Scalable Rhodium-Catalyzed Synthesis of Anticancer Succinimide Spiro-Fused Sultams for Pharmaceutical Applications
Scalable Rhodium-Catalyzed Synthesis of Anticancer Succinimide Spiro-Fused Sultams for Pharmaceutical Applications
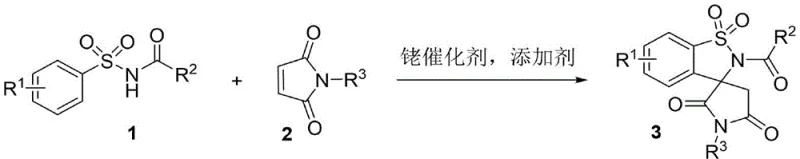
The pharmaceutical industry continuously seeks novel scaffolds that offer unique three-dimensional geometries to improve binding affinity and metabolic stability in drug candidates. Patent CN113444107B introduces a groundbreaking synthetic methodology for constructing succinimide spiro-fused sultam compounds, a privileged structural motif known for diverse biological activities ranging from antibacterial to HIV-1 protease inhibition. This technology leverages a rhodium-catalyzed tandem reaction between arylsulfonamides and N-substituted maleimides, providing a direct and efficient route to these complex heterocycles. For R&D directors and procurement specialists, this innovation represents a significant leap forward in accessing high-value pharmaceutical intermediates with reduced synthetic complexity. The ability to generate such intricate spirocyclic cores in a single operational step addresses critical bottlenecks in early-stage drug discovery and process development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of spiro-fused sultam skeletons has been fraught with synthetic challenges that hinder their widespread adoption in medicinal chemistry programs. Prior art, such as the method reported by Wrobel, relies on a cumbersome multi-step sequence involving the condensation of o-benzoylsulfonylimine with ethyl 2-cyanoacetate, followed by hazardous cyanation with potassium cyanide (KCN), intramolecular cyclization, and subsequent decarboxylation. This traditional pathway suffers from severe drawbacks, including poor atom economy, low overall yields due to cumulative losses across multiple isolation steps, and the use of highly toxic reagents that complicate waste management and operator safety. Furthermore, the lengthy reaction times and stringent conditions required for each transformation increase the cost of goods sold (COGS) and extend the lead time for obtaining material for biological testing, creating a significant barrier for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN113444107B utilizes a streamlined one-pot tandem reaction strategy that dramatically simplifies the synthetic landscape. By employing a cationic rhodium(III) catalyst system, specifically [RhCp*Cl2]2, in conjunction with a simple acetate additive, the process directly couples readily available arylsulfonamides with N-substituted maleimides. This approach eliminates the need for pre-functionalized substrates or dangerous cyanation steps, operating efficiently under neutral oxidative conditions. The reaction proceeds through a cascade of C-H activation and annulation events to forge the spiro-center and the sultam ring simultaneously. This not only enhances the step economy but also improves the environmental profile of the synthesis by reducing solvent consumption and waste generation, aligning perfectly with modern green chemistry principles demanded by global supply chains.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Annulation
The core of this technological breakthrough lies in the sophisticated organometallic mechanism driven by the pentamethylcyclopentadienyl rhodium(III) complex. The catalytic cycle initiates with the coordination of the sulfonamide nitrogen to the rhodium center, facilitating a directed ortho-C-H activation of the aromatic ring to form a stable five-membered rhodacycle intermediate. This key metallacycle then undergoes migratory insertion of the electron-deficient alkene moiety from the N-substituted maleimide. Subsequent nucleophilic attack by the sulfonamide nitrogen onto the activated carbonyl or imine species triggers the cyclization event, ultimately releasing the spiro-fused product and regenerating the active catalyst species. Understanding this mechanism is crucial for R&D teams aiming to optimize reaction parameters, as the electronic properties of the substituents on both the sulfonamide and maleimide rings can significantly influence the rate of C-H cleavage and the stability of the transition states involved in the spiro-ring formation.
From an impurity control perspective, the choice of additives and solvents plays a pivotal role in ensuring high purity profiles essential for API intermediate production. The patent data indicates that sodium acetate (NaOAc) serves as an optimal base to facilitate the C-H activation step without promoting side reactions such as polymerization of the maleimide or hydrolysis of the sultam ring. Furthermore, the use of ethyl acetate (EA) as the preferred solvent not only provides excellent solubility for the organic substrates but also offers a safer, more sustainable alternative to chlorinated solvents often used in similar transformations. The robustness of the catalyst system allows the reaction to proceed effectively even under air atmosphere, minimizing the need for rigorous degassing procedures and inert gas protection, which simplifies the engineering requirements for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Succinimide Spiro-Fused Sultams Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The standard protocol involves charging a reaction vessel with the arylsulfonamide substrate, the N-substituted maleimide coupling partner, the rhodium dimer catalyst, and sodium acetate in ethyl acetate. The mixture is then heated to 120°C for approximately 10 hours, allowing the tandem cyclization to reach completion. Workup is straightforward, involving aqueous quenching followed by extraction and standard silica gel chromatography. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and quality consistency across different batches.
- Combine arylsulfonamide and N-substituted maleimide in ethyl acetate with [RhCp*Cl2]2 catalyst and sodium acetate additive.
- Heat the reaction mixture to 120°C under air atmosphere for 10 hours to facilitate the tandem cyclization.
- Quench with water, extract with dichloromethane, and purify via silica gel chromatography to isolate the target sultam.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this rhodium-catalyzed methodology offers tangible strategic benefits beyond mere chemical elegance. The primary advantage stems from the drastic simplification of the supply chain for raw materials; arylsulfonamides and maleimides are commodity chemicals available from multiple global vendors, reducing the risk of single-source dependency. Additionally, the elimination of toxic reagents like potassium cyanide removes the need for specialized hazardous waste disposal contracts and extensive safety monitoring infrastructure, leading to substantial operational cost savings. The high atom economy of the tandem reaction ensures that a greater proportion of the input mass is converted into the desired product, minimizing raw material costs per kilogram of output and enhancing the overall sustainability metrics of the manufacturing process.
- Cost Reduction in Manufacturing: The one-pot nature of this synthesis significantly lowers processing costs by consolidating multiple reaction steps into a single unit operation. This consolidation reduces labor hours, energy consumption for heating and cooling cycles, and solvent usage associated with intermediate isolations. Furthermore, the use of earth-abundant iron or cobalt alternatives was tested but found inferior, validating the specific value of the rhodium catalyst which, despite being a precious metal, is used in low loading (typically 2-5 mol%) and can potentially be recovered or recycled in large-scale continuous flow setups, further driving down the effective catalyst cost per batch.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, specifically its tolerance to air and moisture, greatly enhances supply chain reliability by reducing the sensitivity of the manufacturing process to environmental fluctuations. This resilience minimizes the risk of batch failures due to minor deviations in inert gas quality or seal integrity, ensuring consistent delivery schedules. Moreover, the broad substrate scope demonstrated in the patent means that a single platform technology can be adapted to produce a wide library of analogues by simply swapping commercially available starting materials, allowing for rapid response to changing market demands or clinical trial requirements without re-validating entirely new processes.
- Scalability and Environmental Compliance: Scaling this process is facilitated by the use of ethyl acetate, a Class 3 solvent with low toxicity and favorable environmental regulations compared to dichloromethane or DMF. The absence of gaseous byproducts or exothermic runaway risks associated with cyanation steps makes the process inherently safer for large-scale reactors. The high yields observed across a diverse range of substrates, including those with electron-withdrawing and electron-donating groups, indicate that the process is chemically robust and less prone to generating difficult-to-remove impurities, thereby simplifying downstream purification and ensuring compliance with strict residual solvent and heavy metal guidelines required for pharmaceutical applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: What are the advantages of this Rh-catalyzed method over traditional synthesis?
A: Unlike traditional multi-step methods requiring toxic cyanation reagents, this one-pot Rh-catalyzed tandem reaction offers higher atom economy, simpler operation, and avoids hazardous steps, significantly improving process safety and efficiency.
Q: Is this synthesis method scalable for commercial production?
A: Yes, the method utilizes robust conditions (air atmosphere, common solvents like ethyl acetate) and readily available starting materials, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the biological potential of these spiro-fused sultam compounds?
A: The synthesized compounds exhibit significant anticancer activity, particularly against REC-1 and Ramos cancer cell lines, with some derivatives showing IC50 values in the sub-micromolar range, indicating strong potential for drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Succinimide Spiro-Fused Sultams Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this rhodium-catalyzed spirocyclization technology in accelerating the development of next-generation anticancer therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of succinimide spiro-fused sultams delivered meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your specific project needs. Contact us today to request a Customized Cost-Saving Analysis tailored to your target molecules. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments, helping you secure a reliable supply chain for these high-value anticancer scaffolds while maximizing your R&D investment.
