Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Earth-Abundant Iron Catalysts for Pharmaceutical Applications
Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Earth-Abundant Iron Catalysts for Pharmaceutical Applications
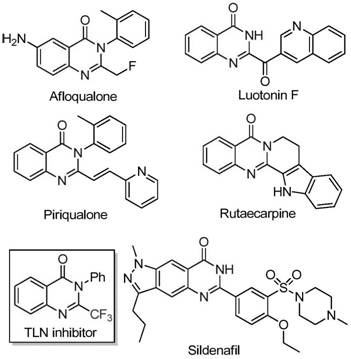
The rapid evolution of medicinal chemistry has increasingly highlighted the critical role of fluorinated heterocycles in modern drug discovery, particularly within the realm of oncology and anti-inflammatory therapeutics. Patent CN111675662B introduces a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, addressing long-standing challenges in synthetic efficiency and cost-effectiveness. Quinazolinone scaffolds are ubiquitous in bioactive natural products and pharmaceutical agents, exhibiting potent anticancer, anticonvulsant, and antifungal properties, yet the introduction of the trifluoromethyl group has historically been a bottleneck due to harsh reaction conditions and expensive reagents. This new methodology leverages earth-abundant iron catalysis to streamline the construction of these complex molecular architectures, offering a robust pathway for the reliable pharmaceutical intermediate supplier seeking to optimize their production pipelines. By shifting away from precious metal catalysis, this technology not only enhances economic viability but also aligns with green chemistry principles, ensuring a sustainable supply chain for high-value active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of trifluoromethyl-substituted quinazolinones has relied heavily on cyclization reactions involving synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate reacting with anthranilamides or isatoic anhydrides. These conventional pathways are frequently plagued by severe operational constraints, including the requirement for stringent anhydrous conditions, elevated temperatures that risk substrate decomposition, and the use of stoichiometric amounts of costly fluorinating agents. Furthermore, the substrate scope in legacy methods is often narrow, failing to accommodate sensitive functional groups which limits the structural diversity accessible to medicinal chemists during lead optimization phases. The reliance on expensive starting materials and the generation of significant chemical waste due to low atom economy further exacerbate the cost burden, making large-scale production economically unfeasible for many generic drug manufacturers. Consequently, there has been a persistent industry demand for a more versatile and economical synthetic strategy that can overcome these thermodynamic and kinetic barriers without sacrificing yield or purity.
The Novel Approach
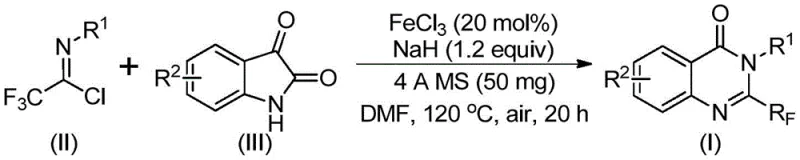
The innovative strategy disclosed in the patent utilizes readily available trifluoroethylimidoyl chloride and isatin derivatives as key building blocks, catalyzed by inexpensive ferric chloride to drive the cyclization process. This novel approach fundamentally alters the reaction landscape by enabling a tandem sequence of alkali-promoted carbon-nitrogen bond formation followed by iron-catalyzed decarbonylation and intramolecular cyclization. Unlike previous methods that struggle with functional group compatibility, this system exhibits remarkable tolerance, successfully synthesizing derivatives with methyl, halogen, and methoxy substituents at ortho-, meta-, and para-positions with high efficiency. The reaction proceeds smoothly in polar aprotic solvents like DMF under aerobic conditions, eliminating the need for complex inert atmosphere setups that typically increase operational complexity and capital expenditure. By utilizing isatins, which are commodity chemicals, the method drastically reduces the raw material cost baseline, positioning this technology as a superior choice for cost reduction in API manufacturing where margin compression is a constant pressure.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of nucleophilic substitution and transition metal-mediated bond activation, initiated by the deprotonation of the isatin nitrogen by sodium hydride to generate a reactive nucleophile. This nucleophile attacks the electrophilic carbon of the trifluoroethylimidoyl chloride, forming a transient trifluoroacetamidine intermediate which serves as the precursor for the subsequent cyclization event. The presence of ferric chloride is pivotal, as it likely coordinates with the carbonyl oxygen or the imine nitrogen to facilitate the decarbonylation step, a process that is energetically demanding under standard thermal conditions alone. This iron-mediated activation lowers the energy barrier for the extrusion of carbon monoxide, allowing the system to rearrange into the thermodynamically stable quinazolinone core while retaining the critical trifluoromethyl group at the 2-position. The use of 4A molecular sieves in the reaction mixture plays a crucial role in scavenging trace moisture, which could otherwise hydrolyze the sensitive imidoyl chloride or deactivate the base, thereby ensuring consistent reaction kinetics and high reproducibility across different batches.
From an impurity control perspective, the mechanism inherently minimizes side reactions such as homocoupling or over-fluorination, which are common pitfalls in fluorine chemistry. The specific stoichiometry of 1.2 equivalents of sodium hydride ensures complete deprotonation of the isatin without causing excessive degradation of the sensitive trifluoromethyl moiety, while the 20 mol% loading of FeCl3 provides sufficient catalytic turnover without introducing overwhelming levels of metal contaminants. The reaction temperature profile, starting at 40°C for initial adduct formation and ramping to 120°C for cyclization, allows for precise control over the reaction trajectory, preventing the formation of polymeric byproducts that often occur at single high-temperature steps. This controlled thermal progression, combined with the chemoselectivity of the iron catalyst, results in a clean crude reaction profile that simplifies downstream purification, ultimately delivering high-purity OLED material or pharmaceutical intermediates with minimal chromatographic effort.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis is designed for operational simplicity, requiring standard laboratory equipment and avoiding the need for specialized high-pressure reactors or glovebox techniques. The process begins with the careful addition of ferric chloride and sodium hydride to a reaction vessel containing the substrate mixture in DMF, followed by the incorporation of activated 4A molecular sieves to maintain anhydrous conditions throughout the extended reaction period. The two-stage heating protocol is critical; maintaining the mixture at 40°C for the first 8 to 10 hours allows for the gentle formation of the C-N bond, while the subsequent heating to 120°C for 18 to 20 hours drives the irreversible decarbonylation and ring closure to completion. Upon cooling, the reaction mixture is filtered to remove the molecular sieves and inorganic salts, and the filtrate is concentrated and purified via silica gel column chromatography to isolate the target compound. For detailed procedural specifics regarding reagent grades, workup protocols, and safety considerations, please refer to the standardized synthesis guide below.
- Mix ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in anhydrous DMF.
- Stir the reaction mixture at 40°C for 8-10 hours to initiate the nucleophilic attack and intermediate formation.
- Heat the reaction to 120°C under air atmosphere for 18-20 hours to complete the decarbonylation and cyclization, followed by filtration and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed route represents a strategic opportunity to de-risk the supply of critical heterocyclic intermediates while simultaneously driving down the cost of goods sold. The shift from precious metal catalysts to ferric chloride removes a significant volatility factor from the raw material basket, as iron salts are abundant, price-stable commodities compared to fluctuating palladium or rhodium markets. Furthermore, the use of isatins and imidoyl chlorides as starting materials leverages existing global supply chains for bulk chemicals, ensuring that sourcing remains resilient even during periods of geopolitical disruption or logistics bottlenecks. The robustness of the reaction conditions, specifically the tolerance to air and moisture relative to other organometallic processes, reduces the likelihood of batch failures due to environmental excursions, thereby enhancing overall supply chain reliability and predictability for downstream customers.
- Cost Reduction in Manufacturing: The elimination of expensive ligands and precious metals directly translates to substantial cost savings in the bill of materials, while the simplified workup procedure reduces solvent consumption and labor hours associated with complex metal scavenging steps. By avoiding the need for cryogenic conditions or ultra-high purity inert gases, the utility costs per kilogram of product are significantly lowered, improving the overall margin profile for commercial scale-up of complex pharmaceutical intermediates. Additionally, the high yields reported across a broad substrate scope mean that less starting material is wasted, maximizing the value extracted from every pound of raw material purchased and processed through the facility.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like isatins and iron chloride ensures that the supply chain is not dependent on single-source suppliers of exotic reagents, mitigating the risk of shortages that can halt production lines. The reaction's ability to proceed under aerobic conditions simplifies the engineering requirements for manufacturing plants, allowing for production in standard glass-lined or stainless steel reactors without the need for specialized nitrogen-blanketed infrastructure. This flexibility enables faster technology transfer between sites and easier scaling from pilot plant to full commercial production, ensuring that reducing lead time for high-purity pharmaceutical intermediates becomes a achievable reality rather than just a goal.
- Scalability and Environmental Compliance: The use of DMF as a solvent, while requiring careful handling, is well-understood in industrial settings with established recovery and recycling protocols that minimize environmental impact and waste disposal costs. The absence of toxic heavy metals in the catalyst system simplifies the regulatory burden associated with residual metal testing in the final drug substance, accelerating the path to regulatory approval and market entry. Moreover, the high atom economy of the cyclization process, which incorporates most of the starting material atoms into the final product, aligns with modern sustainability mandates and reduces the volume of hazardous waste generated per unit of product manufactured.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic methodology, derived directly from the experimental data and beneficial effects outlined in the patent documentation. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this route into their existing process development workflows. The answers provided reflect the specific capabilities of the iron-catalyzed system in handling diverse substrates and its performance metrics under optimized conditions.
Q: What are the primary advantages of using FeCl3 over precious metal catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) eliminates the need for expensive palladium or rhodium catalysts, significantly reducing raw material costs and simplifying the removal of heavy metal residues, which is critical for pharmaceutical compliance.
Q: Can this synthetic route tolerate diverse functional groups on the isatin substrate?
A: Yes, the method demonstrates excellent functional group tolerance, successfully accommodating substituents such as methyl, fluoro, bromo, chloro, and methoxy groups at various positions on the aromatic ring without compromising yield.
Q: Is this process suitable for large-scale industrial production?
A: The protocol utilizes commercially available reagents, operates under aerobic conditions without inert gas protection, and employs simple workup procedures like filtration and chromatography, making it highly amenable to kilogram-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed technology in advancing the development of next-generation therapeutics targeting oncology and inflammatory diseases. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to market supply is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone delivered meets the highest international standards for pharmaceutical applications. We are committed to leveraging our technical expertise to optimize this specific route further, tailoring the process to your unique volume requirements and quality targets.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be integrated into your supply chain to achieve significant operational efficiencies. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this iron-catalyzed method for your specific project needs. We encourage you to contact us today to obtain specific COA data for our catalog compounds and to schedule a consultation for route feasibility assessments, ensuring your project stays on track for successful commercialization.
