Advanced Rare Earth Catalysis for Efficient Indole Derivative Manufacturing and Commercial Scale-Up
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing nitrogen-containing heterocycles, particularly indole scaffolds which serve as privileged structures in countless bioactive molecules. A significant technological breakthrough in this domain is detailed in Chinese Patent CN110818608B, which discloses a novel application of rare earth silamides as catalysts for the preparation of indole and its derivatives. This innovation addresses long-standing challenges in synthetic efficiency by utilizing a hydrogen transfer reduction strategy that converts readily available 2-indolones into valuable indole products. The protocol leverages the unique Lewis acidity and coordination properties of rare earth elements, specifically employing catalysts of the formula M[N(SiMe3)2]3 where M represents elements such as Yttrium, Lanthanum, or Neodymium. By integrating pinacolborane as a mild and stable hydrogen source, this methodology circumvents the need for hazardous reducing agents or expensive noble metal catalysts, thereby establishing a new benchmark for sustainable and scalable chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indole derivatives has relied heavily on classical transformations such as the Fischer indole synthesis, which, while foundational, suffers from significant limitations regarding substrate scope and selectivity. The Fischer method typically requires phenylhydrazine derivatives and ketones, restricting the diversity of accessible molecular architectures and often leading to complex mixtures that are difficult to purify on an industrial scale. Furthermore, alternative modern approaches have introduced severe safety and cost liabilities; for instance, certain cyclization reactions necessitate the use of tert-butyllithium, a pyrophoric reagent that poses extreme operational risks and requires specialized handling infrastructure. Other reported methods utilize platinum-based catalysts for the dehydroreduction of indolines, but the reliance on precious metals introduces substantial raw material costs and potential contamination issues that are unacceptable for high-purity pharmaceutical applications. These conventional pathways often involve multi-step sequences with poor atom economy, generating excessive waste and complicating the supply chain for critical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the patented technology introduces a streamlined catalytic cycle that directly transforms 2-indolones into indoles using a rare earth silamide catalyst and pinacolborane. This approach fundamentally shifts the paradigm by replacing dangerous reagents with stable, easy-to-handle boranes that are resistant to air and moisture, significantly enhancing operational safety. The reaction proceeds through a selective hydrogen transfer mechanism that preserves sensitive functional groups, allowing for the synthesis of complex substituted indoles without the need for protecting group strategies. As illustrated in the reaction scheme below, the transformation is direct and efficient, utilizing simple starting materials that are commercially abundant.
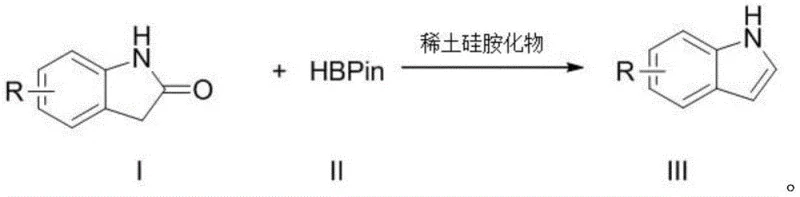
The versatility of this system is further evidenced by its compatibility with a wide range of substituents, including halogens and alkyl groups, ensuring that diverse chemical libraries can be accessed from a single robust platform. The elimination of transition metal residues and the use of non-toxic byproducts simplify the downstream purification process, resulting in higher overall throughput and reduced environmental impact.
Mechanistic Insights into Rare Earth Silamide-Catalyzed Hydrogen Transfer
The efficacy of this synthetic route is rooted in the sophisticated mechanistic pathway facilitated by the rare earth center, which acts as a dynamic hub for bond activation and formation. The proposed catalytic cycle initiates with the reaction between the trisilamido rare earth catalyst and pinacolborane to generate a reactive rare earth hydride intermediate, designated as species A in the mechanistic model. This metal-hydride species then undergoes insertion into the carbonyl group of the 2-indolone substrate, forming a key alkoxy-rare earth intermediate B. Subsequent interaction with another equivalent of pinacolborane regenerates the active hydride species A while producing a borate intermediate C, effectively closing the catalytic loop for the reduction step. The borate intermediate C then undergoes elimination to form an iminium cation intermediate D, which is subsequently reduced by pinacolborane to yield an imine intermediate E, releasing hydrogen and boron ester byproducts. Finally, the imine intermediate undergoes tautomerization to aromatize the five-membered ring, delivering the final indole product with high fidelity.
Understanding the electronic influence of the substituents on the benzene ring is crucial for optimizing reaction outcomes, as the nature of the R group—whether it be hydrogen, methyl, chloro, fluoro, bromo, or methoxy—can subtly influence the electrophilicity of the carbonyl carbon and the stability of the intermediates. The rare earth catalyst's ability to accommodate these electronic variations without loss of activity underscores its utility as a general-purpose tool for heterocycle synthesis. Furthermore, the mechanism avoids the formation of radical species that often lead to polymerization or side reactions in other reduction protocols, thereby ensuring a clean impurity profile that is essential for regulatory compliance in drug substance manufacturing.
How to Synthesize Indole Derivatives Efficiently
Implementing this advanced catalytic protocol requires strict adherence to anhydrous and oxygen-free conditions to maintain the integrity of the rare earth catalyst and prevent premature deactivation. The process begins by charging a reactor with the 2-indolone substrate and pinacolborane in a precise molar ratio, typically utilizing a slight excess of the borane reagent to drive the equilibrium toward completion. The choice of solvent is paramount, with non-polar hydrocarbons like toluene or n-hexane proving superior to coordinating ethers, a detail that process chemists must rigorously control to achieve the reported high yields. While the general procedure is straightforward, scaling this reaction requires careful thermal management to handle the exothermic nature of the hydride formation and ensure uniform mixing throughout the batch.
- Under an inert atmosphere (nitrogen or argon), charge a reactor with 2-indolone substrate, pinacolborane (2.4 equivalents), and a rare earth silamide catalyst such as Y[N(SiMe3)2]3 (5-10 mol%).
- Add anhydrous toluene, xylene, or n-hexane as the reaction solvent and heat the mixture to a temperature range of 80-120°C, preferably maintaining 120°C for optimal kinetics.
- Stir the reaction mixture for 24 to 36 hours to ensure complete conversion, followed by standard workup and purification procedures to isolate the high-purity indole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this technology offers compelling advantages by decoupling production costs from the volatile pricing of precious metals and hazardous reagents. The shift towards earth-abundant rare earth catalysts, which can be derived from inexpensive oxide precursors like Y2O3, fundamentally alters the cost structure of indole manufacturing, making it more resilient to market fluctuations. Additionally, the use of pinacolborane as a hydrogen source eliminates the logistical complexities and safety premiums associated with transporting and storing pyrophoric lithium reagents or high-pressure hydrogen gas. This simplification of the raw material portfolio allows supply chain managers to secure more reliable sourcing channels and reduce inventory holding costs related to specialized safety equipment.
- Cost Reduction in Manufacturing: The replacement of expensive platinum group metal catalysts with cost-effective rare earth silamides results in a drastic reduction in direct material costs per kilogram of product. Furthermore, the high selectivity of the reaction minimizes the formation of byproducts, which reduces the burden on purification units and lowers the consumption of solvents and energy during downstream processing. The ability to operate at atmospheric pressure without specialized high-pressure reactors also translates to lower capital expenditure requirements for manufacturing facilities.
- Enhanced Supply Chain Reliability: By utilizing reagents that are stable to air and moisture, such as pinacolborane, the risk of supply disruption due to hazardous material transport restrictions is significantly mitigated. The robustness of the catalyst system ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed runs or out-of-specification results. This reliability is critical for maintaining continuous supply to downstream pharmaceutical customers who demand strict adherence to delivery schedules.
- Scalability and Environmental Compliance: The process generates minimal waste compared to traditional stoichiometric reductions, aligning with increasingly stringent global environmental regulations and sustainability goals. The simplicity of the workup procedure facilitates easier scale-up from laboratory to commercial tonnage, allowing manufacturers to respond rapidly to surges in market demand without extensive process re-engineering. The absence of heavy metal residues simplifies waste disposal protocols and reduces the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rare earth catalyzed synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential adopters.
Q: What are the primary advantages of using rare earth silamides over traditional platinum catalysts for indole synthesis?
A: Rare earth silamides offer a cost-effective alternative to expensive platinum group metals while avoiding the safety hazards associated with highly reactive reagents like tert-butyllithium. The process utilizes stable, commercially available pinacolborane and operates under milder conditions with high atom economy.
Q: How does solvent choice impact the yield of this rare earth catalyzed reduction?
A: Solvent selection is critical; non-coordinating solvents like toluene, xylene, and n-hexane achieve yields exceeding 90%, whereas coordinating solvents like tetrahydrofuran significantly inhibit the catalyst, dropping yields below 60%.
Q: Is this method suitable for synthesizing indoles with electron-withdrawing or donating substituents?
A: Yes, the method demonstrates excellent functional group tolerance, successfully synthesizing derivatives with fluoro, chloro, bromo, methyl, and methoxy substituents at various positions on the benzene ring with high isolated yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this rare earth catalyzed pathway for the production of high-value indole intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet rigorous quality standards. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the absence of residual catalysts and impurities, guaranteeing that every batch is ready for immediate use in sensitive medicinal chemistry campaigns.
We invite global partners to collaborate with us to leverage this innovative technology for their specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, along with specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your time-to-market with our advanced manufacturing capabilities.
