Scalable Production of Optically Pure 2-Methyl Alkyl Glycol Derivatives for Advanced Pharmaceutical Synthesis
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for accessing chiral building blocks that possess medium-length carbon chains with specific stereochemical configurations. Patent CN101104579B introduces a groundbreaking preparation method for optically pure 2-methyl alkyl glycol derivatives, addressing a critical gap in the supply of bifunctional structures characterized by chiral methyl side chains. These compounds serve as indispensable precursors for the synthesis of high-value bioactive substances, including Vitamin E, Vitamin K, and various insect sex pheromones, as well as functional materials like ferroelectric liquid crystals. The innovation lies in the strategic utilization of optically pure lactones, specifically 4R-4-methylvalerolactone and 3R-3-methylbutyrolactone, which are transformed into stable, versatile chain compounds. This approach not only solves the instability issues associated with the parent lactones but also provides a scalable route to medium-length bifunctional synthetic blocks that were previously difficult to access in bulk quantities.
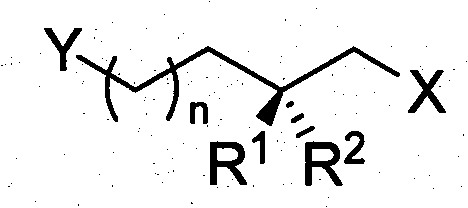
Traditionally, the synthesis of chiral synthetic building blocks with bifunctional structural features and chiral methyl side chains has relied heavily on asymmetric synthesis techniques involving chiral prosthetic groups such as Evans reagents or S(R)AMP, or direct reactions with chiral substrates like L-tartrate derivatives. While effective for laboratory-scale exploration, these conventional methods suffer from significant limitations when applied to industrial manufacturing. The synthetic routes are often excessively long, requiring multiple steps that erode overall yield, and the reagents involved, particularly specialized chiral catalysts and auxiliaries, are prohibitively expensive. Furthermore, these processes are generally restricted to gram-scale synthesis, making them unsuitable for the tonnage requirements of commercial API production. Alternative biotechnological approaches, such as microbial asymmetric ester hydrolysis, or modifications of natural products like citronellol, offer some relief but are constrained by the limited variety of available building blocks and unfavorable carbon chain lengths that are either too short or too long for specific applications.
The novel approach detailed in this patent fundamentally shifts the paradigm by leveraging the chiral pool inherent in steroidal sapogenin waste streams. By utilizing optically pure 4-methylvalerolactone and 3-methylbutyrolactone derived from the oxidative degradation of resources like diosgenin, the method converts these unstable cyclic esters into stable, open-chain diol derivatives. This transformation is achieved through controlled ring-opening reactions followed by selective protection strategies that differentiate the two hydroxyl groups. The result is a series of medium-length chiral methyl bifunctional synthetic building blocks that retain high optical purity while offering enhanced stability and handling properties. This strategy effectively turns a waste byproduct of the steroid industry into a high-value feedstock, significantly reducing the raw material costs associated with chiral synthesis while ensuring a consistent supply of structurally diverse intermediates suitable for complex organic synthesis.
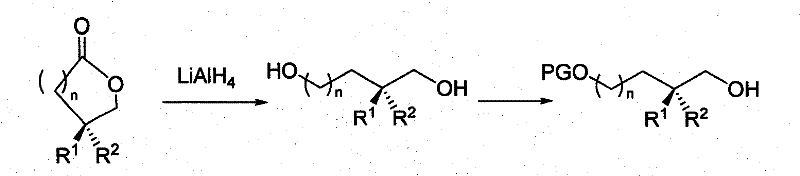
Mechanistically, the synthesis relies on the precise manipulation of the lactone ring to generate differentiated functionality. The process begins with the reduction or alcoholysis of the optically pure lactone. For instance, reduction with lithium aluminum hydride (LiAlH4) cleanly opens the ring to yield the corresponding diol, preserving the stereocenter at the methyl branch. Alternatively, acid-catalyzed methanolysis produces a hydroxy ester, which allows for orthogonal protection strategies. The key to the method's success lies in the selective protection of one hydroxyl group over the other. Reagents such as tert-butyldimethylsilyl chloride (TBDMSCl), methoxymethyl chloride (MOMCl), or benzyl bromide are employed under controlled conditions to protect the primary or secondary alcohol selectively. This differentiation is crucial as it enables subsequent transformations at the unprotected site, such as oxidation to an aldehyde using PDC or Swern conditions, or conversion to a halide using triphenylphosphine and carbon tetrabromide. The ability to toggle between different oxidation states and protecting groups without racemization ensures that the optical integrity of the chiral methyl center is maintained throughout the synthetic sequence.
Impurity control is inherently built into this chiral pool strategy. Since the starting lactones are derived from natural steroidal sapogenins which possess defined stereochemistry, the resulting building blocks inherit this optical purity, eliminating the need for costly chiral resolution steps or enantioselective catalysis that often introduce trace metal impurities. The reaction conditions, typically involving standard organic solvents like tetrahydrofuran or dichloromethane at moderate temperatures, minimize the formation of side products such as elimination byproducts or over-oxidized species. Furthermore, the selective protection steps are optimized to prevent bis-protection or incomplete reaction, which simplifies downstream purification. The use of robust protecting groups like silyl ethers or acetals ensures that the intermediates remain stable during storage and transport, addressing a major logistical pain point associated with the parent lactones which are prone to polymerization. This stability profile is critical for maintaining high purity specifications required by regulatory bodies for pharmaceutical intermediates.
How to Synthesize Optically Pure 2-Methyl Alkyl Glycol Derivatives Efficiently
The synthesis of these valuable chiral intermediates follows a logical progression from readily available lactone precursors to highly functionalized diol derivatives. The process is designed to be operationally simple, avoiding the need for exotic equipment or hazardous high-pressure conditions, which facilitates easy technology transfer from laboratory to pilot plant. The initial step involves the quantitative reduction of the lactone to a diol or its conversion to a hydroxy ester, setting the stage for divergent functionalization. Subsequent steps involve the careful selection of protecting groups to mask one hydroxyl function, thereby exposing the other for specific chemical modifications such as halogenation or oxidation. This modular approach allows chemists to access a wide library of derivatives from a single common intermediate, maximizing the utility of the starting material. For detailed standard operating procedures and specific reaction parameters regarding stoichiometry and workup, please refer to the standardized synthesis guide below.
- Start with optically pure lactones such as 4R-4-methylvalerolactone or 3R-3-methylbutyrolactone derived from steroidal sapogenin degradation.
- Perform ring-opening reactions using alcohols and acid/base catalysts, or direct reduction with LiAlH4 to obtain the corresponding diols.
- Execute selective protection of one hydroxyl group using reagents like TBDMSCl or MOMCl, followed by further functionalization to halides or aldehydes as needed.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this manufacturing technology offers distinct advantages that directly impact the bottom line and operational reliability. By sourcing raw materials from the degradation waste of steroidal sapogenins, the process decouples production from the volatile pricing of synthetic chiral catalysts and petrochemical feedstocks. This integration with existing steroid processing infrastructure creates a circular economy model where waste is valorized, leading to substantial cost savings in raw material acquisition. The elimination of expensive transition metal catalysts and chiral auxiliaries further reduces the cost of goods sold (COGS), as there is no need for rigorous heavy metal removal steps or the procurement of high-cost specialty reagents. Additionally, the simplified post-treatment procedures, often involving basic filtration and distillation, reduce solvent consumption and energy usage, contributing to a more sustainable and economically efficient manufacturing footprint.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the utilization of low-cost, renewable starting materials derived from agricultural waste streams rather than expensive synthetic chiral pools. By avoiding the use of precious metal catalysts and complex chiral ligands, the manufacturing process significantly lowers the input costs associated with reagent procurement. Furthermore, the high yields reported in the experimental examples indicate minimal material loss during conversion, which maximizes the throughput per batch. The stability of the final protected diol derivatives also reduces losses due to degradation during storage, ensuring that the inventory value is preserved over time. These factors combine to create a highly competitive cost structure for producing medium-length chiral building blocks compared to traditional asymmetric synthesis routes.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the abundance of steroidal sapogenin resources, which are produced on a massive scale globally for the hormone industry. This ensures a steady and reliable feedstock supply that is less susceptible to the disruptions often seen with niche synthetic reagents. The robustness of the chemical transformations, which utilize common industrial solvents and reagents, means that production can be easily scaled or shifted between manufacturing sites without requiring specialized equipment. The ability to produce a wide range of derivatives from a common intermediate also allows for flexible inventory management, where a single stock of protected diol can be rapidly converted into various halides or aldehydes based on immediate customer demand, thereby reducing lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to proceed efficiently from gram to multi-kilogram scales without loss of efficiency or selectivity. The reaction conditions are mild, typically operating at room temperature or under reflux, which minimizes energy consumption and thermal hazards. From an environmental standpoint, the valorization of steroid degradation waste reduces the ecological burden of the parent industry, while the absence of heavy metals simplifies wastewater treatment and waste disposal compliance. The use of standard organic solvents allows for established recovery and recycling protocols, aligning with green chemistry principles and reducing the overall environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these optically pure 2-methyl alkyl glycol derivatives. The answers are derived directly from the patented methodology and reflect the practical realities of implementing this chemistry in a commercial setting. Understanding these details is crucial for R&D teams evaluating route feasibility and procurement officers assessing supplier capabilities. The technology offers a unique blend of stereochemical control and functional versatility that makes it a preferred choice for complex molecule synthesis.
Q: What are the primary raw materials for these chiral building blocks?
A: The process utilizes optically pure lactones like 4R-4-methylvalerolactone and 3R-3-methylbutyrolactone, which are efficiently obtained from the oxidative degradation of steroidal sapogenins, turning waste streams into high-value intermediates.
Q: How does this method improve upon traditional asymmetric synthesis?
A: Unlike traditional methods relying on expensive chiral auxiliaries like Evans reagents or complex metal catalysts which are limited to gram scales, this chiral pool approach leverages renewable resources for large-scale production with simplified post-treatment and higher overall yields.
Q: What functional group transformations are possible with these derivatives?
A: The resulting diols feature differentiated hydroxyl groups that can be selectively protected, oxidized to aldehydes, or converted into halides and thioethers, providing versatile handles for constructing complex molecules like Vitamin E analogues or liquid crystal monomers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl Alkyl Glycol Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral building blocks play in the development of next-generation pharmaceuticals and agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation to verify optical purity and chemical identity. Our expertise in chiral pool synthesis allows us to optimize these patented routes for maximum yield and cost-efficiency, providing our partners with a competitive edge in their own supply chains.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Whether you require custom synthesis of a specific derivative or a feasibility assessment for a new target molecule, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term supply goals and drive innovation in your product pipeline.
