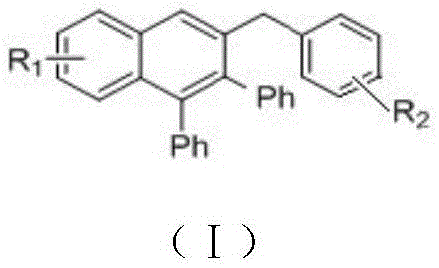
Advanced Ruthenium-Catalyzed Synthesis of Polyaromatic Substituted Naphthalene Derivatives for Commercial Scale-Up
The pharmaceutical and optoelectronic industries are constantly seeking more efficient pathways to construct complex polycyclic aromatic hydrocarbons, which serve as critical scaffolds for advanced functional materials and drug candidates. Patent CN107973691B introduces a groundbreaking methodology for the preparation of polyaromatic substituted naphthalene derivatives through a ruthenium-catalyzed cyclization reaction. This innovation specifically addresses the long-standing challenges associated with traditional synthesis routes by utilizing a cheap ruthenium catalyst, [RuCl2(p-cymene)]2, to activate the beta-hydrogen of aromatic ketones. The significance of this technology lies in its ability to construct the six-membered naphthalene core under remarkably mild conditions without the need for stoichiometric oxidants or hazardous additives. For R&D directors and procurement specialists, this represents a pivotal shift towards greener, more cost-effective manufacturing processes that maintain high purity standards while drastically simplifying the downstream purification workflow.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polyaromatic substituted naphthalene derivatives has relied heavily on transition metal-catalyzed C-H activation strategies that are often plagued by severe economic and environmental drawbacks. Conventional protocols typically necessitate the use of expensive ligands to stabilize the metal center and, more critically, require equivalent amounts of metal salts such as copper or silver to act as terminal oxidants to close the catalytic cycle. These stoichiometric oxidants not only inflate the raw material costs significantly but also generate substantial quantities of heavy metal waste, creating a burdensome environmental compliance issue for large-scale production facilities. Furthermore, these traditional reactions often demand harsh conditions, including high temperatures and strong acidic or basic environments, which can lead to poor functional group tolerance and the formation of complex impurity profiles that are difficult to separate, ultimately compromising the overall yield and quality of the final API intermediate or electronic material.
The Novel Approach
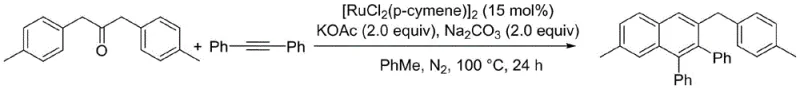
In stark contrast to the cumbersome legacy methods, the novel approach detailed in the patent utilizes a streamlined ruthenium-catalyzed system that eliminates the need for external oxidants entirely. By leveraging the unique redox properties of the [RuCl2(p-cymene)]2 catalyst, the reaction proceeds through an internal hydrogen transfer mechanism that activates the aromatic ketone beta-H bond directly. This oxidative annulation occurs smoothly in non-polar solvents like toluene at moderate temperatures around 100°C, using simple inorganic bases such as sodium carbonate and potassium acetate. This paradigm shift not only reduces the E-factor of the process by removing heavy metal oxidant waste but also enhances the safety profile of the operation by avoiding aggressive reagents. The result is a robust synthetic route that delivers high-purity polyaromatic naphthalene derivatives with excellent atom economy, making it an ideal candidate for reliable polyaromatic substituted naphthalene derivatives supplier networks aiming for sustainable growth.
Mechanistic Insights into Ruthenium-Catalyzed Cyclization
The core of this technological advancement lies in the sophisticated mechanistic pathway where the ruthenium catalyst facilitates the cleavage of the C-H bond adjacent to the carbonyl group (beta-position) of the aromatic ketone. Upon coordination with the alkyne substrate, diphenylacetylene, the ruthenium center promotes a migratory insertion followed by reductive elimination to form the new carbon-carbon bonds required for the naphthalene ring closure. Crucially, this cycle regenerates the active catalytic species without consuming an external oxidant, as the hydrogen atoms are likely eliminated as molecular hydrogen or captured by the base in a benign manner. This mechanism ensures that the reaction remains catalytic with respect to the metal, allowing for high turnover numbers even with relatively low catalyst loadings of 15 mol%. For process chemists, understanding this mechanism is vital for optimizing reaction parameters and predicting potential side reactions, ensuring that the scale-up from gram to kilogram levels maintains the high efficiency observed in the laboratory.
Impurity control in this ruthenium-catalyzed system is inherently superior due to the absence of stoichiometric metal oxidants which often leave behind difficult-to-remove inorganic residues. The primary impurities in such cyclization reactions typically arise from incomplete conversion or oligomerization of the alkyne, but the mild conditions employed here minimize thermal degradation pathways. The use of simple inorganic bases like Na2CO3 and KOAc ensures that the reaction medium remains buffered, preventing acid-catalyzed decomposition of sensitive functional groups such as halogens or methoxy groups present on the aromatic rings. This high level of chemoselectivity is paramount for producing high-purity OLED material precursors or pharmaceutical intermediates where trace metal contamination must be kept below strict ppm limits. The clean reaction profile simplifies the workup procedure, often requiring only standard column chromatography to isolate the target molecule, thereby reducing solvent consumption and processing time significantly compared to multi-step purification protocols needed for older methods.
How to Synthesize Polyaromatic Substituted Naphthalene Derivatives Efficiently
The practical implementation of this synthesis route is designed for straightforward execution in standard laboratory and pilot plant settings, requiring no specialized high-pressure equipment or exotic reagents. The process begins by charging a sealed reaction vessel with the aromatic ketone substrate and diphenylacetylene in a precise molar ratio, typically 2:1, to drive the equilibrium towards the desired product. Following the addition of the ruthenium catalyst and the dual-base system in toluene, the mixture is subjected to an inert nitrogen atmosphere to prevent any potential oxidation of the catalyst or substrates by air. The reaction is then heated to 100°C for a duration of 24 hours, allowing sufficient time for the cyclization to reach completion. Detailed standardized synthesis steps see the guide below.
- Combine diphenylacetylene and aromatic ketone in a sealed tube with [RuCl2(p-cymene)]2 catalyst, toluene solvent, sodium carbonate, and potassium acetate.
- Purge the reaction vessel with nitrogen three times to ensure an inert atmosphere and heat the mixture to 100°C for 24 hours.
- Upon completion, separate the target polyaromatic substituted naphthalene derivative using column chromatography with petroleum ether as the eluent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ruthenium-catalyzed technology offers profound strategic benefits that extend far beyond simple yield improvements. The elimination of expensive stoichiometric oxidants like silver or copper salts translates directly into substantial cost savings in raw material procurement, while simultaneously removing the logistical burden of disposing of hazardous heavy metal waste streams. This simplification of the supply chain reduces the dependency on volatile markets for specialty oxidants and mitigates regulatory risks associated with environmental compliance, ensuring a more stable and predictable production schedule. Furthermore, the use of commodity chemicals such as toluene, sodium carbonate, and potassium acetate ensures that the process is resilient to supply shocks, as these materials are readily available globally from multiple vendors, enhancing the overall reliability of the manufacturing operation.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the removal of stoichiometric oxidants and the use of a relatively inexpensive ruthenium dimer catalyst that operates efficiently at moderate loadings. By avoiding the purchase of high-cost silver or copper salts which are consumed in a 1:1 ratio with the substrate in traditional methods, the variable cost per kilogram of the final product is drastically lowered. Additionally, the simplified purification process reduces the consumption of silica gel and organic solvents during chromatography, further driving down the operational expenditure. This lean manufacturing approach allows for competitive pricing strategies in the global market for fine chemical intermediates without compromising on quality margins.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain stability, as the process is less sensitive to minor fluctuations in temperature or reagent quality compared to highly sensitive palladium-catalyzed couplings. The availability of the key starting materials, aromatic ketones and diphenylacetylenes, from established chemical suppliers ensures that production can be scaled rapidly to meet sudden increases in demand from downstream pharmaceutical or electronics clients. Moreover, the reduced generation of hazardous waste simplifies the logistics of waste management, preventing potential production stoppages due to waste disposal capacity limits or regulatory inspections, thus guaranteeing consistent delivery timelines.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to commercial production is facilitated by the use of standard heating and stirring equipment, with no requirement for high-pressure reactors or cryogenic cooling systems. The green chemistry attributes of the process, specifically the absence of heavy metal oxidant waste and the use of recyclable solvents like toluene, align perfectly with modern corporate sustainability goals and stringent environmental regulations. This compliance advantage reduces the administrative overhead related to environmental reporting and permitting, allowing the manufacturing facility to operate with greater agility and lower long-term liability risks associated with toxic waste accumulation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ruthenium-catalyzed cyclization technology, derived directly from the experimental data and scope defined in the patent literature. These insights are intended to clarify the operational parameters and potential applications for stakeholders evaluating this method for their own production pipelines. Understanding these nuances is essential for making informed decisions about process adoption and integration into existing manufacturing workflows.
Q: Does this ruthenium-catalyzed method require stoichiometric oxidants?
A: No, unlike conventional methods that often require equivalent amounts of copper or silver salts as oxidants, this patented process operates without any additives or external oxidants, significantly reducing heavy metal waste and purification costs.
Q: What are the optimal reaction conditions for maximizing yield?
A: The patent data indicates that using toluene as the solvent, a combination of sodium carbonate and potassium acetate as bases, and a catalyst loading of 15 mol% at 100°C for 24 hours provides the highest yields, reaching up to 68% for certain substrates.
Q: Can this synthesis method be applied to halogenated substrates?
A: Yes, the method demonstrates excellent tolerance for various substituents including fluoro, chloro, bromo, and iodo groups on the aromatic rings, making it highly versatile for synthesizing functionalized intermediates for further cross-coupling reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyaromatic Substituted Naphthalene Derivatives Supplier
As the demand for high-performance organic materials continues to surge in the pharmaceutical and optoelectronic sectors, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM becomes a strategic imperative for securing your supply chain. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. Our rigorous QC labs and adherence to stringent purity specifications guarantee that every batch of polyaromatic substituted naphthalene derivatives meets the exacting standards required for next-generation drug candidates and blue light emitting materials, providing you with a competitive edge in your respective markets.
We invite you to engage with our technical procurement team to discuss how this innovative ruthenium-catalyzed route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your development timeline remains on track with a reliable partner dedicated to chemical excellence and sustainable innovation.
