Scalable Synthesis of Optically Pure 2-Methyl Alkyl Glycol Derivatives for Advanced Chiral Intermediates
The pharmaceutical and fine chemical industries constantly seek reliable sources of chiral synthetic building blocks that combine structural complexity with commercial viability. Patent CN101104577B introduces a groundbreaking preparation method for optically pure 2-methyl alkyl glycol derivatives, addressing the critical need for medium-length bifunctional structures with chiral methyl side chains. These compounds, characterized by the general formula Y-(CH2)n-CH(R1)(R2)-CH2-X, serve as indispensable precursors for high-value targets including Vitamin E, Vitamin K, and various insect sex pheromones. Unlike traditional asymmetric synthesis routes that often rely on expensive chiral auxiliaries, this technology leverages renewable resources to deliver stable, versatile intermediates. The structural versatility allows for diverse functionalization at both termini, enabling chemists to construct complex molecular architectures with precise stereochemical control.
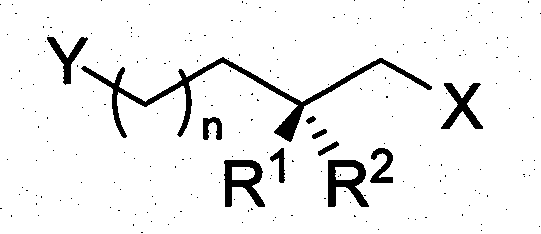
Historically, the synthesis of chiral building blocks with methyl side chains has been plagued by significant limitations in scalability and cost-efficiency. Conventional approaches frequently employ asymmetric synthesis techniques involving chiral prosthetic groups like Evans reagents or S(R)AMP, or direct reactions with chiral substrates such as BINOL-TiX2 complexes. While effective for laboratory-scale gram synthesis, these methods suffer from prohibitively long synthetic routes and the high cost of chiral reagents, rendering them unsuitable for industrial manufacturing. Alternatively, biotechnological methods like microbial asymmetric ester hydrolysis offer some promise but are often restricted by the availability of specific substrates and limitations on carbon chain length, typically failing to provide medium-length chains efficiently. Furthermore, natural product modification strategies using citronellol or citronellal are constrained by the finite variety of available natural scaffolds, limiting the structural diversity accessible to process chemists.
The novel approach detailed in this patent overcomes these barriers by utilizing optically pure lactones derived from the oxidative degradation of steroidal sapogenins, a renewable and abundant resource. By transforming unstable lactones like 4R-4-methylvalerolactone and 3R-3-methylbutyrolactone into stable chain compounds, the invention provides a robust platform for generating medium-length chiral synthons. This strategy not only valorizes waste streams from steroid drug production but also yields products with superior storage stability compared to their lactone precursors. The method enables the differentiation of two hydroxyl groups through selective protection, allowing for orthogonal functionalization that is crucial for multi-step organic synthesis. This represents a paradigm shift from expensive, linear asymmetric synthesis to a more economical, chiral-pool-based strategy that maintains high optical purity throughout the transformation.
Mechanistic Insights into Lactone Reduction and Selective Protection
The core of this synthetic strategy involves the controlled ring-opening of optically pure lactones followed by differentiated functionalization of the resulting diol system. The process begins with the reduction of the lactone carbonyl using powerful reducing agents such as lithium aluminum hydride (LiAlH4) in inert solvents like tetrahydrofuran, or alternatively, via methanolysis in the presence of acid or base catalysts. This step converts the cyclic ester into a linear 2-methyl alkyl glycol, preserving the stereocenter established in the starting lactone. The resulting diol possesses two hydroxyl groups with distinct steric environments; the primary hydroxyl is generally more accessible than the secondary hydroxyl adjacent to the chiral methyl center. This inherent difference in reactivity is exploited to achieve regioselective mono-protection.

Following the initial reduction, the selective protection of one hydroxyl group is achieved using a variety of protecting group reagents, including silyl chlorides (e.g., TBDMSCl), alkoxymethyl chlorides (e.g., MOMCl), or benzyl halides. The choice of reagent and conditions, such as the use of imidazole or triethylamine as bases, allows for precise control over which hydroxyl group is masked. For instance, bulky silyl groups often preferentially protect the less hindered primary alcohol, while specific acidic conditions might favor the secondary position depending on the substrate. Once one hydroxyl is protected, the remaining free hydroxyl group serves as a handle for further diversification. It can be converted into halides using phosphine-halogen complexes, oxidized to aldehydes using PDC or Swern conditions, or transformed into thioethers and acetals. This modularity ensures that a single chiral diol intermediate can be funneled into a wide array of downstream derivatives, maximizing the utility of the starting material.
How to Synthesize Optically Pure 2-Methyl Alkyl Glycol Derivatives Efficiently
The synthesis of these valuable chiral intermediates follows a logical sequence of reduction, protection, and functionalization steps that are amenable to standard process equipment. The protocol typically initiates with the dissolution of the optically pure lactone starting material in an anhydrous solvent, followed by the careful addition of a reducing agent under controlled temperature conditions to manage exotherms. After quenching and workup, the crude diol is subjected to selective protection conditions where stoichiometry and reaction time are critical parameters to ensure mono-substitution. The resulting protected alcohol can then be isolated and carried forward to the final functionalization step, whether it be halogenation, oxidation, or acetal formation. Detailed standardized synthetic steps see the guide below.
- Ring-opening of optically pure lactones (e.g., 4R-4-methylvalerolactone) via reduction with LiAlH4 or methanolysis to form diols or hydroxy esters.
- Selective mono-protection of the resulting diol hydroxyl groups using reagents like TBDMSCl, MOMCl, or benzyl bromide to differentiate functional handles.
- Functionalization of the remaining free hydroxyl group into halides, aldehydes, acetals, or thioethers to create diverse synthetic intermediates.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this lactone-based synthesis route offers compelling advantages in terms of cost structure and supply security. By shifting away from precious metal catalysts and expensive chiral ligands associated with traditional asymmetric synthesis, the manufacturing cost profile is significantly optimized. The reliance on steroidal sapogenin waste streams as a feedstock creates a circular economy model that insulates production from the volatility of petrochemical-derived chiral pool prices. This raw material strategy not only reduces direct material costs but also simplifies the supply chain by leveraging existing, large-scale steroid processing infrastructure. Consequently, this leads to substantial cost savings in chiral intermediate manufacturing without compromising on the optical purity required for regulatory compliance in pharmaceutical applications.
- Cost Reduction in Manufacturing: The elimination of costly chiral auxiliaries and transition metal catalysts drastically lowers the bill of materials for these intermediates. Furthermore, the high yields demonstrated in the patent examples, such as the 92% yield in the reduction step and 98% in the protection step, indicate a highly efficient process with minimal waste generation. This efficiency translates directly into lower processing costs per kilogram, as fewer raw materials are required to produce the same amount of active intermediate. The simplified post-treatment procedures, often involving basic filtration and distillation rather than complex chromatography, further reduce operational expenditures related to solvent consumption and labor.
- Enhanced Supply Chain Reliability: Sourcing starting materials from the degradation of steroidal sapogenins, which are produced in massive quantities for the steroid drug industry, ensures a stable and continuous supply of feedstock. Unlike niche chiral reagents that may face shortages or long lead times, these lactone precursors are derived from a robust, established industrial base. Additionally, the conversion of unstable lactones into stable chain-like glycol derivatives mitigates risks associated with raw material degradation during storage and transport. This stability allows for larger batch production and inventory buffering, ensuring that downstream customers receive consistent quality material regardless of short-term fluctuations in production schedules.
- Scalability and Environmental Compliance: The synthetic route utilizes common industrial reagents and solvents, facilitating seamless scale-up from pilot plant to commercial tonnage production. The avoidance of heavy metal catalysts simplifies waste treatment and reduces the environmental footprint of the manufacturing process, aligning with increasingly stringent global environmental regulations. The ability to produce medium-length chain derivatives that were previously difficult to access via biotechnology expands the addressable market for these intermediates. This scalability ensures that the technology can meet the growing demand for chiral building blocks in the agrochemical and pharmaceutical sectors without requiring specialized or exotic manufacturing capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these optically pure derivatives. The answers are derived directly from the patented methodology and its demonstrated benefits in organic synthesis. Understanding these aspects helps stakeholders evaluate the feasibility of integrating these building blocks into their own development pipelines. The robustness of the chemistry ensures that these intermediates can be reliably sourced for long-term projects.
Q: What are the primary starting materials for these chiral building blocks?
A: The process utilizes optically pure lactones such as 4R-4-methylvalerolactone and 3R-3-methylbutyrolactone, which can be sustainably sourced from the oxidative degradation waste of steroidal sapogenins.
Q: How does this method improve stability compared to traditional lactone precursors?
A: While the parent lactones are prone to polymerization and instability during storage, converting them into chain-like 2-methyl alkyl glycol derivatives significantly enhances their chemical stability and ease of handling for downstream synthesis.
Q: What are the key applications for these 2-methyl alkyl glycol derivatives?
A: These bifunctional chiral synthons are critical for the synthesis of Vitamin E, Vitamin K, insect sex pheromones, bioactive natural products, and ferroelectric liquid crystal monomers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl Alkyl Glycol Derivatives Supplier
As a leading CDMO and manufacturer, NINGBO INNO PHARMCHEM possesses the technical expertise to translate this patented laboratory methodology into robust commercial processes. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including optical rotation and enantiomeric excess, to guarantee that every batch meets the high standards required for API synthesis. We understand the critical nature of chiral integrity in drug development and are committed to delivering intermediates that facilitate your regulatory success.
We invite you to contact our technical procurement team to discuss how we can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for switching to this lactone-based route or require specific COA data and route feasibility assessments for a new target molecule, our experts are ready to assist. Partnering with us ensures access to a reliable pharmaceutical intermediates supplier dedicated to innovation, quality, and long-term supply chain partnership. Let us help you optimize your synthesis and secure your production pipeline with our advanced chiral technologies.
