Scalable Two-Step Synthesis of Imrecoxib: A Cost-Effective Route for API Manufacturing
Scalable Two-Step Synthesis of Imrecoxib: A Cost-Effective Route for API Manufacturing
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for the production of active pharmaceutical ingredients (APIs) and their critical intermediates. Patent CN108997188B discloses a groundbreaking synthetic method for Erexib (Imrecoxib), a potent COX-2 selective inhibitor used for pain relief in osteoarthritis. This novel approach fundamentally restructures the manufacturing landscape by replacing hazardous, multi-step sequences with a streamlined two-step process. By focusing on the direct oxidation of a hydroxy-amine precursor followed by a condensation cyclization, this technology addresses the critical pain points of impurity control and environmental compliance. For R&D directors and supply chain managers, this represents a significant opportunity to optimize the production of high-purity pharmaceutical intermediates while drastically reducing the ecological footprint of the synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Imrecoxib has been plagued by complex reaction sequences that rely on hazardous reagents and expensive coupling agents. As illustrated in prior art routes, traditional methods often commence with 4-methylsulfonyl styrene oxide, necessitating nucleophilic ring-opening, amidation, and subsequent oxidation steps. A major bottleneck in these conventional pathways is the reliance on Jones reagent or pyridinium chlorochromate for oxidation. These chromium-based oxidants not only present severe toxicity risks but also leave behind metal residues that are notoriously difficult to purge from the final product, compromising the purity required for bulk drug substances. Furthermore, alternative routes have employed costly condensing agents such as carbonyldiimidazole (CDI) to drive amidation, which inflates the raw material costs and complicates the downstream purification processes due to the formation of urea byproducts.
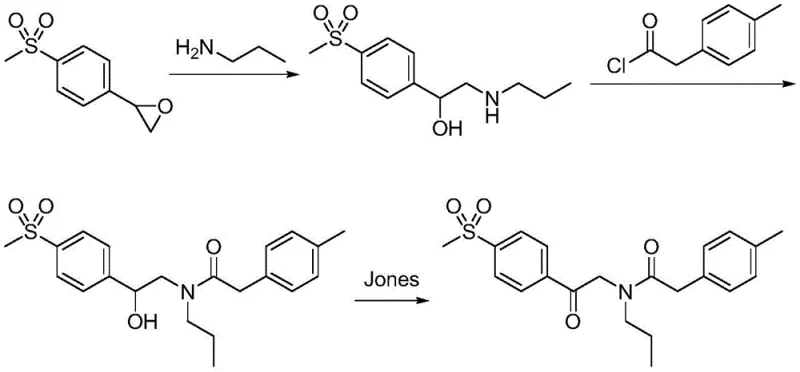
The Novel Approach
In stark contrast to the cumbersome legacy methods, the technology disclosed in CN108997188B introduces a highly efficient strategy that bypasses the need for toxic heavy metals and expensive activators. The new route initiates with the direct oxidation of N-N-propyl-beta-hydroxy-4-methylsulfonyl phenethylamine, a readily accessible starting material. Instead of chromium reagents, the process employs benign and cost-effective oxidants such as potassium permanganate, hydrogen peroxide, or pyridinium chlorochromate in a controlled solvent system. This is immediately followed by a condensation cyclization with p-tolyl acetate using simple alkali reagents. This strategic simplification reduces the number of unit operations, minimizes solvent consumption, and ensures that the impurity profile remains tightly controlled throughout the synthesis, thereby facilitating easier isolation of the target molecule.
Mechanistic Insights into Oxidation and Base-Catalyzed Cyclization
The core of this technological advancement lies in the precise execution of the oxidation step and the subsequent intramolecular cyclization. In the first stage, the secondary alcohol moiety of the phenethylamine derivative is selectively oxidized to a ketone. The patent specifies that this transformation can be achieved under mild conditions (20 to 60°C) using a variety of oxidants, including potassium permanganate or hydrogen peroxide, in a biphasic system containing water and organic solvents like dichloroethane or acetonitrile. This flexibility allows manufacturers to choose the most economically viable oxidant without sacrificing yield, which is reported to be exceptionally high, often exceeding 90%. The absence of over-oxidation or side reactions at the amine nitrogen is a critical mechanistic feature that ensures the integrity of the propyl-amino chain is maintained for the subsequent ring closure.
Following the formation of the ketone intermediate, the synthesis proceeds via a Claisen-type condensation mechanism facilitated by a strong base. The 2-n-propylamino-1-p-methylsulfonylacetophenone reacts with p-tolyl acetate (either methyl or ethyl ester) in the presence of alkali reagents such as sodium isopropoxide or sodium hydroxide. The base deprotonates the alpha-carbon of the ester, generating a nucleophilic enolate that attacks the ketone carbonyl. This is followed by dehydration and intramolecular nucleophilic attack by the amine nitrogen onto the ester carbonyl, closing the five-membered pyrrolidinone ring. This cascade reaction is highly atom-economical and occurs smoothly at temperatures between 20 to 90°C, delivering the final Imrecoxib scaffold with remarkable efficiency and minimal byproduct formation.
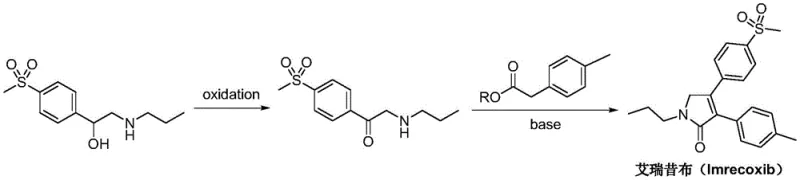
How to Synthesize Imrecoxib Efficiently
The implementation of this synthetic route requires careful attention to stoichiometry and temperature control to maximize the yield and purity of the final API intermediate. The process begins with the dissolution of the hydroxy-amine precursor in a suitable solvent, followed by the controlled addition of the oxidant and water. Once the oxidation is complete, typically within 2 to 8 hours, the intermediate is isolated and subjected to the cyclization conditions. The detailed standardized operating procedures, including specific molar ratios of the ketone intermediate to the p-tolyl acetate ester (1.0:1.1 to 1.5) and the precise addition rates of the alkali catalyst, are critical for reproducibility. For a comprehensive breakdown of the exact experimental parameters and workup procedures, please refer to the technical guide below.
- Oxidize N-propyl-beta-hydroxy-4-methylsulfonyl phenethylamine using an oxidant like potassium permanganate or hydrogen peroxide in a solvent-water system at 20-60°C to form the ketone intermediate.
- React the resulting 2-n-propylamino-1-p-methylsulfonylacetophenone with p-tolyl acetate (methyl or ethyl ester) in the presence of a strong alkali reagent.
- Maintain the condensation cyclization reaction between 20-90°C for 6-12 hours, followed by recrystallization to obtain high-purity Imrecoxib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers transformative benefits that extend far beyond simple yield improvements. By eliminating the dependency on chromium-based oxidants, the process removes the substantial costs associated with hazardous waste treatment and regulatory compliance regarding heavy metal limits. The substitution of expensive coupling agents like CDI with commodity chemicals such as p-tolyl acetate and sodium hydroxide results in a drastic reduction in raw material expenditure. Furthermore, the simplified two-step sequence reduces the overall cycle time and equipment occupancy, allowing for higher throughput in existing manufacturing facilities without the need for significant capital investment in new infrastructure.
- Cost Reduction in Manufacturing: The economic impact of this route is driven by the replacement of high-cost reagents with inexpensive, bulk-available commodities. The elimination of transition metal catalysts removes the need for specialized scavenging resins or complex purification steps required to meet strict residual metal specifications. Additionally, the high conversion rates observed in both the oxidation and cyclization steps minimize the loss of valuable starting materials, leading to a significantly improved overall mass balance and lower cost of goods sold (COGS) for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The robustness of this synthesis is underpinned by the use of stable and widely available starting materials. Unlike specialized epoxides or acyl chlorides that may suffer from supply volatility or short shelf-lives, the precursors used in this route, such as p-tolyl acetate and common alkali bases, are produced on a massive global scale. This ensures a consistent and reliable supply chain, mitigating the risk of production delays caused by raw material shortages. The simplicity of the reaction conditions also reduces the likelihood of batch failures, ensuring steady delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental perspective, this process aligns perfectly with modern green chemistry principles. The avoidance of toxic chromium waste simplifies effluent treatment and reduces the environmental liability of the manufacturing site. The reaction solvents, such as ethanol and isopropanol, are relatively benign and can be easily recovered and recycled. This eco-friendly profile not only facilitates regulatory approval but also enhances the corporate sustainability metrics of the manufacturer, making it an attractive option for multinational corporations with strict vendor code of conduct requirements regarding environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Imrecoxib synthesis. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, aiming to clarify the operational advantages and quality implications of this new method for potential partners and licensees.
Q: How does this new synthetic route improve upon traditional methods involving Jones reagent?
A: Traditional methods often rely on Jones reagent (chromic acid), which introduces toxic chromium residues that are difficult to remove and pose environmental hazards. The new route utilizes safer oxidants like potassium permanganate or hydrogen peroxide, eliminating heavy metal contamination and simplifying purification.
Q: What are the key cost drivers reduced in this Imrecoxib manufacturing process?
A: The process eliminates the need for expensive condensing agents like CDI and avoids the high costs associated with hazardous waste disposal for chromium byproducts. Additionally, the use of readily available esters and common alkali reagents significantly lowers raw material procurement costs.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is specifically designed for industrial scalability. It features simple operation steps, mild reaction conditions (20-90°C), and high yields (reported up to 95%), making it robust for commercial scale-up from pilot plants to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imrecoxib Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this optimized synthetic route to redefine the market availability of high-quality Imrecoxib. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of enforcing stringent purity specifications, guaranteeing that every batch of pharmaceutical intermediate meets the highest global standards for safety and efficacy. We are committed to leveraging this advanced chemistry to deliver superior value to our clients.
We invite you to collaborate with us to explore the full commercial potential of this technology. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can support your supply chain with reliable, cost-effective, and high-purity Imrecoxib solutions.
