Advanced Synthesis of Potent Bronchodilator Naphthalene Derivatives for Commercial Scale-up
The pharmaceutical landscape for respiratory treatments is constantly evolving, driven by the need for agents that offer superior efficacy without the debilitating side effects of legacy drugs. Patent CN1045435C introduces a groundbreaking class of naphthalene derivatives characterized by a unique structural motif that combines a substituted naphthalene core with a nitrogen-containing six-membered heterocyclic group. These compounds have demonstrated exceptional bronchodilating activity, making them highly valuable candidates for the prophylaxis and treatment of asthma. The structural versatility allows for various substitutions at the R1, R2, and R3 positions, enabling fine-tuning of pharmacokinetic properties. As a reliable pharmaceutical intermediate supplier, understanding the nuances of this chemical space is critical for developing next-generation therapeutics that meet modern safety standards.
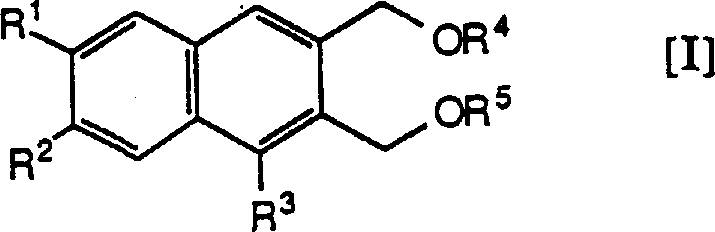
The development of these novel compounds addresses a significant gap in current asthma management strategies. Traditional antiasthmatic drugs, while effective to a degree, often suffer from limitations such as an inability to effectively suppress bronchoconstriction or the induction of serious cardiac side effects. The compounds described in this patent exhibit inhibitory activity against bronchoconstriction that is reported to be 3 to 100 times stronger than that of theophylline, a standard reference drug. Furthermore, unlike theophylline, these derivatives do not display adverse effects on the heart, such as hypotension or palpitations. This distinct safety profile positions them as high-purity pharmaceutical intermediates with substantial commercial potential for companies seeking to innovate in the respiratory therapy sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex naphthalene-based therapeutics has been plagued by inefficient routes that rely on harsh reaction conditions and difficult-to-remove impurities. Conventional methods often struggle with regioselectivity when introducing substituents onto the naphthalene ring, leading to complex mixtures that require extensive and costly purification steps. Additionally, many traditional pathways utilize expensive or hazardous reagents that pose significant challenges for commercial scale-up and environmental compliance. The lack of robust methods for constructing the specific 2,3-bis(hydroxymethyl) substitution pattern on the naphthalene core has further hindered the rapid development of analogues. These inefficiencies translate directly into higher manufacturing costs and longer lead times for high-purity pharmaceutical intermediates, creating bottlenecks in the supply chain for drug developers.
The Novel Approach
The methodology outlined in the patent offers a transformative solution by leveraging a modular synthetic strategy centered on the reduction of diester precursors. This approach allows for the precise installation of the critical hydroxymethyl groups at the 2 and 3 positions of the naphthalene ring with high fidelity. By utilizing readily available starting materials such as substituted benzaldehydes and maleic acid diesters, the route minimizes reliance on exotic reagents. The process is designed to be adaptable, accommodating a wide range of substituents on both the naphthalene core and the heterocyclic ring. This flexibility facilitates the rapid generation of diverse compound libraries for structure-activity relationship studies. Ultimately, this novel approach streamlines the path from bench-scale discovery to cost reduction in pharmaceutical intermediate manufacturing by simplifying the overall synthetic sequence.
Mechanistic Insights into Hydride-Mediated Ester Reduction
The core transformation in this synthesis involves the reduction of a 2,3-bis(methoxycarbonyl)naphthalene derivative to the corresponding 2,3-bis(hydroxymethyl) analogue. This reaction is typically conducted using powerful hydride reducing agents such as lithium aluminum hydride or sodium bis(methoxyethoxy)aluminum hydride in aprotic solvents like tetrahydrofuran or diethyl ether. The mechanism proceeds through the nucleophilic attack of the hydride species on the carbonyl carbon of the ester groups, forming a tetrahedral intermediate that subsequently collapses to release the alkoxide. Careful control of reaction temperature, often ranging from -20°C to room temperature, is essential to prevent over-reduction or degradation of the sensitive heterocyclic moiety attached at the 1-position. The choice of reducing agent can be tuned based on the specific electronic nature of the substituents, ensuring optimal conversion rates and minimizing byproduct formation.
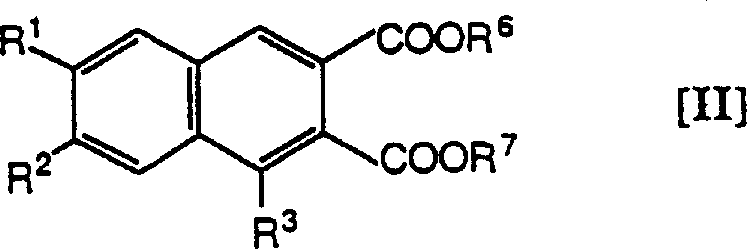
Following the reduction, the resulting diol intermediates can be further functionalized to introduce protecting groups or to modify the heterocyclic ring system. For instance, the hydroxyl groups can be protected as acyl or alkyl ethers to facilitate subsequent reactions on the nitrogen-containing ring without interference. The patent details various methods for converting pyridyl groups into pyridones or N-alkylated derivatives through oxidation and alkylation steps. These transformations are crucial for unlocking the full therapeutic potential of the scaffold. The ability to selectively protect and deprotect functional groups ensures that the final active pharmaceutical ingredient is obtained with the desired substitution pattern and high chemical purity. This level of control over the synthetic pathway is vital for maintaining consistent quality in large-scale production environments.
How to Synthesize Naphthalene Derivative Intermediates Efficiently
The synthesis of these high-value intermediates requires a disciplined approach to reaction engineering and process optimization. The patented methods provide a clear roadmap for constructing the complex molecular architecture efficiently. By following the established protocols for ester reduction and heterocyclic coupling, manufacturers can achieve reproducible results with minimal waste. The detailed procedures cover everything from reagent addition rates to workup and purification strategies, ensuring that the process is robust enough for industrial application. For those looking to implement this chemistry, the following guide outlines the critical operational parameters derived from the patent examples.
- Preparation of the naphthalene diester precursor via condensation of substituted benzaldehydes with maleic diesters.
- Selective reduction of the diester groups to hydroxymethyl moieties using hydride reducing agents like sodium bis(methoxyethoxy)aluminum hydride.
- Functionalization of the heterocyclic ring through alkylation or oxidation to achieve the final active pharmaceutical ingredient structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical feasibility. The reliance on commodity chemicals such as maleic diesters and substituted benzaldehydes ensures a stable and secure supply of raw materials, mitigating the risk of shortages that often plague specialty chemical markets. Furthermore, the synthetic steps are designed to be operationally simple, utilizing standard unit operations like filtration, extraction, and crystallization that are easily scalable in existing manufacturing facilities. This compatibility with standard infrastructure reduces the need for significant capital expenditure on new equipment, thereby accelerating the time to market for new drug candidates. The overall efficiency of the process contributes to substantial cost savings in the long run.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for transition metal catalysts in key steps, which removes the expensive and time-consuming requirement for heavy metal scavenging and residual analysis. By utilizing hydride reductions that generate soluble byproducts, the purification process is significantly simplified, reducing solvent consumption and waste disposal costs. The high yields reported in the examples, such as 91.1% in specific reduction steps, indicate a material-efficient process that maximizes the output from every kilogram of starting material. This efficiency directly translates to a lower cost of goods sold, making the final API more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are widely available from multiple global suppliers, reducing dependency on single-source vendors and enhancing supply chain resilience. The robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality, ensuring consistent production output. Additionally, the ability to store stable intermediates, such as the protected diesters, allows for better inventory management and production planning. This flexibility enables manufacturers to respond quickly to fluctuations in demand without compromising on delivery schedules or product quality.
- Scalability and Environmental Compliance: The process avoids the use of highly toxic reagents where possible, aligning with modern green chemistry principles and reducing the environmental footprint of manufacturing. The solvents used, such as ethyl acetate and alcohols, are relatively benign and can be recovered and recycled efficiently, further lowering operational costs. The scalability of the reduction and coupling reactions has been demonstrated through the successful preparation of multi-gram quantities in the patent examples, suggesting a smooth path to ton-scale production. This scalability ensures that the supply chain can support commercial volumes without the need for complex process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these naphthalene derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details is essential for R&D teams evaluating the feasibility of incorporating these intermediates into their drug development pipelines. The information covers aspects ranging from chemical stability to regulatory considerations.
Q: What represents the primary advantage of this naphthalene scaffold over traditional asthma medications?
A: Unlike traditional drugs such as theophylline which often cause severe cardiac side effects like hypotension and palpitations, these novel naphthalene derivatives exhibit potent bronchodilating activity with significantly reduced cardiac toxicity, offering a safer therapeutic profile.
Q: Which reducing agents are preferred for the commercial scale-up of the diol intermediate?
A: The patent data indicates that sodium bis(methoxyethoxy)aluminum hydride is particularly preferred for reducing the esterified carboxyl groups to hydroxymethyl groups, although lithium aluminum hydride and sodium borohydride are also viable depending on the specific substitution pattern.
Q: How does the synthesis route ensure high purity for regulatory compliance?
A: The process utilizes robust purification techniques such as recrystallization from solvent systems like ethyl acetate and hexane, alongside silica gel chromatography where necessary, ensuring the removal of impurities and meeting stringent pharmaceutical quality standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthalene Derivative Supplier
The technical potential of these naphthalene derivatives represents a significant opportunity for advancing respiratory medicine, but realizing this potential requires a manufacturing partner with deep expertise in complex organic synthesis. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from pilot plant to full-scale manufacturing. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of intermediate meets the highest industry standards. We understand the critical nature of supply continuity in the pharmaceutical sector and are committed to being a dependable extension of your supply chain.
We invite you to engage with our technical procurement team to discuss how we can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how optimizing this synthetic route can improve your project's economics. We encourage you to reach out for specific COA data and route feasibility assessments to verify the suitability of our materials for your application. Our team is ready to collaborate with you to overcome any engineering bottlenecks and accelerate your path to commercial success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
