Revolutionizing Ergothioneine Production: A Scalable 4-Step Chemical Synthesis Strategy
Revolutionizing Ergothioneine Production: A Scalable 4-Step Chemical Synthesis Strategy
The global demand for ergothioneine, a naturally occurring amino acid with potent antioxidant properties, has surged due to its recognition as a vital dietary supplement for cellular protection and DNA maintenance. However, traditional sourcing methods, such as extraction from mushrooms or complex biosynthetic fermentation, often struggle to meet the volume and consistency required by the modern nutraceutical and pharmaceutical industries. Addressing this critical supply gap, the recent technological disclosure in patent CN116102502A introduces a robust, four-step chemical synthesis pathway that fundamentally alters the production landscape. This method leverages inexpensive histidine derivatives as starting materials, bypassing the biological limitations of fungal cultivation while delivering a product with high optical purity and structural integrity. For industry stakeholders, this represents a pivotal shift towards a more reliable and economically viable supply chain for high-value antioxidants.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of ergothioneine has been fraught with significant logistical and economic challenges inherent to biological sourcing. Extraction from natural sources like ergot fungi or mushrooms is not only labor-intensive but also subject to severe fluctuations in raw material availability due to seasonal agricultural cycles and geographic constraints. Furthermore, the purification of ergothioneine from complex biological matrices often requires extensive chromatography, leading to substantial product loss and elevated operational costs. On the other hand, existing chemical synthesis routes reported in prior art have frequently suffered from excessive step counts, often exceeding six or seven distinct transformations. These lengthy sequences invariably result in cumulative yield erosion, where the efficiency of each individual step compounds negatively, rendering the final process economically unfeasible for kilogram-to-ton scale manufacturing. Additionally, many legacy chemical methods rely on harsh reaction conditions or expensive chiral catalysts that introduce difficulties in waste management and regulatory compliance.
The Novel Approach
In stark contrast to these inefficient legacy processes, the methodology outlined in the patent data presents a streamlined four-step sequence (Steps a through d) that maximizes atom economy and operational simplicity. The core innovation lies in the strategic protection and functionalization of the histidine backbone, allowing for precise control over the regioselectivity of subsequent reactions. By utilizing a specific protection-methylation-deprotection-sulfuration logic, the route effectively constructs the unique imidazole-2-thione moiety characteristic of ergothioneine without compromising the stereochemistry of the alpha-carbon. This approach eliminates the need for complex enzymatic cascades or difficult-to-source biological precursors. The reaction scheme below illustrates this concise transformation, highlighting how a simple histidine derivative is converted into the final quaternary ammonium zwitterion through a logical progression of stable intermediates.
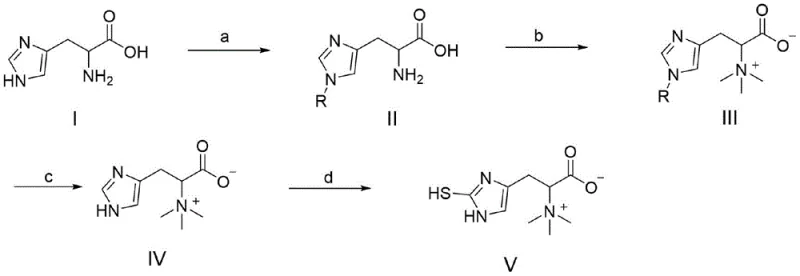
The visual representation of the synthesis underscores the modularity of the process. Step (a) involves the protection of the imidazole nitrogen, a crucial maneuver to prevent unwanted side reactions during the subsequent quaternization. Step (b) introduces the trimethylammonium group, establishing the permanent positive charge essential for the molecule's biological transport mechanisms. Step (c) cleanly removes the protecting group under mild acidic hydrolysis, and finally, Step (d) installs the sulfur functionality via a halogenation-nucleophilic substitution sequence. This linear progression ensures that each intermediate can be isolated and characterized, providing quality control checkpoints that are often absent in one-pot biosynthetic fermentations.
Mechanistic Insights into Regioselective Protection and Sulfuration
From a mechanistic perspective, the success of this synthesis hinges on the careful management of the imidazole ring's reactivity. The imidazole nucleus in histidine contains two nitrogen atoms with distinct electronic environments, and selective protection is paramount to ensuring the sulfur atom ends up at the C2 position rather than N1 or N3. The patent specifies the use of bulky protecting groups such as trityl (triphenylmethyl), benzhydryl, or benzyl moieties. These groups provide significant steric hindrance, directing the initial alkylation and subsequent reactions away from the protected nitrogen. This steric control is vital for maintaining the structural fidelity of the final product. Furthermore, the final sulfuration step (Step d) employs a sophisticated halogenation strategy using reagents like dibromohydantoin or bromosuccinimide. This generates an electrophilic intermediate at the C2 position of the imidazole ring, which is then attacked by a sulfur nucleophile such as L-cysteine or ammonium thiocyanate. This mechanism avoids the use of toxic hydrogen sulfide gas often utilized in older thione syntheses, thereby enhancing the safety profile of the manufacturing process.
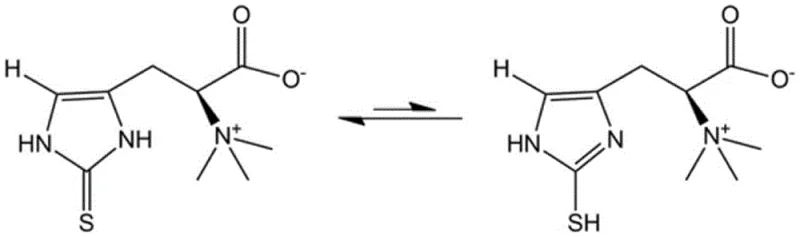
Another critical aspect of the chemical behavior of ergothioneine is its tendency to exist in equilibrium between thione and thiol tautomeric forms, a phenomenon that impacts its stability and analytical characterization. As depicted in the structural analysis below, the molecule predominantly exists in the thione form at physiological pH, but the synthetic process must account for potential oxidation to cystine or disulfide byproducts. The described method mitigates this by controlling the redox environment during the final crystallization steps. The use of specific recrystallization solvents, such as isopropanol and water mixtures, helps to stabilize the desired tautomer and exclude cysteine impurities. Understanding this tautomeric equilibrium is essential for R&D teams aiming to replicate the process, as slight variations in pH or solvent polarity during workup could shift the equilibrium and affect the apparent purity or melting point of the isolated solid.
How to Synthesize L-Ergothioneine Efficiently
Implementing this synthesis requires strict adherence to the reaction parameters defined in the patent to ensure optimal yield and purity. The process begins with the selection of high-quality L-histidine as the chiral pool starting material, which dictates the optical purity of the final ergothioneine. The protection step (Step a) is typically conducted in chlorinated solvents like dichloromethane at controlled temperatures (10-40°C) to prevent racemization. Following isolation, the methylation step (Step b) utilizes methylating agents like dimethyl sulfate in polar protic solvents such as methanol. The subsequent hydrolysis (Step c) requires careful pH adjustment to remove the protecting group without degrading the sensitive imidazole ring. Finally, the sulfuration (Step d) is the most critical stage, requiring precise stoichiometry of the halogenating agent and sulfur source to minimize the formation of disulfide byproducts. For a detailed, standardized operating procedure including exact reagent quantities and workup protocols, please refer to the technical guide below.
- Step A: Protect the imidazole nitrogen of histidine using trityl or benzyl protecting groups in a chlorinated solvent with a base.
- Step B: Methylate the alpha-amino group using dimethyl sulfate or methyl iodide to form the quaternary ammonium intermediate.
- Step C: Hydrolyze the protecting group under acidic conditions to regenerate the imidazole ring.
- Step D: Perform halogenation followed by reaction with a sulfur source like L-cysteine or ammonium thiocyanate to install the thione/thiol functionality.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from biological extraction to this defined chemical synthesis offers transformative advantages in terms of cost structure and supply reliability. The primary driver of cost reduction is the replacement of expensive, variable biological feedstocks with commodity chemicals. Histidine, trityl chloride, and dimethyl sulfate are produced on a massive industrial scale, ensuring that their prices remain stable and predictable regardless of agricultural harvest conditions. This decoupling from biological cycles eliminates the risk of supply shocks caused by crop failures or fungal contamination, which have historically plagued the ergothioneine market. Furthermore, the shortened synthetic route directly correlates to reduced processing time and lower utility consumption. Fewer reaction steps mean fewer unit operations, less solvent usage, and reduced labor hours per kilogram of finished product, all of which contribute to a significantly lower cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The economic benefits of this route are derived from the elimination of costly purification bottlenecks associated with fermentation broths. In biological processes, separating the target molecule from a soup of cellular debris and metabolic byproducts often requires expensive resin columns and large volumes of water. In contrast, this chemical route produces intermediates that can be purified via simple filtration and crystallization, techniques that are inherently cheaper and easier to scale. Additionally, the avoidance of transition metal catalysts removes the need for expensive scavenging resins to meet heavy metal specifications, further driving down the cost reduction in antioxidant manufacturing. The high yields reported in the early steps (often exceeding 90%) ensure that raw material waste is minimized, maximizing the value extracted from every kilogram of histidine purchased.
- Enhanced Supply Chain Reliability: Supply continuity is a paramount concern for manufacturers of finished nutraceutical formulations. This synthetic method enhances reliability by utilizing a robust inventory of chemically stable intermediates. Unlike live cultures or fresh mushroom extracts which have limited shelf lives, the protected histidine intermediates described in Steps II and III are stable solids that can be stockpiled strategically to buffer against demand spikes. This capability allows suppliers to maintain consistent lead times even during periods of surging market demand. Moreover, the use of standard organic solvents and reagents means that production can be easily transferred between different manufacturing sites globally without the need for specialized bioreactor infrastructure, creating a more resilient and geographically diversified supply network for high-purity nutraceutical ingredients.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces heat transfer and mixing challenges, but this route is designed with scalability in mind. The reaction temperatures are moderate (mostly below 100°C), and the exotherms are manageable, allowing for safe operation in large-scale glass-lined steel reactors. From an environmental perspective, the process generates less biological waste compared to fermentation, simplifying wastewater treatment protocols. While the use of chlorinated solvents requires proper recovery systems, the overall waste profile is more defined and easier to treat than the complex organic load of a fermentation broth. This facilitates easier regulatory approval and environmental compliance, reducing the administrative burden on the supply chain team and ensuring long-term operational sustainability for commercial scale-up of complex amino acid derivatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ergothioneine synthesis technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on yield expectations, impurity profiles, and process robustness. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement specialists negotiating supply contracts.
Q: How does this synthetic route improve yield compared to extraction?
A: Unlike natural extraction which is limited by fungal biomass and seasonal variability, this 4-step chemical synthesis utilizes readily available histidine starting materials. The patent data indicates step-wise yields exceeding 90% in protection and hydrolysis steps, significantly boosting overall throughput compared to biological fermentation bottlenecks.
Q: What are the critical purity considerations for ergothioneine synthesis?
A: The process addresses the challenge of tautomeric stability (thione vs. thiol forms) and residual cysteine impurities. The described method includes specific recrystallization protocols using isopropanol and water mixtures to ensure the final product meets stringent purity specifications required for nutraceutical applications, minimizing toxicological risks associated with heavy metal catalysts often found in alternative routes.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the route is designed for scalability. It avoids cryogenic conditions and exotic reagents, relying on standard solvents like dichloromethane, methanol, and water. The reaction temperatures are moderate (mostly 20-80°C), allowing for implementation in standard glass-lined or stainless steel reactors without requiring specialized high-pressure or low-temperature infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ergothioneine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a new synthetic route must be translated into tangible commercial reality. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facility is equipped with state-of-the-art reactor trains capable of handling the specific solvent systems and temperature profiles required for this ergothioneine synthesis, including dedicated lines for chlorinated solvent recovery and crystallization. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the high standards required for human consumption, effectively mitigating the risks associated with impurity carryover from earlier synthetic steps.
We invite forward-thinking partners to collaborate with us to leverage this advanced manufacturing technology. Whether you require a Customized Cost-Saving Analysis to compare this chemical route against your current biosynthetic supply or need specific COA data to validate the impurity profile against your internal standards, our technical procurement team is ready to assist. We encourage you to reach out for detailed route feasibility assessments and to discuss how we can secure your supply chain with high-purity ergothioneine produced through this innovative, scalable, and cost-effective chemical process.
