Revolutionizing Erexib Production: A High-Yield, Chromium-Free Synthetic Route for Commercial Scale-Up
Introduction to Advanced Erexib Manufacturing
The pharmaceutical landscape for non-steroidal anti-inflammatory drugs (NSAIDs) continues to evolve, with Erexib (Imrecoxib) standing out as a highly selective COX-2 inhibitor offering superior safety profiles regarding gastrointestinal protection. The efficient production of this active pharmaceutical ingredient (API) is critical for meeting global demand while adhering to stringent environmental and quality standards. Patent CN110386891B introduces a groundbreaking preparation method that fundamentally shifts the paradigm from hazardous, multi-step oxidations to a streamlined, high-efficiency amidation and cyclization strategy. This technical insight explores how this novel approach delivers a reliable API intermediate supplier solution by achieving total yields exceeding 96% and purity levels greater than 99.8%, effectively addressing the bottlenecks of legacy manufacturing processes.
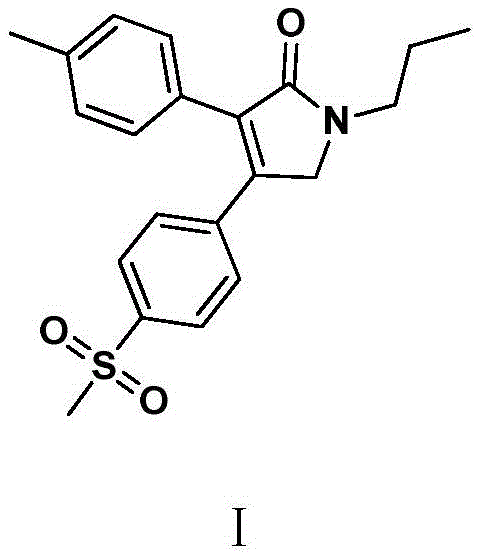
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Erexib has been plagued by significant operational and environmental hurdles that hinder cost reduction in pharmaceutical manufacturing. Traditional Route 1, for instance, relies heavily on the oxidation of hydroxy intermediates using Jones reagent or chromium trioxide-pyridine complexes. This not only introduces toxic hexavalent chromium into the process stream—necessitating complex and costly heavy metal removal protocols—but also suffers from low overall yields ranging merely from 26.1% to 38.5%. Furthermore, alternative pathways involving alpha-bromo ketones often require harsh conditions, such as heating to 160°C in acetic acid, which compromises reaction selectivity and generates substantial byproducts that are difficult to separate via standard recrystallization techniques.
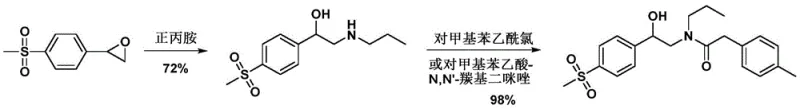
The Novel Approach
In stark contrast, the methodology disclosed in CN110386891B bypasses these oxidative and high-temperature pitfalls entirely by utilizing a convergent two-step sequence. The process initiates with a direct amidation between alpha-N-propylamino-4-methylsulfonyl acetophenone and a 4-methyl phenylacetate derivative, forming a stable amide intermediate with exceptional efficiency. This is followed by a sophisticated intramolecular cyclization that avoids the instability issues associated with alpha-halo ketones. By eliminating the need for column chromatography and enabling simple filtration for purification, this route drastically simplifies the downstream processing workflow, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Ester-Buffered Intramolecular Cyclization
The core innovation of this synthesis lies in the mechanistic control exerted during the cyclization of the amide intermediate (IV). In conventional base-mediated cyclizations, the generation of strong alkoxides often leads to the hydrolysis of the newly formed amide bond or the degradation of the pyrrolidone ring itself. The patented process ingeniously incorporates an ester additive, such as methyl acetate or ethyl acetate, directly into the reaction medium. This ester serves as a sacrificial scavenger that rapidly reacts with any free hydroxide ions generated during the reaction, effectively buffering the system. This conversion of a strong base into a weaker carboxylate species preserves the integrity of the sensitive functional groups, ensuring that the reaction proceeds selectively towards the desired cyclic product rather than degrading into open-chain impurities.
Furthermore, the protocol emphasizes the kinetic control of carbanion concentration through a controlled dropwise addition of the substrate. By maintaining a low instantaneous concentration of the active enolate species, the process suppresses intermolecular condensation reactions that would otherwise lead to oligomeric byproducts. This precise manipulation of reaction kinetics is crucial for achieving the reported liquid phase purity of over 99.8% without the need for extensive purification. Such mechanistic finesse demonstrates a deep understanding of physical organic chemistry, translating directly into robust process reliability for high-purity API intermediates.
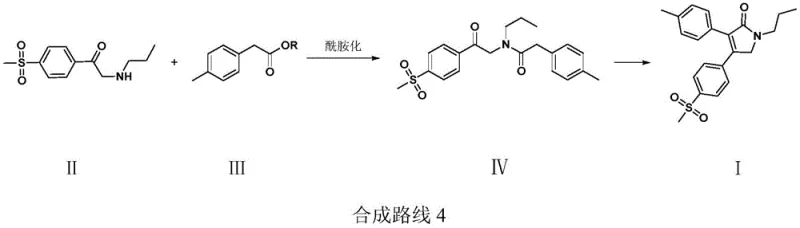
How to Synthesize Erexib Efficiently
Implementing this synthesis requires careful attention to solvent selection and temperature control to maximize the benefits of the ester-buffering effect. The process begins with the preparation of the key amide intermediate in solvents like toluene or DMF, followed by the critical cyclization step in alcoholic solvents. The following guide outlines the standardized operational parameters derived from the patent examples to ensure reproducibility and optimal yield. For detailed standard operating procedures and specific stoichiometric ratios, please refer to the technical guide below.
- Perform an amidation reaction between alpha-N-propylamino-4-methylsulfonyl acetophenone (II) and a 4-methyl phenylacetate (III) in a solvent like toluene or DMF at 70-100°C to obtain the amide intermediate (IV).
- Prepare a reaction mixture containing a base (e.g., sodium methoxide) and an ester buffer (e.g., methyl acetate) in a solvent such as methanol or THF.
- Dropwise add the intermediate (IV) to the base/ester mixture at 40-60°C to facilitate intramolecular cyclization while suppressing oligomerization and hydrolysis side reactions.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain directors, the transition to this novel synthetic route offers profound advantages that extend beyond mere chemical elegance. The elimination of chromium-based oxidants removes a major regulatory and disposal burden, significantly lowering the environmental compliance costs associated with waste treatment. Moreover, the stability of the intermediates allows for more flexible inventory management, reducing the risk of raw material degradation during storage and transport. This robustness translates directly into a more resilient supply chain capable of meeting tight production schedules without the delays often caused by complex purification failures or equipment corrosion from harsh reagents.
- Cost Reduction in Manufacturing: The dramatic improvement in overall yield, rising from roughly 30% in legacy methods to over 96% in this new process, fundamentally alters the cost structure of production. By nearly tripling the output per unit of raw material input, manufacturers can achieve substantial savings on starting materials. Additionally, the removal of expensive heavy metal scavengers and the simplification of purification from column chromatography to simple filtration drastically reduces operational expenditures and solvent consumption, driving down the total cost of goods sold.
- Enhanced Supply Chain Reliability: The use of commercially available and stable starting materials, such as alpha-N-propylamino-4-methylsulfonyl acetophenone, mitigates the supply risks associated with unstable alpha-halo ketones used in older routes. The mild reaction conditions (50-100°C) reduce the strain on reactor equipment, minimizing maintenance downtime and extending asset life. This operational stability ensures consistent batch-to-batch quality and reliable delivery timelines, which is critical for maintaining continuous API production lines.
- Scalability and Environmental Compliance: The process is inherently designed for green chemistry principles, generating minimal wastewater and avoiding toxic heavy metals. This makes the technology highly scalable from pilot plants to multi-ton commercial reactors without encountering the exponential increase in waste treatment costs typical of oxidation-heavy processes. The ability to produce high-purity material with a smaller environmental footprint aligns perfectly with modern corporate sustainability goals and regulatory requirements for reducing lead time for high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Erexib synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on purity profiles, scalability, and process safety.
Q: How does the new Erexib synthesis method improve upon traditional chromium-based routes?
A: The novel method eliminates the use of toxic chromium oxidants (like Jones reagent) found in prior art, thereby removing the need for expensive heavy metal clearance steps and significantly reducing environmental waste.
Q: What is the role of the ester additive in the cyclization step?
A: The ester acts as a buffering agent that reacts with byproduct hydroxides (formed from the base), converting strong bases into weaker ones. This prevents the hydrolysis of the sensitive amide bond and the pyrrolidone ring, ensuring high product purity.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process operates under mild conditions (50-100°C) without requiring high-pressure equipment or extreme temperatures (like the 160°C required in older routes), making it highly scalable and safe for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Erexib Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced NSAIDs like Erexib depends on accessing cutting-edge synthetic technologies. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of Erexib intermediate meets the highest global standards, providing our partners with the confidence needed to accelerate their drug development pipelines.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this efficient, chromium-free synthesis route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to obtain specific COA data and comprehensive route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of this critical therapeutic agent.
