Revolutionizing Pharmaceutical Intermediate Production with Scalable Iodine-Promoted Trifluoromethyl Triazole Synthesis Technology
The recently granted Chinese patent CN110467579B represents a significant advancement in heterocyclic chemistry by introducing a novel synthetic route for producing critical pharmaceutical building blocks—specifically the class of compounds known as 5-trifluoromethyl substituted 1,2,4-triazoles which serve as essential structural motifs in numerous bioactive molecules including antifungal agents like fluconazole and cancer therapeutics such as letrozole. This innovative methodology directly addresses longstanding industry challenges by eliminating both anhydrous/anaerobic reaction requirements and toxic heavy metal catalysts that have historically complicated manufacturing processes while introducing significant cost burdens through additional purification steps required to meet regulatory standards for pharmaceutical intermediates. The process leverages readily accessible starting materials including sodium acetate as a mild base promoter alongside commercially available trifluoroethylimidoyl chloride derivatives and hydrazones to construct these valuable triazole scaffolds through a streamlined iodine-mediated cyclization sequence that operates effectively under standard laboratory conditions without specialized equipment or environmental controls. Notably, this approach demonstrates exceptional functional group tolerance across diverse substrates enabling precise molecular engineering of triazole derivatives with trifluoromethyl groups positioned at different locations through strategic substrate design rather than relying on post-synthetic modifications that often reduce overall yield and increase complexity. The method's operational simplicity combined with its demonstrated scalability from gram-scale laboratory validation to potential multi-ton manufacturing represents a paradigm shift from conventional synthetic routes that frequently depend on expensive trifluoromethylation reagents or harsh reaction environments incompatible with commercial production economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for incorporating trifluoromethyl groups into nitrogen-containing heterocycles typically require either pre-functionalized substrates subjected to challenging trifluoromethylation reactions using specialized reagents such as Umemoto's or Togni's reagents that are costly and often unstable under standard processing conditions or rely on transition metal-catalyzed cross-coupling methodologies that introduce significant purification challenges due to persistent metal contamination concerns requiring extensive downstream processing to meet pharmaceutical quality standards. These conventional methods frequently demand strictly anhydrous and anaerobic environments that are difficult to maintain consistently at manufacturing scale leading to batch-to-batch variability in product quality while substantially increasing operational complexity through specialized reactor requirements and continuous monitoring systems that drive up capital expenditure and operational costs. Furthermore many existing protocols exhibit limited substrate scope with poor functional group tolerance restricting their applicability across diverse molecular architectures required in modern pharmaceutical development pipelines where structural diversity is essential for optimizing drug efficacy and safety profiles during lead compound optimization stages. The reliance on heavy metal catalysts not only increases raw material expenses but also necessitates additional processing steps including multiple chromatographic purifications or specialized extraction techniques to remove trace metal residues below regulatory thresholds thereby extending production timelines and reducing overall process efficiency while generating additional waste streams requiring costly disposal protocols.
The Novel Approach
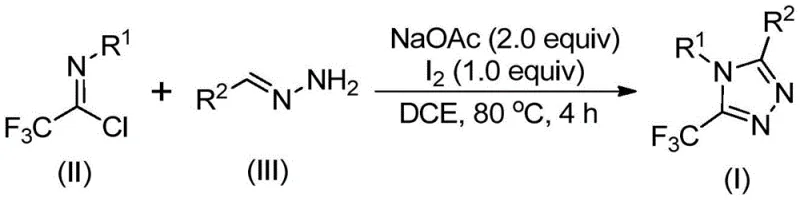
The patented methodology overcomes these limitations through an elegant iodine-promoted cyclization strategy that operates under ambient atmospheric conditions without requiring moisture or oxygen exclusion while completely avoiding transition metals through a carefully designed reaction sequence starting with base-mediated coupling between readily available trifluoroethylimidoyl chloride derivatives and hydrazones followed by iodine-mediated cyclization under mild thermal activation at precisely controlled temperatures between 80°C and optimal reaction times of three hours before introducing elemental iodine as a non-metal promoter for the final ring-closure step. This approach leverages commercially accessible starting materials including sodium acetate as a mild base that facilitates carbon-nitrogen bond formation without generating hazardous byproducts while utilizing dichloroethane as a preferred solvent that effectively dissolves all components without requiring specialized handling procedures or expensive solvent recovery systems typically needed with more volatile or toxic alternatives. The process demonstrates remarkable functional group tolerance across diverse substrates enabling strategic modification of R¹ and R² positions through commercially available precursors which allows pharmaceutical manufacturers to rapidly generate customized triazole scaffolds meeting specific drug development requirements without redesigning the core synthetic pathway thereby significantly accelerating lead optimization timelines while maintaining high purity standards essential for clinical-stage materials. Crucially this method eliminates both the capital investment required for specialized moisture-free reactors and the operational costs associated with metal removal processes creating immediate economic advantages while simultaneously improving environmental sustainability through reduced waste generation compared to conventional approaches.
Mechanistic Insights into Iodine-Promoted Triazole Cyclization
The reaction proceeds through a multi-step sequence initiated by base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and hydrazone substrates facilitated by sodium acetate which deprotonates the hydrazone nitrogen enabling nucleophilic attack on the electrophilic carbon of the imidoyl chloride moiety to form a key trifluoroacetamidine intermediate that subsequently undergoes spontaneous isomerization under thermal activation at elevated temperatures. This intermediate then participates in base-promoted oxidative iodination where elemental iodine acts as both an oxidant and promoter generating an iodinated species that undergoes intramolecular electrophilic substitution at the ortho position relative to the nitrogen atom followed by aromatization through proton elimination completing the five-membered triazole ring formation with precise regiocontrol ensuring the trifluoromethyl group occupies the desired position without requiring additional directing groups or protecting strategies. The mechanism's elegance lies in its avoidance of transition metals entirely while maintaining high regioselectivity across diverse substrate combinations through careful manipulation of electronic properties inherent in the starting materials rather than relying on external catalysts that introduce complexity and contamination risks.
Impurity profile management is achieved through precise control of reaction stoichiometry where maintaining a molar ratio of sodium acetate to elemental iodine between 2.0–3.0:1 ensures complete conversion while minimizing side reactions that could generate regioisomers or decomposition products; additionally the use of dichloroethane as solvent provides optimal polarity balance that facilitates intermediate solubility without promoting unwanted side reactions commonly observed in more polar solvents like DMSO which can lead to over-reaction or decomposition under prolonged heating conditions. The post-reaction workup involving simple filtration followed by silica gel chromatography effectively removes any residual iodine species or minor byproducts while preserving product integrity through gentle purification conditions that avoid thermal degradation pathways observed in more aggressive isolation techniques; this straightforward purification protocol contributes significantly to achieving high purity levels exceeding typical industry standards without requiring specialized equipment or additional processing steps that would increase manufacturing costs or reduce overall yield efficiency.
How to Synthesize Trifluoromethyl Triazoles Efficiently
This patented methodology provides pharmaceutical manufacturers with a robust pathway for producing high-purity triazole intermediates through a carefully optimized sequence that begins with strategic selection of commercially available starting materials including sodium acetate as a mild base promoter alongside appropriately substituted trifluoroethylimidoyl chloride derivatives and hydrazones tailored to achieve specific molecular targets required for drug development programs; the process operates effectively under standard laboratory conditions without requiring specialized environmental controls thereby simplifying technology transfer from research settings to manufacturing environments while maintaining consistent product quality across different scales of operation. The following standardized procedure details the critical parameters necessary for successful implementation of this innovative synthesis route which has been validated across multiple substrate combinations demonstrating exceptional reproducibility and reliability essential for commercial pharmaceutical intermediate production where consistency is paramount.
- Combine sodium acetate with trifluoroethylimidoyl chloride and hydrazone substrates in dichloroethane solvent under ambient atmospheric conditions without requiring moisture or oxygen exclusion.
- Heat the reaction mixture to precisely controlled temperatures between 80°C and maintain for three hours to facilitate carbon-nitrogen bond formation and intermediate cyclization.
- Introduce elemental iodine as a non-metal promoter followed by one-hour reaction period before standard workup involving filtration and silica gel chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points faced by procurement and supply chain professionals in pharmaceutical manufacturing by transforming what was previously considered a complex specialty chemical process into a streamlined operation with significant economic benefits; the elimination of expensive transition metal catalysts removes both raw material costs and extensive downstream processing requirements while operating under standard atmospheric conditions simplifies facility requirements reducing capital expenditure needs for specialized reactor systems typically required for moisture-sensitive processes.
- Cost Reduction in Manufacturing: The complete avoidance of transition metal catalysts eliminates not only their substantial procurement costs but also removes multiple downstream processing steps required for metal removal including specialized chromatography columns or extraction techniques that consume significant time and resources while generating additional waste streams requiring costly disposal protocols; this streamlined approach substantially reduces overall manufacturing costs through simplified process flow without compromising product purity standards essential for pharmaceutical applications.
- Enhanced Supply Chain Reliability: Utilizing readily available starting materials including sodium acetate which is globally sourced from multiple suppliers ensures consistent raw material availability while eliminating dependency on specialized reagents with limited supply chains; this strategic material selection combined with robust process stability under ambient conditions significantly reduces supply chain vulnerability by enabling flexible sourcing options and minimizing production disruptions caused by single-source dependencies common in traditional synthetic routes.
- Scalability and Environmental Compliance: The method's compatibility with standard manufacturing equipment operating under atmospheric conditions facilitates seamless scale-up from laboratory validation to commercial production volumes while generating minimal hazardous waste compared to conventional approaches; this inherent scalability combined with reduced environmental impact through elimination of heavy metals aligns perfectly with modern sustainability initiatives without requiring additional capital investment in specialized waste treatment systems.
Frequently Asked Questions (FAQ)
The following questions address common technical concerns raised by procurement teams regarding implementation of this patented methodology; each response is grounded in specific experimental data from patent CN110467579B demonstrating practical applicability across diverse manufacturing scenarios while highlighting key advantages relevant to commercial production environments where reliability and cost-effectiveness are paramount considerations.
Q: How does this method eliminate heavy metal contamination concerns while maintaining high regioselectivity?
A: The iodine-promoted mechanism avoids transition metals entirely through a base-mediated oxidative cyclization pathway that achieves precise regiocontrol via substrate design flexibility without requiring costly metal removal steps.
Q: What scalability advantages arise from operating without anhydrous/anaerobic conditions?
A: Eliminating stringent environmental controls significantly simplifies reactor design and process validation while reducing energy consumption during scale-up from laboratory to commercial manufacturing volumes.
Q: How does substrate design flexibility support diverse pharmaceutical intermediate production?
A: The broad functional group tolerance enables strategic modification of R¹ and R² positions through commercially available precursors to synthesize customized triazole scaffolds meeting specific drug development requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazoles Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through state-of-the-art QC labs equipped with advanced analytical instrumentation capable of detecting impurities at parts-per-billion levels; this technical expertise combined with our deep understanding of regulatory requirements ensures seamless integration of this innovative triazole synthesis into your manufacturing pipeline while delivering consistent high-quality intermediates essential for pharmaceutical applications where product integrity is non-negotiable.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who will provide specific COA data and route feasibility assessments tailored to your unique production requirements; contact us today to discuss how our CDMO capabilities can transform your triazole intermediate supply chain into a strategic competitive advantage.
