Advanced Synthesis of Tryptamine Precursors via Isatin Condensation for Commercial Scale-up
The pharmaceutical industry constantly seeks more efficient and safer pathways for synthesizing complex heterocyclic scaffolds, particularly those serving as core structures for bioactive molecules. Patent CN1729174B introduces a groundbreaking methodology for the preparation of tryptamine, horsfiline, and coerulescine derivatives, which are critical intermediates in the development of various therapeutic agents. This invention specifically addresses the limitations of prior art by utilizing a novel condensation reaction between isatin derivatives and malonic acid, followed by unique activation steps. Unlike traditional routes that rely heavily on unstable hydrazine intermediates, this approach leverages the stability of oxindole scaffolds to achieve superior purity and yield. The technical breakthrough lies in the ability to generate versatile esters and amides directly from the condensation product, enabling a streamlined flow towards diverse tryptamine analogs. For R&D teams focused on API development, this patent offers a compelling alternative that enhances process safety and chemical efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tryptamine derivatives has been dominated by routes originating from substituted aniline compounds, which necessitate conversion into phenylhydrazines before undergoing cyclization. As illustrated in the conventional Reaction Scheme 2, this process typically involves the reduction of diazonium salts using excessive amounts of tin(II) chloride, generating significant heavy metal waste and posing environmental hazards. Furthermore, the subsequent Fischer indolization step often suffers from low yields due to the instability of the indole product under the strongly acidic reaction conditions required. Side reactions, such as the formation of carbazoles and methylation of the indole nitrogen, frequently complicate the purification process and reduce the overall mass balance. These inherent drawbacks make the traditional aniline-to-hydrazine pathway less attractive for modern, green chemistry-focused manufacturing environments where waste reduction and atom economy are paramount concerns for supply chain sustainability.
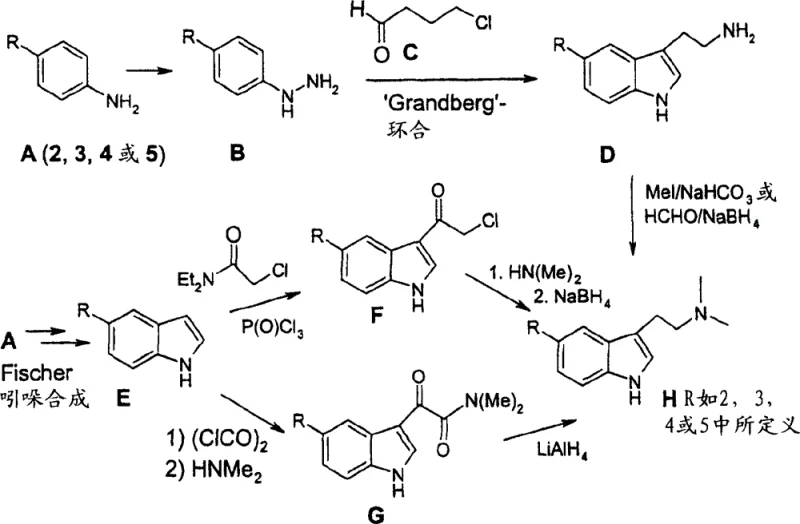
The Novel Approach
In stark contrast, the novel approach detailed in CN1729174B circumvents the hazardous hydrazine stage entirely by employing a direct condensation of isatin with malonic acid in a pyridine medium. This method generates a stable pyridinium salt intermediate which can be further derivatized in a one-pot fashion to produce valuable esters or amides. The elimination of strong mineral acids and heavy metal reducing agents significantly simplifies the workup procedure and improves the environmental profile of the synthesis. By stabilizing the intermediate as an oxindole derivative, the process allows for precise control over functional group introduction at the 3-position of the indole ring. This strategic shift not only enhances the chemical yield but also provides a robust platform for introducing diverse substituents, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates where consistency and reliability are critical.
Mechanistic Insights into Isatin-Malonic Acid Condensation
The core mechanistic advantage of this invention resides in the nucleophilic activation of the condensation product derived from isatin and malonic acid. Initially, the reaction in pyridine yields a pyridinium salt, which traditionally presents challenges in further functionalization without isolation. However, the patent reveals that treating this intermediate with a suitable nitrogenous base, such as triethylamine, renders it sufficiently nucleophilic to react directly with activated carbonates or chloroformamides. This transformation is depicted in Reaction Scheme 7, where the in situ generation of the nucleophile allows for the seamless formation of esters or N,N-disubstituted alkylamide derivatives. The ability to merge the condensation and derivatization steps into a single operational sequence minimizes material handling and reduces the risk of intermediate degradation. This mechanistic elegance ensures that the sensitive 3-hydroxy-2-oxindole core is preserved while efficiently installing the necessary side chains for downstream reduction to tryptamines.
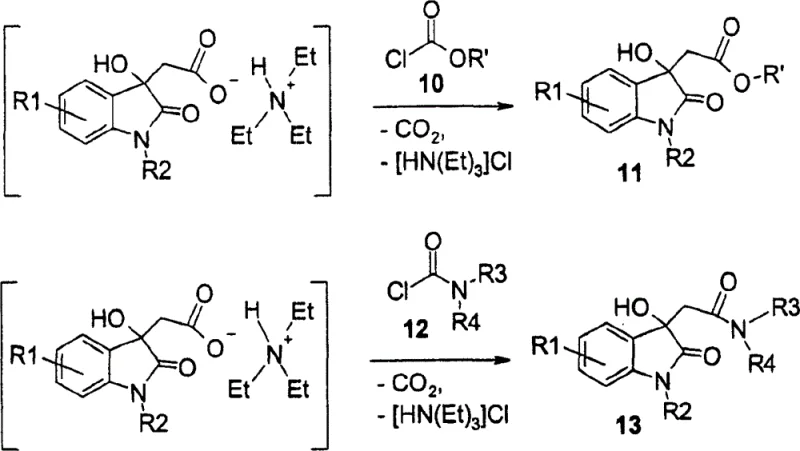
Furthermore, the impurity control mechanism is inherently superior due to the crystalline nature of the intermediates formed during the process. The precipitation of the pyridinium salt and the subsequent amide products allows for effective purification through simple filtration and slurry washing, removing soluble by-products before they can interfere with subsequent reduction steps. The patent data indicates that the resulting amides, such as those prepared in Example 1, exhibit high purity suitable for direct use in the next synthetic stage without extensive chromatographic purification. This characteristic is vital for maintaining a clean impurity profile throughout the synthesis, ensuring that the final active pharmaceutical ingredient meets stringent regulatory specifications. The robustness of this mechanism against side reactions like decarboxylation or polymerization underlines its suitability for large-scale manufacturing where batch-to-batch consistency is non-negotiable.
How to Synthesize Oxindole Amide Intermediates Efficiently
The practical implementation of this synthesis route involves a carefully controlled sequence of addition and temperature management to maximize yield and safety. The process begins with the suspension of isatin and malonic acid in pyridine, followed by heating to initiate the condensation and release of carbon dioxide. Once the intermediate salt is formed, the addition of a tertiary amine base solubilizes the mixture, allowing for the controlled introduction of the activating agent, such as dimethylcarbamoyl chloride. Detailed standardized synthesis steps for this specific transformation are provided in the guide below, outlining the precise molar ratios and thermal profiles required to replicate the high yields reported in the patent examples. Adhering to these parameters ensures the successful formation of the target 3-substituted oxindole scaffolds.
- Condense isatin derivatives with malonic acid in pyridine to form a pyridinium salt intermediate.
- Activate the intermediate with a tertiary amine base and react with an activated carbonate or amide chloride.
- Reduce the resulting oxindole derivative using borane complexes or metal hydrides to obtain the final indole or tryptamine structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers substantial advantages by eliminating the reliance on hazardous and regulated reagents like phenylhydrazines and tin chloride. The removal of these materials simplifies the sourcing strategy and reduces the regulatory burden associated with handling toxic substances, thereby enhancing supply chain reliability. Moreover, the use of readily available starting materials such as isatin and malonic acid ensures a stable supply base, mitigating the risk of raw material shortages that can disrupt production schedules. The process is designed to be scalable, with exothermic reactions that are manageable under standard industrial cooling conditions, facilitating a smooth transition from pilot plant to full commercial production. This scalability ensures that manufacturers can meet increasing demand for tryptamine-based APIs without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal catalysts and the reduction in waste treatment costs associated with tin residues lead to significant operational savings. By streamlining the synthesis into fewer steps and avoiding complex purification protocols, the overall cost of goods sold is optimized, making the final intermediates more competitive in the global market. The ability to perform derivatization in a one-pot manner further reduces solvent consumption and energy usage, contributing to a leaner and more cost-effective manufacturing process.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals like pyridine and malonic acid reduces dependency on specialized suppliers, ensuring a more resilient supply chain. The robust nature of the intermediates allows for longer storage stability if necessary, providing flexibility in inventory management. This reliability is crucial for maintaining continuous production flows in the face of global logistical challenges, ensuring that downstream API synthesis is never halted due to intermediate shortages.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional methods, aligning with increasingly strict environmental regulations. The crystallization-driven purification minimizes the need for resource-intensive chromatography, making the process inherently greener and easier to scale. This environmental compliance not only reduces disposal costs but also enhances the corporate sustainability profile, which is becoming a key factor in vendor selection for major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis method. These answers are derived directly from the experimental data and technical descriptions provided in the patent documentation, offering clarity on process capabilities and limitations. Understanding these aspects is essential for technical teams evaluating the feasibility of adopting this route for their specific project needs.
Q: How does this method improve upon traditional Fischer indolization?
A: This method avoids the use of hazardous phenylhydrazines and strong acidic conditions required in Fischer indolization, leading to better stability and higher purity of the final indole products.
Q: What are the key starting materials for this synthesis?
A: The primary starting materials are substituted isatins and malonic acid, which are reacted in the presence of pyridine and subsequently activated with tertiary amines and acyl chlorides.
Q: Can this process be scaled for commercial production?
A: Yes, the patent describes robust exothermic management and crystallization steps that facilitate scaling from laboratory to commercial production volumes without significant loss of yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tryptamine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative chemistry described in CN1729174B can be effectively translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of tryptamine derivatives meets the highest international standards. Our commitment to quality assurance means that clients can rely on us for consistent supply of high-purity intermediates that facilitate smoother regulatory filings and faster time-to-market for their drug candidates.
We invite you to collaborate with us to explore how this advanced synthesis technology can optimize your supply chain and reduce manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can support your long-term strategic goals in pharmaceutical development.
