Revolutionizing Spiro-Sultam Production: A Robust Rhodium-Catalyzed Platform for Pharmaceutical Intermediates
Revolutionizing Spiro-Sultam Production: A Robust Rhodium-Catalyzed Platform for Pharmaceutical Intermediates
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, sustainable, and biologically potent chemical scaffolds. A significant breakthrough in this domain is detailed in patent CN113444107B, which discloses a novel synthetic method for succinimide spiro-fused sultam compounds. These unique heterocyclic structures are of immense interest in medicinal chemistry due to their rigid three-dimensional geometry and proven potential as calcium-sensing receptor agonists and HIV-1 protease inhibitors. The patent introduces a highly efficient tandem reaction between arylsulfonamides and N-substituted maleimides, catalyzed by a rhodium complex. This approach not only streamlines the construction of the complex spiro-core but also ensures high atom economy and operational simplicity, addressing critical pain points in modern drug discovery pipelines.
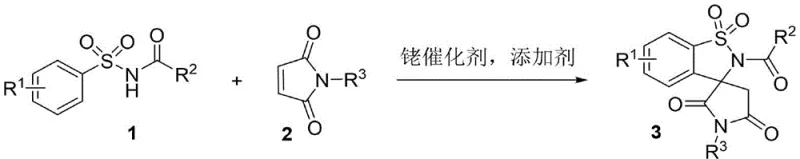
For procurement managers and supply chain heads, the implications of this technology are profound. By shifting away from legacy multi-step syntheses towards this direct catalytic coupling, manufacturers can achieve cost reduction in pharmaceutical intermediates manufacturing through simplified processing and reduced waste generation. The ability to access a diverse library of these bioactive scaffolds rapidly positions this methodology as a cornerstone for developing next-generation anticancer agents, specifically targeting hematologic malignancies like REC-1 and Ramos cell lines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spirocyclic sultams has been fraught with significant technical and safety challenges that hindered their widespread adoption in commercial drug production. Prior art, such as the method reported by Wrobel, relied on a cumbersome multi-step sequence involving the condensation of o-benzoylsulfonylimide with ethyl 2-cyanoacetate. This legacy pathway necessitated the use of hazardous reagents like potassium cyanide (KCN) for subsequent cyanation steps, followed by intramolecular cyclization and decarboxylation. From a process safety and environmental compliance perspective, handling large quantities of cyanide salts presents unacceptable risks and requires specialized containment infrastructure. Furthermore, the overall atom economy of such linear syntheses is inherently poor, leading to substantial material loss and increased disposal costs for chemical waste. The prolonged reaction times and low total yields associated with these traditional routes further exacerbate production costs, making the final high-purity pharmaceutical intermediates economically unviable for large-scale applications.
The Novel Approach
In stark contrast, the methodology described in CN113444107B represents a paradigm shift towards green and efficient chemical synthesis. This novel approach utilizes a direct tandem reaction between readily available arylsulfonamides and N-substituted maleimides in the presence of a rhodium catalyst and a simple inorganic base additive. The reaction proceeds efficiently under air atmosphere, eliminating the need for rigorous inert gas protection which often complicates reactor operations. By forging the spiro-center and the sultam ring simultaneously in a single pot, this method drastically reduces the number of unit operations required. The use of benign solvents like ethyl acetate further enhances the environmental profile of the process. This streamlined workflow not only accelerates the commercial scale-up of complex pharmaceutical intermediates but also ensures a cleaner impurity profile, reducing the burden on downstream purification teams and facilitating faster regulatory approval timelines for new drug candidates.
Mechanistic Insights into Rhodium-Catalyzed Tandem Cyclization
The success of this synthesis hinges on the precise orchestration of a rhodium(III)-catalyzed C-H activation cascade. The mechanism initiates with the coordination of the sulfonamide nitrogen to the cationic rhodium species generated in situ from the dimeric precatalyst [RhCp*Cl2]2. This coordination directs the metal center to activate the ortho-C-H bond of the aryl ring, forming a stable five-membered rhodacycle intermediate. Subsequently, the electron-deficient double bond of the N-substituted maleimide inserts into the Rh-C bond. This insertion step is critical as it establishes the carbon-carbon bond that will eventually become part of the spiro-junction. Following migratory insertion, an intramolecular nucleophilic attack by the sulfonamide nitrogen onto the carbonyl group of the maleimide moiety occurs, closing the sultam ring and releasing the rhodium catalyst to re-enter the cycle. This elegant mechanistic pathway ensures high regioselectivity and minimizes the formation of byproducts, which is essential for maintaining the integrity of the final active pharmaceutical ingredient.
From an R&D perspective, understanding the role of additives and ligands is vital for optimizing yield and purity. The patent highlights sodium acetate (NaOAc) as the optimal additive, likely serving to neutralize the HCl byproduct generated during catalyst activation and to facilitate the C-H cleavage step via a concerted metalation-deprotonation (CMD) mechanism. The choice of solvent also plays a pivotal role; ethyl acetate was found to provide superior yields compared to polar aprotic solvents like DMSO or chlorinated solvents like DCE. This solvent compatibility is a significant advantage for process chemists, as ethyl acetate is easier to remove and recycle than high-boiling polar solvents. The robustness of this catalytic system allows for a wide tolerance of functional groups, enabling the synthesis of diverse derivatives without the need for extensive protecting group strategies, thereby simplifying the overall synthetic design.
How to Synthesize Succinimide Spiro-Fused Sultams Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and pilot plant equipment. The general procedure involves charging a reaction vessel with the arylsulfonamide substrate, the N-substituted maleimide coupling partner, the rhodium catalyst, and the sodium acetate additive in ethyl acetate. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are outlined in the guide below.
- Combine arylsulfonamide and N-substituted maleimide in a reaction vessel with a rhodium catalyst such as [RhCp*Cl2]2.
- Add an inorganic base additive like sodium acetate and an organic solvent such as ethyl acetate to the mixture.
- Heat the reaction mixture to 120°C under air atmosphere for approximately 10 hours to facilitate the tandem cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the transition to this rhodium-catalyzed platform offers tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Unlike legacy methods requiring specialized and hazardous reagents like cyanides, this process relies on commodity chemicals—sulfonamides and maleimides—that are widely available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source bottlenecks and price volatility. Furthermore, the elimination of toxic reagents reduces the regulatory burden associated with hazardous material storage and transport, leading to lower insurance and compliance costs. The high atom economy of the tandem reaction means that less raw material is wasted, directly contributing to cost reduction in fine chemical manufacturing by maximizing the output per kilogram of input.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by its operational efficiency. By consolidating multiple synthetic steps into a single tandem reaction, manufacturers save significantly on labor, energy, and solvent consumption. The removal of the cyanation and decarboxylation steps eliminates the need for specialized waste treatment facilities required for cyanide detoxification. Additionally, the use of earth-abundant rhodium catalysts in low loading amounts (typically 2-5 mol%) ensures that catalyst costs do not prohibitive, especially when considering the high value of the resulting anticancer intermediates. The simplified purification process, often requiring only standard silica gel chromatography or crystallization, further reduces the cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Reliability is paramount in the pharmaceutical supply chain, and this method enhances it through robustness and flexibility. The reaction tolerates a wide range of substituents on both the sulfonamide and maleimide components, as evidenced by the extensive substrate scope demonstrated in the patent data. This flexibility allows manufacturers to quickly pivot between different analogues to meet changing R&D demands without retooling the entire production line. Moreover, the reaction proceeds under air atmosphere, removing the dependency on high-purity nitrogen or argon supplies, which can be a logistical constraint in some manufacturing regions. This operational resilience ensures consistent delivery schedules and reduces lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden inefficiencies, but this protocol is designed with scalability in mind. The use of ethyl acetate, a solvent with a favorable safety profile and low environmental impact, aligns with modern green chemistry principles and corporate sustainability goals. The reaction temperature of 120°C is easily achievable with standard heating media like thermal oil, avoiding the need for extreme high-pressure or cryogenic conditions. The high yields observed across various substrates indicate that the process is robust against minor variations in reaction parameters, a key requirement for successful technology transfer from lab to plant. This ease of scale-up facilitates the rapid production of clinical trial materials and eventual commercial batches, ensuring a continuous supply for downstream drug formulation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these spiro-sultam compounds. The answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing a reliable foundation for decision-making.
Q: What are the advantages of this rhodium-catalyzed method over traditional synthesis?
A: This method eliminates the need for toxic potassium cyanide and multi-step sequences found in older protocols, offering a one-pot tandem reaction with superior atom economy and operational simplicity.
Q: What is the biological activity profile of these spiro-sultam compounds?
A: The synthesized compounds exhibit significant anticancer activity, particularly against REC-1 and Ramos cancer cell lines, with IC50 values demonstrating potent antiproliferative effects compared to controls.
Q: Is this process scalable for commercial manufacturing?
A: Yes, the process utilizes readily available starting materials, operates under air atmosphere without stringent inert gas requirements, and uses common solvents like ethyl acetate, making it highly suitable for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Succinimide Spiro-Fused Sultams Supplier
As the demand for sophisticated heterocyclic scaffolds in oncology research continues to surge, partnering with an experienced CDMO is crucial for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing rhodium-catalyzed processes to ensure stringent purity specifications are met consistently. With state-of-the-art rigorous QC labs, we guarantee that every batch of succinimide spiro-fused sultams meets the highest international standards, ready for immediate integration into your drug discovery programs.
We invite you to leverage our expertise to accelerate your project timelines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and reduce overall project costs.
