Advanced Silver-Catalyzed Para-Amination Dearomatization for Commercial Pharmaceutical Intermediate Production
The landscape of organic synthesis is constantly evolving, driven by the need for more efficient and environmentally benign methodologies to construct complex molecular architectures. A significant breakthrough in this domain is documented in patent CN111362841B, which details a novel method for the para-amination dearomatization of phenol compounds. This technology addresses critical challenges in the functionalization of phenolic moieties, which are ubiquitous in natural products, dyes, and pharmaceuticals. By utilizing a transition metal-catalyzed approach in an aqueous phase, this invention offers a robust pathway to synthesize 4-amino substituted cyclohexadienone derivatives. These derivatives serve as vital building blocks for the construction of complex molecules and active pharmaceutical ingredients. The strategic implementation of this patent allows manufacturers to access high-purity intermediates with superior regioselectivity, marking a substantial advancement over conventional synthetic routes that often suffer from harsh conditions and poor selectivity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for the dearomatization of phenols often rely on high-valence iodine reagents or harsh transition metal-catalyzed alkylation and arylation processes. These conventional approaches frequently encounter significant hurdles, particularly regarding chemo- and regioselectivity. The free phenolic hydroxyl group is inherently acidic and nucleophilic, which typically leads to preferential O-H substitution rather than the desired C-H substitution. Furthermore, in electrophilic reactions, the ortho and para positions on the aromatic ring are both nucleophilic, which drastically reduces the regioselectivity of the reaction. Phenols, being electron-rich aromatic hydrocarbons, are also highly susceptible to oxidative decomposition under rigorous reaction conditions. These limitations result in complex mixture profiles, difficult purification processes, and ultimately, lower overall yields, making them less ideal for the cost reduction in pharmaceutical intermediate manufacturing required by modern industry standards.
The Novel Approach
In stark contrast to these traditional limitations, the novel approach described in the patent utilizes silver oxide (Ag2O) as a catalyst in a green solvent water phase. This method achieves para-amination dearomatization with remarkable efficiency and selectivity. The reaction conditions are simple and mild, typically requiring an ice bath and reaction times ranging from 0.5 to 10 hours. By employing diethyl azodicarboxylate as the amination reagent, the process effectively converts the phenolic C-H bond into a C-N bond with high precision. The use of water as a solvent not only aligns with green chemistry principles but also simplifies the downstream processing. The raw materials are cheap and easily obtained, which is a crucial factor for reliable agrochemical intermediate supplier and pharma supply chains. This methodology effectively bypasses the issues of oxidative decomposition and poor selectivity, providing a streamlined route to valuable cyclohexadienone derivatives.
Mechanistic Insights into Ag2O-Catalyzed Para-Amination
The core of this technological advancement lies in the specific catalytic mechanism facilitated by silver oxide. The reaction proceeds through a transition metal-catalyzed pathway that activates the phenolic substrate for nucleophilic attack by the azodicarboxylate. The silver catalyst plays a pivotal role in directing the amination to the para-position, thereby overcoming the inherent preference for ortho-substitution seen in uncatalyzed or acid-catalyzed reactions. This high regioselectivity is essential for producing high-purity OLED material or pharmaceutical intermediates where isomeric impurities can be detrimental to the final product's efficacy. The mild conditions prevent the oxidative degradation of the electron-rich aromatic ring, ensuring that the structural integrity of the molecule is maintained throughout the transformation. This precise control over the reaction pathway is what distinguishes this method from previous attempts at phenol dearomatization.
Impurity control is another critical aspect where this mechanism excels. The use of water as the reaction medium helps in suppressing side reactions that might occur in organic solvents. The specific interaction between the silver catalyst and the reactants ensures that the formation of by-products is minimized. This results in a cleaner reaction profile, which significantly reduces the burden on purification steps such as column chromatography. For R&D directors focused on purity and impurity profiles, this means a more predictable and controllable synthesis process. The ability to achieve yields as high as 95% in certain examples, as documented in the patent data, underscores the efficiency of this catalytic system. Such high yields are indicative of a robust process that can be reliably scaled for commercial production without compromising on quality.
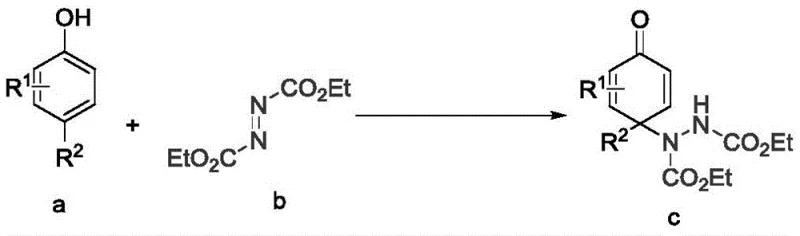
How to Synthesize 4-Amino Substituted Cyclohexadienone Efficiently
The synthesis of these valuable derivatives follows a straightforward protocol that is amenable to standard laboratory and industrial equipment. The process begins with the sequential addition of the catalyst, reactants, and solvent into the reactor, ensuring proper mixing and temperature control. The reaction is then allowed to proceed under ice bath conditions, which is crucial for maintaining the mild environment required for high selectivity. Following the reaction, a standard workup procedure involving extraction and distillation is employed to isolate the crude product. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety considerations. This streamlined process minimizes the need for specialized equipment, making it accessible for a wide range of manufacturing facilities aiming for commercial scale-up of complex polymer additives or fine chemicals.
- Add catalyst Ag2O, reactant I (phenol derivative), reactant II (diethyl azodicarboxylate), and water into the reactor sequentially.
- Place the reactor in an ice bath and stir the mixture for a reaction period ranging from 0.5 to 10 hours to ensure complete conversion.
- Extract the reaction liquid with organic solvent, distill under reduced pressure, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patent offers substantial benefits that directly address the pain points of procurement and supply chain management. The shift to an aqueous phase reaction eliminates the need for large volumes of expensive and hazardous organic solvents. This transition not only reduces the raw material costs but also simplifies waste disposal and environmental compliance procedures. The use of cheap and easily obtained raw materials further enhances the economic viability of the process. For procurement managers, this translates into a more stable and cost-effective supply chain, reducing the risk of disruptions caused by the scarcity of specialized reagents. The simplicity of the reaction conditions also means that the process can be implemented in existing facilities with minimal retrofitting, accelerating the time to market for new products.
- Cost Reduction in Manufacturing: The elimination of expensive organic solvents and the use of readily available catalysts like silver oxide significantly lower the operational expenditures. The high yields reported in the patent examples indicate efficient atom economy, meaning less raw material is wasted. This efficiency directly contributes to substantial cost savings in the overall manufacturing process. Furthermore, the mild reaction conditions reduce energy consumption associated with heating and cooling, adding another layer of cost optimization. These factors combined make the production of these intermediates more economically attractive compared to traditional methods.
- Enhanced Supply Chain Reliability: The reliance on common and easily sourced raw materials ensures a robust supply chain that is less susceptible to market fluctuations. The simplicity of the process reduces the dependency on specialized equipment or hard-to-find reagents. This reliability is crucial for maintaining continuous production schedules and meeting delivery deadlines. For supply chain heads, this means reduced lead time for high-purity pharmaceutical intermediates and a lower risk of production stoppages. The scalability of the process further ensures that supply can be ramped up quickly to meet increasing demand without compromising quality.
- Scalability and Environmental Compliance: The patent explicitly states that the reaction yield is not affected even when the dosage of reactants is enlarged by 100 times. This demonstrates excellent scalability, making it suitable for industrial production. The use of water as a solvent aligns with strict environmental regulations, reducing the environmental footprint of the manufacturing process. This compliance is increasingly important for companies aiming to meet sustainability goals and avoid regulatory penalties. The ease of scale-up ensures that the transition from lab to plant is smooth and efficient.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial aspects of this synthesis method. These answers are derived directly from the patent specifications and are intended to provide clarity on the process capabilities and limitations. Understanding these details is essential for making informed decisions about adopting this technology for your specific application needs. The information provided here reflects the current state of the art as described in the intellectual property documentation.
Q: What are the primary advantages of using water as a solvent in this dearomatization process?
A: Using water as a solvent significantly enhances environmental compliance by eliminating volatile organic compounds (VOCs). It also simplifies the workup procedure and reduces the overall cost of manufacturing by avoiding expensive organic solvents.
Q: How does the silver oxide catalyst affect regioselectivity compared to traditional methods?
A: The silver oxide catalyst promotes high para-selectivity, overcoming the common challenge of ortho-substitution in phenolic compounds. This results in a cleaner impurity profile and higher yields of the desired 4-amino substituted cyclohexadienone derivatives.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the patent data indicates that enlarging the dosage of reactants by 100 times does not negatively affect the yield. This demonstrates excellent scalability and robustness for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino Substituted Cyclohexadienone Supplier
The technical potential of this silver-catalyzed dearomatization route is immense, offering a pathway to high-value intermediates with superior quality. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of this aqueous phase synthesis, ensuring that stringent purity specifications are met consistently. With our rigorous QC labs, we guarantee that every batch of 4-amino substituted cyclohexadienone derivatives meets the highest industry standards. Our commitment to quality and efficiency makes us the ideal partner for bringing this innovative chemistry to the global market.
We invite you to explore how this technology can optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate the viability of this process for your projects. Let us help you leverage this advanced chemistry to gain a competitive edge in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
