Advanced Palladium-Catalyzed Synthesis of 2,3-Disubstituted Indoles for Commercial HCV Inhibitor Production
Introduction to Advanced Indole Synthesis Technology
The pharmaceutical industry's relentless pursuit of effective Hepatitis C Virus (HCV) treatments has driven significant innovation in the synthesis of complex heterocyclic intermediates. Patent CN101155801A discloses a groundbreaking process for the preparation of 2,3-disubstituted indole compounds, which serve as critical scaffolds in the development of potent HCV inhibitors. This technology addresses long-standing challenges in constructing the indole core with high regioselectivity and purity, utilizing a sophisticated palladium-catalyzed borylation strategy followed by Suzuki coupling. By shifting away from traditional organozinc or alkyne-based methodologies, this approach offers a more robust and versatile pathway for generating diverse libraries of biologically active molecules. For R&D directors and process chemists, understanding the nuances of this patent is essential for designing next-generation antiviral agents that meet stringent regulatory standards for impurity control and structural integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
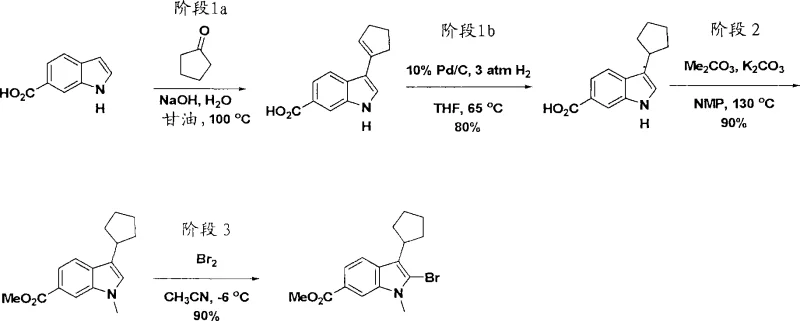
Historically, the synthesis of 2,3-disubstituted indoles has relied heavily on methods that introduce significant operational risks and purity challenges at scale. Method A, for instance, utilizes dipyridyl zinc intermediates in conjunction with palladium catalysts, but this approach is fraught with difficulties when the target molecule contains sensitive aryl bromide moieties. In such scenarios, the nucleophilic zinc reagent can engage in competitive side reactions with the desired product, leading to the formation of polymeric byproducts that are notoriously difficult to remove. Similarly, Method B employs palladium-catalyzed indolation of 2-halo-anilines with internal alkynes, yet this pathway suffers from poor chemoselectivity when the alkyne substituents contain vinyl or aryl halides. These competing oxidative insertion events drastically reduce yield and complicate downstream purification, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
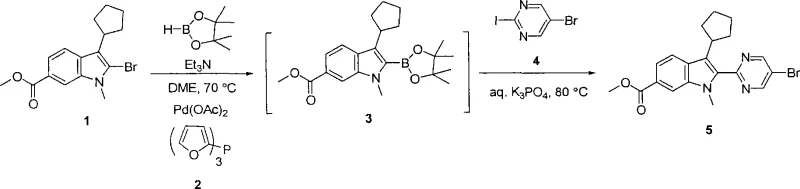
In stark contrast, the novel methodology outlined in the patent leverages a palladium-catalyzed borylation of 2-bromoindole derivatives to generate stable boronate ester intermediates, which are then subjected to Suzuki coupling. This two-step sequence, which can potentially be telescoped into a one-pot operation, circumvents the aggressive reactivity associated with organozinc species. The use of dialkoxyboranes, such as pinacolborane, under mild thermal conditions (70-80°C) ensures compatibility with a wide range of functional groups, including esters and heterocycles. Furthermore, the ability to utilize simple aqueous inorganic bases like potassium phosphate or carbonate simplifies the workup procedure and reduces the environmental footprint of the manufacturing process. This strategic shift not only enhances the overall yield but also provides a cleaner reaction profile, which is paramount for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pd-Catalyzed Borylation and Coupling
The core of this technological advancement lies in the precise orchestration of the palladium catalytic cycle, specifically utilizing ligands such as tris(2-furyl)phosphine (TFP). This ligand plays a dual role by facilitating both the initial borylation of the bromoindole and the subsequent cross-coupling with aryl or heteroaryl halides. The mechanism begins with the oxidative addition of the palladium catalyst to the carbon-bromine bond of the indole substrate, followed by transmetallation with the dialkoxyborane species. The presence of TFP stabilizes the palladium center, allowing the reaction to proceed efficiently at moderate temperatures without the need for exotic or highly toxic reagents. This stability is crucial for maintaining the integrity of the indole ring system, preventing degradation pathways that often plague high-temperature syntheses. Moreover, the ligand's electronic properties promote rapid reductive elimination, ensuring high turnover numbers and minimizing the residence time of reactive intermediates that could lead to impurity formation.
Impurity control is another critical aspect where this mechanism excels, particularly regarding the suppression of homocoupling dimers. In traditional Suzuki reactions, the formation of symmetrical biaryl byproducts is a common issue that erodes yield and complicates purification. However, the specific reaction conditions described, including the use of aqueous base systems and optimized catalyst loading (e.g., 3 mol% Pd(OAc)2), effectively mitigate these side reactions. Additionally, the patent describes an alternative pathway involving bromine-magnesium exchange using trialkylmagnesiate reagents at low temperatures (-20°C to 0°C). This alternative offers a complementary route for substrates that might be sensitive to the thermal conditions of the borylation step, providing process chemists with flexible options to tailor the synthesis based on the specific electronic nature of the substituents. Such mechanistic versatility ensures that the process remains robust across a broad scope of 2,3-disubstituted indole derivatives.
How to Synthesize 2,3-Disubstituted Indole Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize efficiency and product quality. The process generally begins with the conversion of a 2-bromoindole precursor into a boronate ester using a palladium catalyst and a boron source in a solvent like DME or THF. Following the formation of this key intermediate, the reaction mixture can often be directly treated with the coupling partner and an aqueous base to complete the transformation without isolating the unstable boronate species. This telescoping capability is a significant operational advantage, reducing the number of unit operations and solvent exchanges required. Detailed standardized synthetic steps for executing this transformation, including specific reagent quantities and temperature profiles, are provided in the guide below to ensure reproducibility and safety during scale-up.
- React 2-bromoindole compound with dialkoxyborane (e.g., pinacolborane) in the presence of a palladium catalyst (Pd(OAc)2), a ligand (e.g., tris(2-furyl)phosphine), and a base (e.g., Et3N) in DME or THF at 70-80°C to form the boronate ester intermediate.
- Without isolation, react the resulting boronate ester mixture with an aryl or heteroaryl halide (R2-Hal) in the presence of an aqueous inorganic base (e.g., K3PO4 or K2CO3) at 80°C to effect Suzuki coupling.
- Quench the reaction, extract with organic solvent (EtOAc), dry over Na2SO4, and purify the crude solid via slurry in cold acetone or silica gel chromatography to obtain the high-purity 2,3-disubstituted indole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial benefits that directly address the pain points of procurement managers and supply chain leaders in the fine chemical sector. Traditional methods for synthesizing complex indoles often involve multiple isolation steps, hazardous reagents, and extensive purification protocols that drive up costs and extend lead times. By adopting this streamlined palladium-catalyzed approach, manufacturers can significantly reduce the complexity of the production workflow. The ability to perform the reaction in a one-pot fashion eliminates the need for intermediate drying and handling, which translates to lower labor costs and reduced solvent consumption. Furthermore, the use of commercially available starting materials and catalysts ensures a stable supply chain, mitigating the risk of raw material shortages that can disrupt production schedules for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive and sensitive organozinc reagents, coupled with the potential for a one-pot procedure, leads to substantial cost savings in raw materials and processing. By avoiding the formation of polymeric byproducts common in older methods, the process reduces the burden on purification resources, such as chromatography media and crystallization solvents. This efficiency gain allows for a more competitive pricing structure for the final API intermediate, enhancing the overall margin profile for downstream drug manufacturers without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on robust, commodity-grade reagents like pinacolborane and potassium phosphate ensures that the supply chain is less vulnerable to fluctuations in the availability of specialty chemicals. The process tolerates a wide range of functional groups, meaning that a single manufacturing platform can be adapted to produce various analogues within the 2,3-disubstituted indole family. This flexibility allows suppliers to respond rapidly to changing demand signals from pharmaceutical clients, ensuring consistent delivery of high-purity materials even during periods of market volatility or increased production requirements.
- Scalability and Environmental Compliance: The reaction conditions operate at moderate temperatures and utilize solvents that are manageable within standard industrial infrastructure, facilitating seamless scale-up from pilot plants to commercial tonnage. The reduced generation of hazardous waste, particularly through the avoidance of stoichiometric metal waste associated with zinc or magnesium methods, aligns with increasingly strict environmental regulations. This green chemistry profile not only simplifies waste disposal logistics but also enhances the sustainability credentials of the supply chain, a factor that is becoming progressively important for global pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a reliable foundation for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this process into their existing manufacturing portfolios. The answers cover aspects ranging from reaction optimization to impurity management, ensuring a comprehensive overview of the technology's capabilities.
Q: What are the primary advantages of this Pd-catalyzed borylation method over traditional zinc-based coupling?
A: Unlike Method A which utilizes dipyridyl zinc intermediates prone to polymerization with aryl bromides, this Pd-catalyzed borylation route offers superior functional group tolerance and minimizes side reactions, leading to higher purity profiles essential for pharmaceutical applications.
Q: Can this synthesis method be scaled for commercial production of HCV inhibitor intermediates?
A: Yes, the process utilizes robust conditions (70-80°C) and common solvents like DME and THF. The potential for a one-pot procedure significantly reduces processing time and solvent consumption, making it highly suitable for commercial scale-up from kilograms to metric tons.
Q: How does the choice of ligand impact the reaction efficiency and impurity profile?
A: The patent highlights tris(2-furyl)phosphine (TFP) as a preferred ligand. It facilitates both the borylation and the subsequent Suzuki coupling under mild conditions, allowing for the use of simple aqueous inorganic bases and effectively suppressing the formation of homocoupling dimer impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Disubstituted Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving antiviral medications. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity 2,3-disubstituted indoles that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch supplied adheres to the highest international standards, providing our partners with the confidence they need to advance their clinical programs.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your time to market. Let us be your trusted partner in bringing innovative HCV therapies to patients worldwide.
