Advanced One-Step Synthesis of Chiral 2,3-Disubstituted Indoleamines for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex chiral scaffolds, particularly those found in bioactive natural products and drug candidates. A significant breakthrough in this domain is documented in Chinese Patent CN113461589B, which discloses a novel preparation method for chiral 2,3-disubstituted indoleamine compounds. This technology represents a paradigm shift from traditional multi-step syntheses to a streamlined, organocatalytic approach. By leveraging a chiral bissulfonimide as a small-molecule organic catalyst, the invention enables the direct formation of these valuable intermediates through an asymmetric Friedel-Crafts reaction. For R&D directors and procurement strategists, this development signals a move towards greener chemistry that does not compromise on stereochemical purity. The ability to generate high-value indole derivatives in a single operational step addresses critical bottlenecks in supply chain continuity and manufacturing cost structures, positioning this methodology as a cornerstone for next-generation API intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 2,3-disubstituted indoleamines has been fraught with inefficiencies that hinder large-scale adoption. Conventional methodologies typically rely on a two-step sequence involving the asymmetric Friedel-Crafts reaction of 4,7-dihydroindoles with imines, followed by a subsequent oxidation step to aromatize the ring system. This indirect route introduces significant operational complexity, requiring distinct reaction conditions, separate workup procedures, and additional purification stages for the intermediate dihydroindole species. Furthermore, the oxidation step often necessitates the use of stoichiometric oxidants, which can generate substantial chemical waste and pose safety hazards during scale-up. From a commercial perspective, these extra steps translate directly into increased processing time, higher solvent consumption, and reduced overall yield due to cumulative losses at each stage. The reliance on such cumbersome protocols limits the economic viability of producing these compounds, especially when high enantiomeric excess is required for pharmaceutical applications.
The Novel Approach
In stark contrast, the technology outlined in patent CN113461589B offers a direct, one-pot solution that bypasses the need for pre-functionalized dihydroindoles or post-reaction oxidation. The novel approach utilizes readily available 3-substituted indoles and aldimines as starting materials, reacting them directly in the presence of a chiral bissulfonimide catalyst. This strategy effectively collapses the synthetic timeline, eliminating the isolation of unstable intermediates and reducing the total number of unit operations. The reaction proceeds under mild conditions, typically in common organic solvents like toluene, and achieves high conversion rates with exceptional stereocontrol. By simplifying the workflow to a single reaction vessel, manufacturers can drastically reduce equipment occupancy time and labor costs. This direct functionalization not only enhances the atom economy of the process but also aligns perfectly with modern green chemistry principles, making it an attractive option for sustainable cost reduction in API manufacturing.
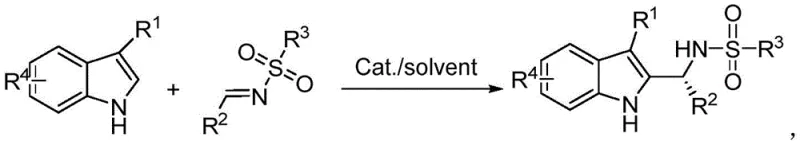
Mechanistic Insights into Chiral Bissulfonimide Catalysis
The success of this transformation hinges on the unique structural and electronic properties of the chiral bissulfonimide catalyst. Unlike traditional chiral phosphoric acids, the bissulfonimide catalyst employed in this invention features a BINOL (1,1'-bi-2-naphthol) skeleton coupled with dual sulfonyl groups. This specific architecture confers a stricter C2 symmetry axis and significantly stronger Brønsted acidity compared to its phosphate counterparts. Mechanistically, the catalyst operates by simultaneously activating both reaction partners through a dual hydrogen-bonding network. The acidic protons of the sulfonimide moiety form hydrogen bonds with the nitrogen atoms of both the indole and the aldimine substrates. This precise organization within the chiral pocket of the catalyst lowers the activation energy for the nucleophilic attack while rigidly controlling the spatial orientation of the approaching molecules. Such tight control is essential for discriminating between the prochiral faces of the imine, thereby ensuring the formation of a single enantiomer with high fidelity.
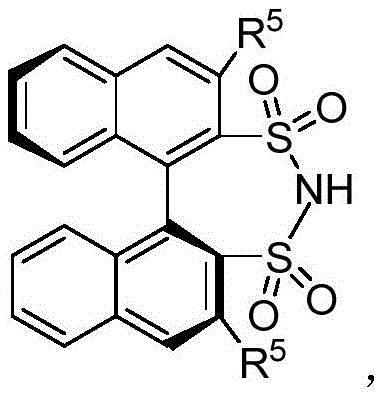
Beyond mere activation, the catalyst plays a pivotal role in impurity control and product quality. The high enantioselectivity observed, with experimental examples demonstrating ee values up to 98%, indicates that the transition state is highly organized, minimizing the formation of the undesired enantiomer. This level of stereochemical purity is critical for pharmaceutical intermediates, where even trace amounts of the wrong isomer can lead to regulatory rejection or toxicological issues. Furthermore, the mild reaction conditions (typically ranging from -10°C to 40°C) prevent thermal degradation of sensitive functional groups on the indole or imine rings, preserving the integrity of the molecular scaffold. The robustness of the catalyst across a wide substrate scope, accommodating various aryl and alkyl substituents, suggests a versatile mechanistic pathway that is tolerant to electronic and steric variations. This versatility ensures that the process remains reliable even when scaling up diverse derivatives for different drug discovery programs.
How to Synthesize Chiral 2,3-Disubstituted Indoleamines Efficiently
Implementing this advanced synthesis route requires careful attention to reaction parameters to maximize yield and enantioselectivity. The process begins with the preparation of a homogeneous catalytic solution, followed by the controlled addition of substrates to manage exotherms and ensure proper mixing. Optimization studies within the patent indicate that solvent choice is critical, with non-polar solvents like toluene providing superior results compared to polar protic solvents which may disrupt the crucial hydrogen-bonding interactions. Temperature control is another key variable; while the reaction can proceed at room temperature, slight heating to 35°C has been shown to accelerate kinetics without compromising stereoselectivity in many cases. The workup procedure is notably simple, involving a standard aqueous quench and extraction, which facilitates easy separation of the organic product from the catalyst and salts. For detailed operational protocols and specific molar ratios tailored to your production needs, please refer to the standardized guide below.
- Prepare a mixed dispersion by stirring a chiral bissulfonimide catalyst with an organic solvent such as toluene.
- Sequentially add aldimine and 3-substituted indole to the mixture and react under stirring at mild temperatures (e.g., 35°C).
- Quench the reaction with water, extract with ethyl acetate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits beyond mere technical elegance. The shift to a one-step process fundamentally alters the cost structure of producing these high-value intermediates. By eliminating the oxidation step and the associated reagents, the process reduces the raw material bill of materials (BOM) and minimizes waste disposal costs. The use of inexpensive, commodity-grade solvents like toluene further drives down operational expenditures compared to processes requiring exotic or highly purified solvents. Additionally, the simplicity of the workup—requiring only water quenching and standard extraction—reduces the demand for specialized downstream processing equipment, allowing existing facilities to adapt quickly without major capital investment. These factors combine to create a leaner, more cost-effective manufacturing model that enhances margin potential for final API products.
- Cost Reduction in Manufacturing: The elimination of the multi-step sequence traditionally required for indoleamine synthesis results in substantial savings on labor, energy, and consumables. Removing the oxidation step avoids the cost of stoichiometric oxidants and the subsequent purification of oxidation byproducts. Furthermore, the high catalytic efficiency means that lower catalyst loadings can often be utilized while maintaining high turnover numbers, reducing the cost contribution of the chiral promoter. The overall simplification of the process flow leads to shorter batch cycles, increasing plant throughput and asset utilization rates without the need for additional reactor capacity.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, specifically 3-substituted indoles and various aldimines, are commercially available and chemically stable, ensuring a robust supply base. Unlike specialized dihydroindole precursors which may have limited suppliers and long lead times, these commoditized building blocks can be sourced from multiple vendors globally. This diversification of the supply base mitigates the risk of shortages and price volatility. Moreover, the mild reaction conditions reduce the safety risks associated with handling hazardous oxidants or extreme temperatures, leading to fewer unplanned shutdowns due to safety incidents and ensuring consistent delivery schedules to downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard organic synthesis techniques that are well-understood in industrial settings. The absence of heavy metal catalysts eliminates the need for expensive and complex metal scavenging steps, which are often a bottleneck in GMP manufacturing. This metal-free approach simplifies regulatory filings and reduces the environmental footprint of the manufacturing process by lowering the E-factor (mass of waste per mass of product). The use of recyclable solvents and the generation of benign aqueous waste streams align with increasingly stringent environmental regulations, future-proofing the supply chain against evolving compliance requirements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing pipelines. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and reliability for decision-making purposes. Whether you are concerned about substrate scope limitations or specific purification requirements, these insights provide a foundational understanding of the process capabilities.
Q: What is the primary advantage of this new synthesis method over conventional routes?
A: The primary advantage is the reduction from a two-step process involving oxidation to a direct one-step asymmetric Friedel-Crafts reaction, significantly improving efficiency and atom economy.
Q: What level of enantioselectivity can be achieved with this catalyst system?
A: The chiral bissulfonimide catalyst system demonstrates high enantioselectivity, with experimental data showing ee values reaching up to 98% under optimized conditions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions, inexpensive solvents like toluene, and readily available starting materials, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 2,3-Disubstituted Indoleamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalytic methods like the one described in CN113461589B for accelerating drug development timelines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs are seamlessly translated into industrial reality. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs dedicated to verifying the enantiomeric excess and chemical purity of every batch. We understand that in the competitive landscape of pharmaceutical intermediates, consistency and reliability are paramount, and our state-of-the-art facilities are designed to deliver exactly that.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific project needs. Our technical team is ready to conduct a Customized Cost-Saving Analysis to quantify the economic benefits of switching to this one-step route for your target molecules. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive proposal tailored to your volume requirements. Let us help you optimize your supply chain and bring your chiral indoleamine projects to market faster and more efficiently.
