Optimizing Vilazodone Intermediate Production via Novel Zinc-Chloride Catalyzed Alkylation
Optimizing Vilazodone Intermediate Production via Novel Zinc-Chloride Catalyzed Alkylation
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value antidepressants, and the recent disclosure in patent CN111087340B represents a significant technological leap in the manufacturing of Vilazodone intermediates. This patent details a streamlined preparation method for 3-(4-chlorobutyl)indole-5-carbonitrile, a critical building block for the dual-action antidepressant Vilazodone. By shifting from traditional multi-step acylation-reduction sequences to a direct Friedel-Crafts alkylation, the inventors have addressed longstanding challenges regarding reagent stability, operational safety, and overall process economics. For R&D directors and procurement specialists, this innovation offers a compelling alternative to legacy routes that rely on hazardous and expensive Lewis acids. The following analysis dissects the technical merits of this one-step transformation, highlighting its potential to redefine supply chain reliability for this high-purity pharmaceutical intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes for this key intermediate have historically been plagued by inefficiencies and safety hazards that hinder scalable manufacturing. As documented in literature such as the Journal of Medicinal Chemistry (2004), the conventional pathway typically involves a two-step sequence: first, a Friedel-Crafts acylation using isobutyl aluminum dichloride to form a ketone intermediate, followed by a selective reduction step. This approach is fundamentally flawed for industrial application due to the extreme sensitivity of the required reagents. Isobutyl aluminum dichloride is highly unstable, pyrophoric, and necessitates specialized pipeline conveying systems and rigorous exclusion of moisture, driving up capital expenditure for reactor infrastructure. Furthermore, the subsequent reduction step often employs Red-Al (sodium bis(2-methoxyethoxy)aluminum hydride), a reagent known for its operational difficulty and low atom economy.
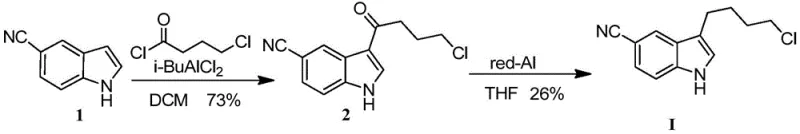
The cumulative effect of these complex steps results in suboptimal yields, with historical data indicating reduction efficiencies as low as 26%. Such poor material throughput not only inflates the cost of goods sold (COGS) but also generates substantial chemical waste, complicating environmental compliance. Additionally, the requirement for column chromatography purification in some legacy methods is entirely incompatible with multi-tonne commercial production, creating a bottleneck that limits supply continuity. These factors collectively render the traditional acylation-reduction strategy economically unsustainable for modern generic drug manufacturers seeking cost-effective solutions.
The Novel Approach
In stark contrast to the cumbersome legacy protocols, the method disclosed in patent CN111087340B introduces a elegant one-step Friedel-Crafts alkylation that directly couples 5-cyanoindole with 1-bromo-4-chlorobutane. This transformative approach utilizes anhydrous zinc chloride as the catalyst, a reagent that is not only commercially ubiquitous and inexpensive but also significantly safer to handle than organo-aluminum compounds. The reaction proceeds smoothly under mild thermal conditions, typically ranging from -10°C to 30°C, eliminating the need for cryogenic cooling or high-temperature reflux that characterizes more aggressive alkylation protocols. By bypassing the ketone intermediate entirely, the process removes the necessity for a separate reduction step, thereby collapsing the synthesis timeline and reducing the number of unit operations required.
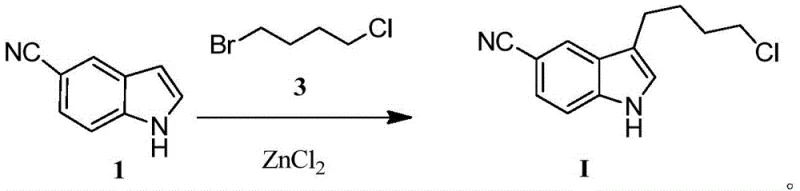
The operational simplicity of this novel route is further evidenced by the straightforward post-reaction workup, which involves standard quenching, extraction, and recrystallization rather than complex chromatographic separation. Experimental data from the patent demonstrates that this direct alkylation achieves impressive yields, with Example 1 reporting an 88% isolated yield and HPLC purity exceeding 99.6%. This dramatic improvement in efficiency translates directly to lower raw material consumption and reduced waste disposal costs. For supply chain managers, the shift to stable, off-the-shelf reagents like zinc chloride and bromo-chlorobutane mitigates the risk of production delays caused by the scarcity or hazardous transport requirements of specialized Lewis acids.
Mechanistic Insights into ZnCl2-Catalyzed Friedel-Crafts Alkylation
The success of this novel synthesis lies in the precise activation of the electrophile by the Lewis acid catalyst within the indole heterocyclic system. In this mechanism, anhydrous zinc chloride acts as a potent Lewis acid by coordinating with the bromine atom of 1-bromo-4-chlorobutane. This coordination polarizes the carbon-bromine bond, facilitating the heterolytic cleavage that generates a reactive carbocation-like species or a tight ion pair at the terminal carbon of the butyl chain. The indole nucleus, specifically the C3 position, is inherently electron-rich due to the resonance donation from the nitrogen lone pair, making it the preferred nucleophilic site for electrophilic aromatic substitution. The zinc chloride catalyst effectively lowers the activation energy for this attack, ensuring that the alkylation occurs regioselectively at the C3 position rather than the less favorable N1 or C2 positions.
Regiocontrol is paramount in this synthesis to prevent the formation of N-alkylated byproducts, which would be difficult to separate and would degrade the quality of the final API. The choice of solvent, dichloromethane, plays a crucial role in stabilizing the transition state and solvating the ionic intermediates without interfering with the catalyst. Furthermore, the controlled addition rate of the alkyl halide at low temperatures (-10°C to -5°C) is critical to suppress poly-alkylation side reactions. By maintaining a low concentration of the activated electrophile, the reaction kinetics favor the mono-alkylation of the indole ring. This kinetic control, combined with the thermodynamic stability of the C3-alkylated product, ensures a clean reaction profile that minimizes the generation of structurally related impurities.
How to Synthesize 3-(4-chlorobutyl)indole-5-carbonitrile Efficiently
Implementing this optimized synthetic route requires strict adherence to the specified molar ratios and thermal profiles to maximize yield and purity. The process begins with the dissolution of the indole starting material in a dry aprotic solvent, followed by the careful addition of the catalyst under inert atmosphere conditions to prevent hydrolysis. The subsequent addition of the alkylating agent must be managed precisely to control the exotherm and maintain the reaction within the optimal temperature window defined in the patent examples. Following the reaction period, the quenching step is designed to neutralize the Lewis acid and facilitate the phase separation of the organic product from inorganic salts.
- Dissolve compound 1 (5-cyanoindole) in dichloromethane and add anhydrous zinc chloride while maintaining the internal temperature between 0°C and 5°C.
- Dropwise add 1-bromo-4-chlorobutane (compound 3) keeping the temperature below -5°C, then warm to 0°C and react for 2 hours.
- Quench the reaction with ice water and sodium hydroxide, separate the organic phase, wash with brine, concentrate, and recrystallize from isopropyl acetate to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this zinc chloride-catalyzed route offers profound strategic advantages beyond mere technical elegance. The primary benefit is the drastic simplification of the raw material portfolio. By eliminating the need for isobutyl aluminum dichloride and Red-Al, companies can source reagents from a broader base of general chemical suppliers, reducing dependency on niche vendors and mitigating supply disruption risks. The stability of zinc chloride allows for bulk purchasing and long-term storage without the degradation issues associated with moisture-sensitive organometallics, enabling better inventory management and cash flow optimization. This shift fundamentally alters the cost structure of the intermediate, moving it from a high-cost specialty chemical category to a more commoditized production model.
- Cost Reduction in Manufacturing: The elimination of the reduction step and the replacement of expensive, hazardous Lewis acids with cheap zinc chloride result in substantial cost savings. The removal of column chromatography purification further reduces operational expenditures by saving on silica gel, solvents, and labor hours. Additionally, the higher yield (88% vs 26%) means that less starting material is required per kilogram of product, directly lowering the variable cost of production and improving the overall margin profile for the final API.
- Enhanced Supply Chain Reliability: The use of common industrial reagents like 1-bromo-4-chlorobutane and dichloromethane ensures that production is not held hostage by the lead times of exotic catalysts. The robustness of the reaction conditions, which tolerate a reasonable range of temperatures and stoichiometry as shown in the patent examples, makes the process resilient to minor variations in plant operations. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, preventing stockouts that could delay drug product launches.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route is vastly superior. Avoiding pyrophoric reagents reduces the risk of fire and explosion in the plant, lowering insurance premiums and safety training costs. The simplified workup generates less hazardous waste, and the high purity of the crude product reduces the solvent load associated with extensive purification. This aligns with green chemistry principles, making it easier to obtain environmental permits for capacity expansion and ensuring long-term regulatory compliance in strict jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in patent CN111087340B, offering clarity on why this route is becoming the preferred standard for Vilazodone intermediate production. Understanding these nuances is essential for technical teams evaluating technology transfer packages.
Q: Why is the zinc chloride method superior to previous isobutyl aluminum dichloride routes?
A: The novel method utilizes anhydrous zinc chloride, which is significantly cheaper, more stable, and easier to handle than the moisture-sensitive and pyrophoric isobutyl aluminum dichloride used in prior art. Furthermore, it eliminates the need for a separate reduction step, streamlining the process from two steps to one.
Q: How does this process improve yield compared to the Red-Al reduction method?
A: Previous methods involving Red-Al reduction suffered from low yields of approximately 26% due to the difficulty in controlling selectivity and handling hazardous reagents. The direct alkylation method described in patent CN111087340B achieves yields exceeding 85%, drastically improving material efficiency.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process operates under mild temperatures (-10°C to 30°C) and uses common solvents like dichloromethane. The post-treatment involves standard extraction and recrystallization, avoiding complex chromatography, which makes it highly scalable and compliant with industrial safety standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(4-chlorobutyl)indole-5-carbonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team has extensively analyzed the Friedel-Crafts alkylation pathway described in CN111087340B and has successfully adapted it for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity specifications (99.5%+) achieved in the lab are consistently met in our pilot and commercial plants. Our rigorous QC labs employ state-of-the-art HPLC and GC-MS instrumentation to monitor every batch, guaranteeing that the impurity profile remains well within the limits required for GMP API synthesis.
We invite global pharmaceutical partners to leverage our optimized manufacturing platform for their Vilazodone supply needs. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this superior alkylation route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data from our recent batches and to discuss route feasibility assessments tailored to your project timelines. Let us collaborate to secure a stable, cost-effective supply of this critical antidepressant intermediate.
