Advanced Manufacturing of 3-(4-Chlorobutyl)indole-5-Carbonitrile for Global Pharma Supply Chains
Advanced Manufacturing of 3-(4-Chlorobutyl)indole-5-Carbonitrile for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust synthetic routes for key intermediates that balance high purity with economic viability. A prime example is found in the synthesis of Vilazodone Hydrochloride, an antidepressant approved by the FDA, where the quality of the starting materials dictates the success of the final drug substance. As detailed in patent CN103058912A, a novel preparation method for 3-(4-chlorobutyl)indole-5-carbonitrile has been developed that fundamentally alters the cost and efficiency landscape for this critical building block. This technical breakthrough addresses the longstanding inefficiencies of previous synthetic pathways by replacing hazardous, low-yielding reduction steps with a safer, high-yielding alternative. For R&D directors and procurement managers alike, understanding this shift is crucial for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent global standards. The transition from complex aluminum hydride reductions to a streamlined sodium borohydride protocol represents a significant leap forward in process chemistry, ensuring that supply chains remain resilient against raw material volatility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this vital indole derivative relied on a pathway that, while chemically feasible, was fraught with operational and economic disadvantages. The conventional route, often cited in earlier literature such as the Journal of Medicinal Chemistry, utilized 5-cyanoindole and 4-chlorobutyryl chloride as starting materials but employed sodium bis(2-methoxyethoxy)aluminum hydride (Red-Al) for the critical reduction step. This reagent is not only significantly more expensive than standard reducing agents but also poses severe safety risks due to its pyrophoric nature and sensitivity to moisture. Furthermore, the legacy process suffered from abysmal efficiency, with total yields reported as low as 19%, necessitating extensive purification via column chromatography to achieve acceptable purity levels. This reliance on chromatographic purification is a major bottleneck in industrial chemistry, consuming vast quantities of silica gel and solvents while drastically increasing processing time and waste generation. Consequently, the traditional method was deemed difficult to realize for suitability for industrialized production, creating a fragile supply link for manufacturers of Vilazodone.
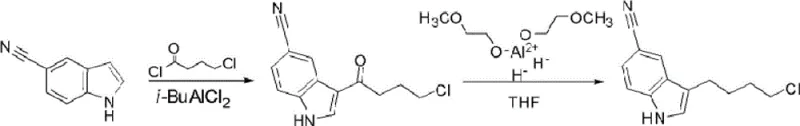
The Novel Approach
In stark contrast, the innovative methodology disclosed in the patent introduces a paradigm shift by utilizing sodium borohydride in combination with trifluoroacetic acid for the reduction phase. This modification transforms the reaction profile, boosting the total yield dramatically to a range of 70-80% while achieving product purity of 98-99.5% without the need for column chromatography. The new route maintains the initial Friedel-Crafts acylation using Lewis acids like aluminum chloride but optimizes the subsequent transformation into the alkyl chain. By operating at controlled low temperatures between -5°C and 0°C, the process ensures high selectivity and minimizes side reactions. The use of common, inexpensive reagents such as sodium borohydride and trifluoroacetic acid drastically lowers the raw material costs compared to the specialized Red-Al reagent. This approach not only simplifies the post-reaction workup but also enhances the overall safety profile of the manufacturing plant, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into Friedel-Crafts Acylation and Borohydride Reduction
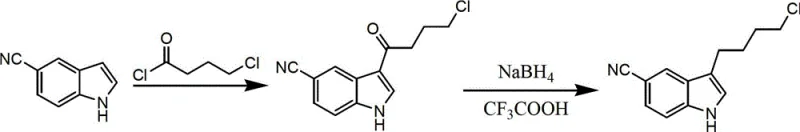
The core of this synthetic strategy lies in the precise orchestration of electrophilic aromatic substitution followed by a specialized reduction mechanism. In the first stage, 5-cyanoindole undergoes a Friedel-Crafts acylation with 4-chlorobutyryl chloride. The presence of a Lewis acid catalyst, typically aluminum chloride used in a molar ratio of 1 to 2 equivalents relative to the indole, activates the acid chloride to form a highly reactive acylium ion. This electrophile attacks the electron-rich C3 position of the indole ring, which is the most nucleophilic site, forming the ketone intermediate 3-(4-chlorobutyryl)indole-5-carbonitrile. The choice of solvent, such as dichloromethane or nitromethane, plays a critical role in stabilizing the charged intermediates and ensuring the reaction proceeds smoothly at the required cryogenic temperatures of -5°C to 0°C. Strict temperature control is essential here to prevent polyacylation or degradation of the sensitive cyano group.
The subsequent reduction step is where the true chemical ingenuity of this patent resides. Instead of using powerful, non-selective hydride donors, the process employs a system of sodium borohydride and trifluoroacetic acid. In this acidic medium, sodium borohydride generates reactive borane species in situ, which are potent enough to reduce the benzylic ketone to the corresponding methylene group but mild enough to leave the nitrile functionality intact. This chemoselectivity is paramount; reducing the nitrile group would lead to unwanted amine byproducts that are difficult to separate. The reaction mixture is maintained below 0°C to control the exotherm and ensure that the reduction stops at the desired alkyl stage. This mechanistic precision eliminates the formation of complex impurity profiles, thereby allowing the final product to be isolated with high purity simply through standard aqueous workup and crystallization, bypassing the need for chromatographic separation entirely.
How to Synthesize 3-(4-Chlorobutyl)indole-5-Carbonitrile Efficiently
Implementing this synthesis requires careful attention to thermal management and reagent addition rates to maximize the benefits of the new protocol. The process begins with the dissolution of 5-cyanoindole in a suitable organic solvent, followed by the slow addition of the acylating agent under Lewis acid catalysis. Once the acylated intermediate is formed and isolated, it is subjected to the reduction conditions using the sodium borohydride/trifluoroacetic acid system. The detailed standardized synthesis steps, including specific molar ratios and quenching procedures, are outlined below to guide process engineers in replicating these high-yield results.
- Dissolve 5-cyanoindole in a solvent such as dichloromethane and react with 4-chlorobutyryl chloride at -5 to 0°C using aluminum chloride as a Lewis acid catalyst to form the acylated intermediate.
- Dissolve the resulting 3-(4-chlorobutyryl)indole-5-carbonitrile in a solvent mixture and cool to -5°C.
- Add sodium borohydride in portions followed by trifluoroacetic acid, maintaining the temperature below 0°C to effect reduction to the final alkyl product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic benefits that extend far beyond simple chemistry. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly translates to enhanced supply chain reliability and reduced operational risk. By eliminating the need for column chromatography, the process removes a major variable that often causes batch-to-batch variability and delays in production schedules. Furthermore, the substitution of expensive, hazardous reagents with commodity chemicals creates a more robust cost structure that is less susceptible to market fluctuations. This stability is essential for long-term supply agreements in the competitive pharmaceutical sector.
- Cost Reduction in Manufacturing: The economic impact of switching from Red-Al to sodium borohydride cannot be overstated. Red-Al is a specialty reagent with a high price point and significant handling costs due to safety requirements. In contrast, sodium borohydride is a bulk commodity chemical available globally at a fraction of the cost. Additionally, the removal of the column chromatography step saves substantial amounts of silica gel and organic solvents, which are major cost drivers in fine chemical production. The increase in yield from roughly 19% to over 75% means that less raw material is wasted per kilogram of final product, effectively lowering the cost of goods sold significantly without compromising quality.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on niche reagents that may have limited suppliers or long lead times. The new method relies on 4-chlorobutyryl chloride, aluminum chloride, and sodium borohydride, all of which are produced on a massive industrial scale with multiple sources worldwide. This diversification of the supply base reduces the risk of shortages. Moreover, the simplified purification process reduces the turnaround time for each batch, allowing manufacturers to respond more quickly to demand spikes. The ability to produce high-purity material without complex purification equipment means that production capacity can be scaled up more easily to meet growing market needs for Vilazodone intermediates.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the new route is vastly superior. Red-Al is moisture-sensitive and can ignite upon contact with air, requiring specialized inert atmosphere equipment and rigorous safety protocols. Sodium borohydride, while still requiring care, is much more manageable in a standard industrial setting. The reduction in solvent usage associated with skipping chromatography also lowers the volume of hazardous waste generated, simplifying waste treatment and disposal compliance. This greener profile aligns with modern ESG (Environmental, Social, and Governance) goals, making the supply chain more sustainable and attractive to environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this intermediate. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, ensuring that stakeholders have accurate information for decision-making. Understanding these nuances helps in evaluating the feasibility of integrating this material into existing production lines.
Q: Why is the new reduction method superior to the traditional Red-Al route?
A: The traditional method uses Red-Al (sodium bis(2-methoxyethoxy)aluminum hydride), which is expensive, hazardous, and yields only 19% product requiring difficult column chromatography. The new method uses cheap Sodium Borohydride/TFA, achieving 70-80% yield with 99% purity without chromatography.
Q: What are the critical reaction conditions for the acylation step?
A: The Friedel-Crafts acylation must be conducted at low temperatures between -5°C and 0°C using a Lewis acid like Aluminum Chloride (1-2 equivalents) to ensure regioselectivity and prevent polymerization or degradation of the indole ring.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes common industrial solvents like dichloromethane and stable reagents like Sodium Borohydride. The elimination of column chromatography and the high yield make it highly scalable for multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(4-Chlorobutyl)indole-5-Carbonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to more efficient synthetic routes is critical for maintaining competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the methodology described in CN103058912A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering this key intermediate with stringent purity specifications, leveraging our rigorous QC labs to ensure every batch meets the 99% purity benchmark required for downstream API synthesis. Our facility is equipped to handle the specific thermal and safety requirements of the Friedel-Crafts and reduction steps, guaranteeing a consistent and high-quality supply.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic advantages of switching to our supply source. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain for Vilazodone intermediates is built on a foundation of technical excellence and commercial reliability.
