Advanced Synthesis of Bioactive Diosgenin Derivatives for Oncology Applications
Advanced Synthesis of Bioactive Diosgenin Derivatives for Oncology Applications
The pharmaceutical landscape is continuously evolving towards more potent and selective therapeutic agents, particularly in the realm of oncology where resistance and toxicity remain critical challenges. Recent intellectual property developments, specifically patent CN110028547B, have unveiled a groundbreaking structural modification of diosgenin, a well-known steroidal sapogenin. This patent details the synthesis of a novel diosgenin 3-OH derivative, designated as Compound III, which demonstrates markedly superior antitumor efficacy compared to its natural precursor. For research and development teams focused on steroid-based drug discovery, this innovation represents a significant leap forward, offering a robust chemical scaffold with enhanced biological activity against liver, lung, and breast cancer cell lines. As a leading entity in the fine chemical sector, we recognize the immense potential of this pathway to serve as a reliable pharmaceutical intermediate supplier for next-generation anticancer drug development.
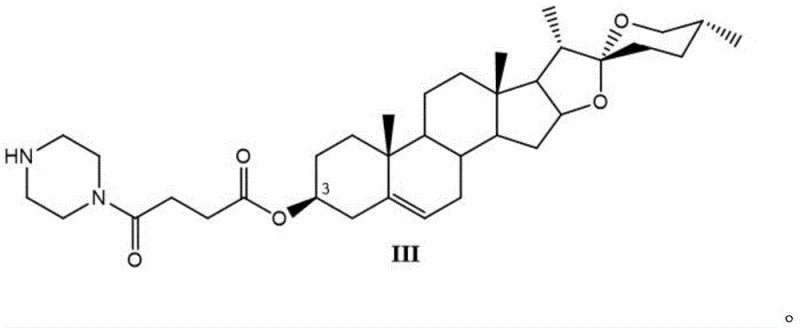
The core innovation lies in the strategic functionalization of the steroid backbone at the 3-hydroxyl position. By introducing a succinate linker terminated with a piperazine moiety, the resulting molecule achieves a balance of lipophilicity and hydrogen-bonding capability that drastically improves cellular uptake and target interaction. This structural refinement addresses the historical limitations of diosgenin, which, despite its abundance in nature, suffers from relatively weak pharmacological potency and a narrow therapeutic spectrum. The ability to transform this abundant natural product into a high-value bioactive intermediate opens new avenues for cost reduction in pharmaceutical manufacturing, allowing producers to leverage low-cost raw materials to create high-margin therapeutic candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the utilization of diosgenin in medicinal chemistry has been hindered by its inherent lack of potency and the difficulty in modifying its rigid steroidal skeleton without causing degradation. Conventional derivatization methods often rely on harsh acidic or basic conditions that can compromise the integrity of the spirostane ring system or the sensitive double bond at the C-5 position. Furthermore, older synthetic routes frequently suffer from poor regioselectivity, leading to complex mixtures of isomers that are difficult and expensive to separate. These inefficiencies result in low overall yields and generate significant chemical waste, posing both economic and environmental burdens on the supply chain. For procurement managers, these factors translate into volatile pricing and unreliable delivery schedules for high-purity steroid intermediates, creating bottlenecks in the early stages of drug development pipelines.
The Novel Approach
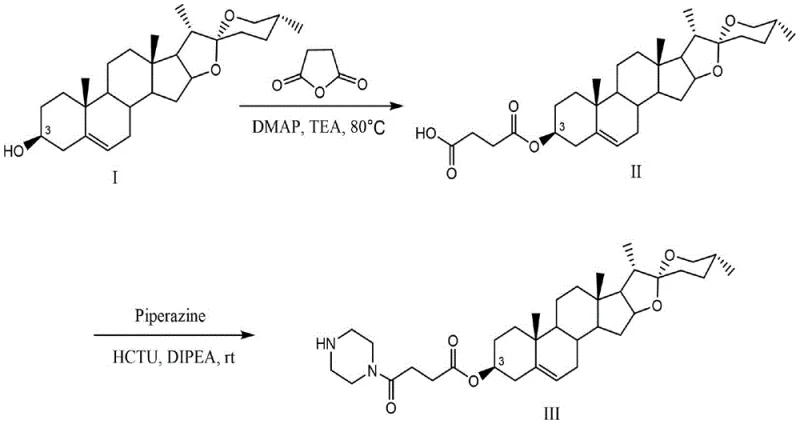
In stark contrast, the methodology disclosed in patent CN110028547B offers a streamlined, two-step synthetic strategy that prioritizes mild conditions and high efficiency. The process begins with a highly selective esterification of diosgenin with succinic anhydride, followed by a coupling reaction with piperazine. This approach avoids the use of aggressive reagents, instead utilizing catalytic amounts of 4-Dimethylaminopyridine (DMAP) and Triethylamine (TEA) at a moderate temperature of 80°C. The second step proceeds at room temperature using HCTU as a coupling agent, further minimizing energy consumption and thermal stress on the molecule. This gentle yet effective protocol ensures the preservation of the stereochemical integrity of the steroid core while achieving impressive isolated yields of up to 94%. Such operational simplicity makes this route ideal for the commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality and supply continuity.
Mechanistic Insights into DMAP-Catalyzed Esterification and HCTU Coupling
The success of this synthetic route relies heavily on the precise orchestration of nucleophilic catalysis and peptide coupling chemistry. In the first step, DMAP acts as a potent nucleophilic catalyst, attacking the succinic anhydride to form a highly reactive acylpyridinium intermediate. This species is significantly more electrophilic than the parent anhydride, facilitating a rapid attack by the 3-hydroxyl group of diosgenin. The presence of Triethylamine serves to neutralize the carboxylic acid byproduct, driving the equilibrium forward and preventing acid-catalyzed side reactions that could degrade the steroid skeleton. This mechanism ensures that the esterification occurs exclusively at the C-3 position, maintaining the purity profile required for regulatory compliance in API synthesis. The mild thermal conditions (80°C) are sufficient to overcome the activation energy barrier without inducing elimination reactions or isomerization of the delta-5 double bond.
The subsequent amidation step leverages the power of uranium-based coupling reagents, specifically HCTU (6-chlorobenzotriazole-1,1,3,3-tetramethyluronium hexafluorophosphate). HCTU activates the carboxylic acid of Intermediate II to form an active ester species, which is then attacked by the nucleophilic nitrogen of piperazine. The use of DIPEA (N,N'-Diisopropylethylamine) as a non-nucleophilic base is critical here; it scavenges the proton released during amide bond formation without competing for the activated ester. This chemoselectivity is paramount for impurity control, as it prevents the formation of N-acylurea byproducts or racemization, although racemization is less of a concern with this specific achiral linker. The result is a clean reaction profile that simplifies downstream purification, directly contributing to reduced processing time and lower solvent consumption in the manufacturing workflow.
How to Synthesize Diosgenin 3-OH Derivative Efficiently
Implementing this synthesis requires careful attention to stoichiometry and reaction monitoring to maximize the benefits of the patented protocol. The process is designed to be robust, tolerating minor variations in mixing rates while demanding precise control over reagent equivalents to minimize waste. Operators should utilize thin-layer chromatography (TLC) to monitor the consumption of starting materials, ensuring that the reaction is quenched at the optimal point to prevent hydrolysis of the activated intermediates. The workup procedure involves standard aqueous extractions and acid-base washes, which are easily adaptable to large-scale reactor setups. For a detailed breakdown of the specific operational parameters, reagent grades, and purification techniques required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Perform esterification of diosgenin with succinic anhydride using DMAP and TEA at 80°C to generate Intermediate II.
- Activate Intermediate II with HCTU and react with piperazine in acetonitrile at room temperature to form the target amide bond.
- Purify the crude product via column chromatography to obtain Compound III with high structural integrity and yield.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic pathway offers compelling advantages that address the core concerns of procurement managers and supply chain directors. The shift from a low-value natural product to a high-value bioactive intermediate creates significant margin potential while mitigating the risks associated with sourcing rare or exotic starting materials. Because the synthesis relies on commodity chemicals like succinic anhydride and piperazine, the supply chain is inherently more resilient to market fluctuations compared to routes dependent on specialized reagents. Furthermore, the high operational yield reduces the volume of raw materials required per kilogram of finished product, leading to substantial cost savings in material procurement. The simplicity of the workup also implies lower utility costs and reduced demand on waste treatment facilities, aligning with modern sustainability goals in chemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of harsh reaction conditions and the use of catalytic rather than stoichiometric amounts of expensive activators significantly lowers the cost of goods sold (COGS). By achieving yields as high as 94%, the process minimizes the loss of valuable steroid starting material, which is often the most expensive component of the bill of materials. Additionally, the ability to perform the second step at room temperature eliminates the need for energy-intensive heating or cooling cycles, further driving down operational expenditures. These efficiencies collectively enable a more competitive pricing structure for the final intermediate, allowing downstream partners to optimize their own R&D budgets.
- Enhanced Supply Chain Reliability: The reliance on widely available, off-the-shelf reagents ensures that production is not bottlenecked by the lead times of custom-synthesized catalysts. Diosgenin itself is a mass-produced commodity derived from yam species, guaranteeing a stable and continuous supply of the core scaffold. The robustness of the reaction conditions means that the process can be transferred between different manufacturing sites with minimal re-validation, providing flexibility in case of regional supply disruptions. This reliability is crucial for maintaining the continuity of clinical trial material supply and eventual commercial launch timelines.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, utilizing solvents like dichloromethane and acetonitrile which are standard in industrial settings and have established recovery protocols. The absence of heavy metal catalysts simplifies the purification process and reduces the burden of residual metal testing, a critical checkpoint in pharmaceutical quality control. Moreover, the high atom economy of the coupling steps results in less chemical waste generation, facilitating easier compliance with increasingly stringent environmental regulations. This green chemistry profile enhances the long-term viability of the manufacturing process and reduces the risk of regulatory shutdowns due to environmental non-compliance.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into their supply chains. The following questions address common inquiries regarding the scalability, purity, and biological relevance of the diosgenin derivative. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making. For further clarification on specific technical parameters or customization options, our technical team is available to provide detailed route feasibility assessments.
Q: What represents the primary advantage of this diosgenin derivative over the parent compound?
A: The derivative exhibits significantly enhanced antitumor activity against HepG2, A549, MDA-MB-231, and MCF-7 cell lines compared to unmodified diosgenin, addressing the limitation of weak bioactivity in the natural product.
Q: Is the synthesis process suitable for large-scale industrial production?
A: Yes, the method utilizes mild reaction conditions (80°C and room temperature), common organic solvents, and achieves high yields (up to 94%), making it highly operable and controllable for commercial scale-up.
Q: What specific catalytic systems are employed in this pathway?
A: The process employs DMAP/triethylamine for the initial esterification and an HCTU/DIPEA coupling system for the subsequent amidation, ensuring efficient bond formation without harsh reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diosgenin Derivative Supplier
At NINGBO INNO PHARMCHEM, we specialize in translating complex academic and patent literature into viable industrial processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising molecules like this diosgenin derivative can reach the market efficiently. We understand that the transition from milligram-scale lab synthesis to ton-scale manufacturing presents unique engineering challenges, which is why we invest heavily in rigorous QC labs and process optimization capabilities. Our commitment to stringent purity specifications ensures that every batch of intermediate meets the exacting standards required for pharmaceutical applications, minimizing the risk of downstream failures.
We invite procurement leaders and R&D directors to collaborate with us to unlock the full potential of this technology. By leveraging our expertise, you can secure a stable supply of high-quality intermediates while optimizing your overall project costs. We encourage you to contact our technical procurement team to request specific COA data and discuss how we can support your specific volume requirements. Let us help you navigate the complexities of steroid chemistry with a Customized Cost-Saving Analysis tailored to your project's unique needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
