Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
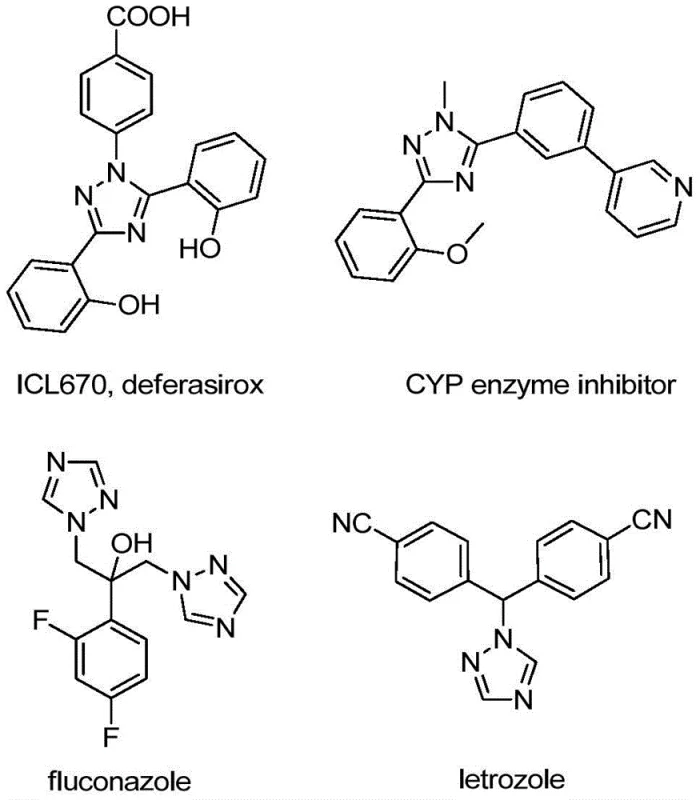
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those functionalized with trifluoromethyl groups, due to their profound impact on metabolic stability and lipophilicity. Patent CN110467579B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in current synthetic routes. This technology leverages a non-metallic iodine-promoted cyclization strategy that operates under mild conditions, eliminating the stringent requirement for anhydrous or oxygen-free environments often associated with traditional organometallic catalysis. For R&D directors and process chemists, this represents a significant opportunity to streamline the synthesis of complex molecular scaffolds found in high-value active pharmaceutical ingredients (APIs) and functional materials. The versatility of this approach allows for the facile introduction of diverse substituents at the 4 and 5 positions of the triazole ring, thereby expanding the chemical space available for drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethylated nitrogen heterocycles has relied heavily on two primary strategies, both of which present substantial logistical and economic challenges for large-scale manufacturing. The first conventional pathway involves the direct trifluoromethylation of pre-synthesized 1,2,4-triazole rings, a process that frequently necessitates the use of specialized and often hazardous trifluoromethylating reagents which are expensive and difficult to handle safely on a multi-kilogram scale. The second mainstream approach utilizes synthons bearing trifluoromethyl groups, such as trifluorodiazoethane, to react with coupling substrates; however, diazo compounds are inherently unstable and pose significant safety risks regarding explosivity, requiring specialized equipment and rigorous safety protocols that drive up capital expenditure. Furthermore, many existing methods rely on transition metal catalysts like palladium or copper, which not only increase raw material costs but also introduce the complex and costly downstream burden of removing trace heavy metal residues to meet stringent regulatory limits for pharmaceutical products.
The Novel Approach
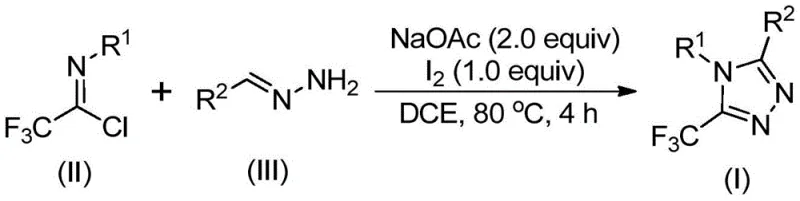
In stark contrast to these legacy methods, the novel methodology outlined in the patent utilizes a simple yet highly effective combination of trifluoroethylimidoyl chloride and hydrazones promoted by elemental iodine. This iodine-promoted system operates efficiently in common organic solvents such as dichloroethane (DCE) at a moderate temperature of 80°C, drastically simplifying the operational complexity of the reaction setup. By avoiding the use of toxic heavy metal catalysts and unstable diazo reagents, this route significantly enhances the safety profile of the manufacturing process while simultaneously reducing the environmental footprint associated with waste disposal. The reaction demonstrates excellent functional group tolerance, allowing for the successful synthesis of various derivatives including those with halogen, alkyl, and alkoxy substituents, as evidenced by the high yields obtained in experimental examples ranging from 72% to 97%. This robustness makes it an ideal candidate for the cost reduction in pharmaceutical intermediate manufacturing, providing a reliable supply chain for critical building blocks.
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic pathway of this transformation is a sophisticated sequence of base-promoted condensation and oxidative aromatization events that ensure high selectivity and purity. The reaction initiates with the nucleophilic attack of the hydrazone nitrogen on the electrophilic carbon of the trifluoroethylimidoyl chloride, facilitated by sodium acetate acting as a mild base to scavenge the generated hydrochloric acid. This initial intermolecular carbon-nitrogen bond formation yields a trifluoroacetamidine intermediate, which subsequently undergoes isomerization to align the reactive centers for cyclization. The addition of elemental iodine serves as a crucial oxidant, promoting the oxidative iodination of the intermediate to generate an iodine-substituted species that is primed for the final ring-closing step. This iodine-mediated activation lowers the energy barrier for the subsequent intramolecular electrophilic substitution, driving the formation of the five-membered triazole ring with high efficiency.
From an impurity control perspective, the mild nature of the reagents and the specific sequence of addition play a pivotal role in minimizing side reactions. Unlike harsh acidic or basic conditions that might degrade sensitive functional groups on the aromatic rings, the sodium acetate/iodine system maintains a relatively neutral to slightly basic environment that preserves the integrity of substituents such as esters, nitriles, or halogens. The final aromatization step is driven thermodynamically by the formation of the stable aromatic triazole system, which effectively pulls the equilibrium towards the desired product and suppresses the accumulation of partially cyclized byproducts. This mechanistic clarity allows process chemists to fine-tune reaction parameters, such as the molar ratio of iodine to substrate (optimized at roughly 1:1 to 1.5:1), to maximize conversion rates and simplify the downstream purification process, ensuring the delivery of high-purity OLED material or pharmaceutical precursors.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
The synthesis protocol described in the patent offers a straightforward operational procedure that is amenable to both laboratory optimization and industrial scale-up. The process begins by charging a reaction vessel with sodium acetate, trifluoroethylimidoyl chloride, and the appropriate hydrazone derivative in an organic solvent, typically dichloroethane, which provides excellent solubility for all reactants. The mixture is heated to 80°C and stirred for an initial period of 2 to 4 hours to allow the condensation and initial cyclization to proceed to completion before the oxidant is introduced. Following this incubation period, elemental iodine is added directly to the reaction system, and heating is continued for an additional 1 to 2 hours to effect the oxidative aromatization, after which the crude product can be isolated via simple filtration and purified by standard column chromatography techniques.
- Mix sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane (DCE).
- Heat the reaction mixture to 80°C and stir for 2 to 4 hours to allow initial condensation and cyclization.
- Add elemental iodine to the system and continue heating for an additional 1 to 2 hours to promote oxidative aromatization, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this iodine-promoted synthesis route offers compelling economic and logistical advantages over traditional metal-catalyzed processes. The elimination of expensive transition metal catalysts such as palladium or rhodium removes a significant variable cost component from the bill of materials, while also negating the need for specialized scavenging resins or complex purification steps required to meet residual metal specifications. Furthermore, the starting materials, including trifluoroethylimidoyl chloride and various hydrazones, are derived from commodity chemicals that are widely available in the global market, ensuring a stable and resilient supply chain that is less susceptible to geopolitical disruptions or single-source bottlenecks. The operational simplicity of the process, which does not require inert atmosphere gloveboxes or cryogenic cooling, further reduces the utility costs and infrastructure investment needed for commercial production.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with inexpensive elemental iodine and sodium acetate results in a drastic reduction in raw material costs, while the simplified workup procedure minimizes labor and solvent consumption. By avoiding the use of hazardous diazo reagents, the facility also saves on the substantial insurance and safety compliance costs associated with handling explosive intermediates. The high atom economy of the reaction, coupled with the ability to recycle solvents like dichloroethane, contributes to a leaner manufacturing model that improves overall gross margins for the final API or intermediate.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials ensures that production schedules can be maintained without the long lead times often associated with custom-synthesized organometallic reagents. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or moisture, reducing the risk of batch failures and ensuring consistent output quality. This reliability is critical for maintaining continuous supply to downstream customers in the pharmaceutical and agrochemical sectors, where interruption of supply can have cascading effects on drug availability.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently from gram-scale laboratory experiments to potential ton-scale production without significant modification of the reaction parameters. The absence of heavy metals simplifies the treatment of wastewater and solid waste, facilitating easier compliance with increasingly stringent environmental regulations regarding heavy metal discharge. Additionally, the use of standard organic solvents allows for integration into existing manufacturing suites without the need for specialized corrosion-resistant equipment, accelerating the timeline from process development to commercial launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 5-trifluoromethyl-1,2,4-triazole synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on reaction scope, safety, and scalability. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the method described in patent CN110467579B utilizes elemental iodine and sodium acetate as promoters, completely avoiding the need for costly palladium or copper catalysts.
Q: What are the typical reaction conditions for this triazole formation?
A: The reaction proceeds efficiently in aprotic solvents like dichloroethane at moderate temperatures around 80°C, without the need for strict anhydrous or anaerobic conditions.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the process uses cheap and readily available starting materials and simple post-treatment steps like filtration and column chromatography, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient heterocycle synthesis in the development of next-generation therapeutics and functional materials. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and high-quality supply of critical intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,4-triazole meets the exacting standards required for clinical and commercial applications. We are committed to leveraging innovative technologies, such as the iodine-promoted cyclization method, to deliver superior value to our global partners.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this metal-free methodology for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your long-term growth and innovation goals in the competitive landscape of fine chemical manufacturing.
