane, toluene, or chloroform. To this mixture, a catalytic amount of the selected chiral phosphoric acid (typically 1-20 mol%) and an additive like molecular sieves are added to scavenge water and drive the equilibrium forward. The reaction is allowed to proceed at ambient or slightly elevated temperatures until completion, as monitored by TLC or HPLC. Detailed standardized synthetic steps for specific derivatives are provided in the technical guide below.
- Prepare a mixed solution of N-substituted isatin and N-alkylamino-substituted pyrrole or indole in a suitable organic solvent such as dichloromethane or toluene.
- Add a chiral phosphoric acid catalyst (1-20 mol%) and an additive such as molecular sieves to the reaction mixture under inert atmosphere.
- Stir the reaction at mild temperatures between 0°C and 50°C for up to 48 hours, then purify the crude product via column chromatography to isolate the target chiral spiro compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic technology presents compelling economic and logistical benefits. The shift from metal-based catalysis to organocatalysis fundamentally alters the cost structure of manufacturing these complex intermediates. By removing the requirement for precious metals like rhodium or specialized ligands, the raw material costs are significantly reduced. Furthermore, the simplified workup procedure, which avoids the need for extensive metal scavenging steps or complex chromatographic separations to remove phosphine oxides, leads to substantial cost savings in processing time and solvent consumption. This efficiency translates directly into a more competitive pricing model for the final intermediate.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated ligands removes a major cost driver from the bill of materials. Additionally, the one-step nature of the reaction reduces labor costs and energy consumption compared to multi-step sequences. The avoidance of stoichiometric reagents that generate difficult-to-remove byproducts further lowers waste disposal costs, contributing to a leaner and more economical manufacturing process that enhances overall margin potential for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Relying on organocatalysts mitigates the supply chain risks associated with the geopolitical volatility of precious metal markets. Chiral phosphoric acids are stable, shelf-stable solids that are easier to source and store than air-sensitive metal complexes. The robustness of the reaction conditions, which do not require stringent anhydrous or oxygen-free environments, reduces the likelihood of batch failures due to environmental excursions. This reliability ensures consistent delivery schedules and reduces the lead time for high-purity pharmaceutical intermediates, allowing for more agile response to market demands.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals make this process inherently safer and more environmentally friendly, aligning with green chemistry principles. Scaling up from gram to kilogram quantities does not require specialized pressure vessels or exotic containment systems. The reduced hazardous waste stream simplifies regulatory compliance and environmental permitting. This scalability facilitates the commercial scale-up of complex pharmaceutical intermediates, ensuring a continuous supply capable of supporting clinical trials and eventual commercial production without the bottlenecks often seen with sensitive metal-catalyzed processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and performance of the method for potential partners and stakeholders evaluating this route for their supply chains.
Q: What are the advantages of using chiral phosphoric acid over transition metal catalysts for spirooxindole synthesis?
A: Chiral phosphoric acid organocatalysis eliminates the need for expensive and toxic transition metals like rhodium or nickel, simplifying purification processes and reducing heavy metal residue risks in pharmaceutical intermediates.
Q: Can this synthesis method be scaled for commercial production of API intermediates?
A: Yes, the reaction operates under mild conditions (0-50°C) without stringent anhydrous or oxygen-free requirements typical of metal catalysis, making it highly suitable for commercial scale-up and cost reduction in API manufacturing.
Q: What is the enantioselectivity achieved in this novel cycloaddition pathway?
A: The method achieves high enantioselectivity, with specific examples demonstrating enantiomeric excess (ee) values up to 88%, providing access to optically pure lead compounds for antitumor drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Spirooxindole Supplier
The technological advancements described in CN108409746B highlight the immense potential of organocatalysis in modern pharmaceutical synthesis. At NINGBO INNO PHARMCHEM, we recognize the value of such innovative routes in delivering high-quality intermediates to the global market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of chiral spirooxindole intermediate meets the highest standards required for drug development. We are committed to translating laboratory breakthroughs into robust, GMP-compliant manufacturing processes.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this efficient synthesis technology. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact us to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us help you accelerate your drug discovery timeline with reliable, cost-effective, and high-purity chemical solutions.
