Revolutionizing Edaravone Production: Continuous Reactive Distillation for Commercial Scale-up
The pharmaceutical industry is constantly seeking robust manufacturing technologies that can bridge the gap between laboratory discovery and commercial viability, particularly for high-volume neuroprotective agents like Edaravone. Patent CN110922360B introduces a groundbreaking methodology for the continuous preparation of Edaravone, addressing the longstanding thermodynamic limitations inherent in traditional batch synthesis. This innovation leverages a sophisticated combination of reactive distillation and continuous crystallization to drive reaction equilibrium forward, ensuring that byproducts such as ethanol and water are synchronously and effectively removed from the reaction system. By integrating a rectifying tower directly with the reaction kettle, the process maintains a dynamic environment where the forward reaction is continuously favored, thereby drastically enhancing the conversion rate of the starting materials. For R&D directors and process engineers, this represents a significant leap forward in process intensification, offering a pathway to higher purity profiles and reduced downstream processing burdens. The ability to operate continuously rather than in discrete batches not only improves thermal efficiency but also establishes a more consistent product quality profile, which is paramount for regulatory compliance in the global pharmaceutical supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for Edaravone typically rely on batch condensation reactions between phenylhydrazine and ethyl acetoacetate, a process that is fundamentally constrained by chemical equilibrium dynamics. In a closed batch system, the accumulation of reaction byproducts, specifically ethanol and water, creates a thermodynamic barrier that inhibits the complete conversion of reactants into the desired pyrazolone derivative. As the concentration of these byproducts increases over time, the rate of the reverse reaction accelerates, leading to a plateau in yield that often necessitates extensive purification steps or the use of excessive amounts of raw materials to drive the reaction to completion. Furthermore, batch processing inherently suffers from operational inefficiencies, including significant downtime between runs for cleaning, charging, and discharging, which disrupts supply continuity and increases the overall cost of goods sold. The inability to continuously remove volatile byproducts in real-time means that energy consumption is often higher, as additional heating or vacuum stripping steps are required post-reaction to isolate the crude product, introducing potential thermal degradation risks to the sensitive API intermediate.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a continuous reactive distillation setup that fundamentally alters the reaction landscape by coupling chemical transformation with simultaneous separation. By communicating the top of the reaction kettle with a rectifying tower, the system allows for the immediate removal of ethanol and water as they are formed, effectively pulling the equilibrium towards the product side according to Le Chatelier's principle. This continuous removal prevents the buildup of inhibitory byproducts, allowing the reaction to proceed to near-completion levels without the need for drastic excesses of reagents. Moreover, the integration of a cooling crystallizer and continuous filtration centrifuge enables the synchronous separation of the main product, Edaravone, directly from the reaction mixture. This "reaction-crystallization" synergy ensures that the product is removed from the liquid phase as soon as it reaches saturation, further driving the reaction forward and minimizing the residence time of the product in potentially degrading conditions. The visual representation of this transformation highlights the elegant simplicity of the molecular conversion, where the hydrazine and beta-keto ester functionalities cyclize to form the stable pyrazolone core.
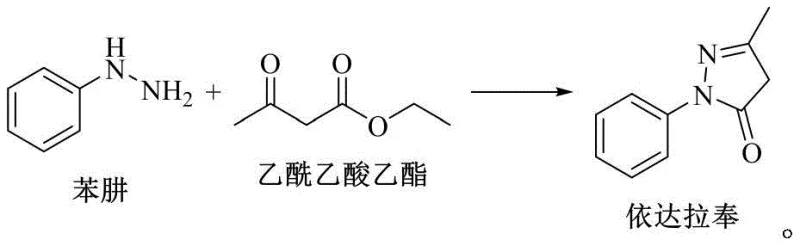
Mechanistic Insights into Reactive Distillation and Cyclization
The core mechanistic advantage of this process lies in the precise manipulation of vapor-liquid equilibrium within the rectifying tower, which serves as an integrated separation unit for the condensation reaction. As phenylhydrazine and ethyl acetoacetate react within the kettle, the generated ethanol and water form a low-boiling azeotrope or mixture that is readily vaporized and transported up the rectifying column. By initially maintaining a full reflux state, the system ensures that the reaction mixture reaches the optimal thermal steady state before product removal begins, stabilizing the kinetic profile of the cyclization. Once the reaction is established, adjusting the reflux ratio allows for the selective discharge of the light components (ethanol and water) while retaining the heavier reactants and product in the liquid phase. This selective volatility management is critical for maintaining high reactant concentrations in the kettle, thereby maximizing the collision frequency between phenylhydrazine and ethyl acetoacetate molecules. The continuous nature of this mechanism ensures that the reaction environment remains chemically "fresh," devoid of the accumulating impurities that typically plague batch reactors, resulting in a cleaner crude profile that simplifies subsequent recrystallization steps.
Furthermore, the impurity control mechanism is enhanced by the continuous crystallization step, which acts as a physical barrier to impurity incorporation. As the reaction liquid flows into the cooling crystallizer, the solubility of Edaravone decreases, prompting nucleation and crystal growth. Because the system is continuously removing the solid product via centrifugation, there is minimal opportunity for side reactions or degradation products to accumulate in the mother liquor to levels that would contaminate the final cake. The recycling loop, where the filtrate containing unreacted phenylhydrazine and ethyl acetoacetate is returned to the reactor, ensures that any partially reacted species are given multiple passes through the reaction zone. This recursive exposure to the catalytic thermal environment guarantees that the overall conversion efficiency is maximized, effectively closing the mass balance loop and minimizing waste generation. For quality control teams, this translates to a more predictable impurity spectrum, facilitating easier validation and regulatory filing for the final drug substance.
How to Synthesize Edaravone Efficiently
The implementation of this continuous synthesis route requires careful calibration of flow rates and thermal gradients to ensure optimal performance. The process begins with the precise metering of phenylhydrazine and ethyl acetoacetate into the reaction kettle, where the molar ratio can be tuned between 1:1 and 1:12 depending on the desired throughput and conversion targets. Detailed standardized operating procedures regarding pump speeds, reflux ratios, and crystallization temperatures are essential for replicating the high yields observed in the patent examples. Operators must monitor the top temperature of the rectifying tower closely to ensure that only the intended byproducts are being distilled off, preventing the loss of valuable starting materials. The following guide outlines the critical operational parameters derived from the patent data to assist process engineers in scaling this technology.
- Feed phenylhydrazine and ethyl acetoacetate into a reaction kettle connected to a rectifying tower, maintaining full reflux initially.
- Adjust the reflux ratio to continuously discharge ethanol and water byproducts while maintaining reaction temperature between 50-80°C.
- Transfer the reaction mixture to a cooling crystallizer for continuous separation, followed by centrifugal filtration and recycling of unreacted materials.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition from batch to continuous processing offers profound strategic advantages that extend far beyond simple yield improvements. The elimination of batch-to-batch variability reduces the risk of production delays caused by out-of-specification results, ensuring a more reliable flow of materials to downstream formulation units. Additionally, the continuous nature of the plant allows for a smaller physical footprint to achieve the same annual output compared to large batch reactors, optimizing capital expenditure and facility utilization rates. The ability to recycle unreacted raw materials directly back into the process significantly lowers the effective consumption of key starting materials, providing a buffer against market volatility in raw material pricing. This closed-loop system not only enhances sustainability metrics but also insulates the manufacturing cost structure from fluctuations in the supply of phenylhydrazine and ethyl acetoacetate.
- Cost Reduction in Manufacturing: The integration of reactive distillation eliminates the need for separate, energy-intensive dehydration steps that are common in batch processing, leading to substantial reductions in utility costs. By continuously removing byproducts, the reaction achieves higher conversion per pass, which means less raw material is wasted in the mother liquor, directly lowering the variable cost per kilogram of API produced. The recycling of unreacted feedstocks further amplifies these savings, as the effective yield approaches theoretical maximums without requiring expensive stoichiometric excesses. Consequently, the overall cost of goods sold is significantly optimized, making the final product more competitive in the global generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Continuous manufacturing inherently provides a steady stream of product, removing the "feast or famine" cycle associated with batch campaigns that can disrupt inventory planning. This consistency allows for leaner inventory strategies, reducing the working capital tied up in safety stock while ensuring that customer orders can be fulfilled with shorter lead times. The robustness of the continuous process against minor perturbations ensures that supply continuity is maintained even during extended production runs, mitigating the risk of stockouts that could impact downstream drug product availability. Furthermore, the simplified workflow reduces the manpower required for operational oversight, allowing skilled personnel to focus on quality assurance rather than manual batch handling.
- Scalability and Environmental Compliance: Scaling a continuous process is often more linear and predictable than scaling batch reactions, as it involves running the equipment for longer durations rather than increasing vessel sizes exponentially. This modularity facilitates easier capacity expansion to meet growing market demand for neuroprotective therapies without massive infrastructure overhauls. From an environmental perspective, the efficient use of raw materials and the reduction of solvent waste align with green chemistry principles, simplifying compliance with increasingly stringent environmental regulations. The minimized waste stream reduces the burden on effluent treatment plants, lowering disposal costs and enhancing the corporate sustainability profile of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous synthesis of Edaravone, based on the specific methodologies disclosed in the patent literature. Understanding these nuances is critical for partners evaluating the feasibility of adopting this advanced manufacturing technology. The answers provided reflect the operational realities and chemical principles governing the reactive distillation process.
Q: How does the continuous reactive distillation method improve Edaravone yield compared to batch processes?
A: The continuous method effectively removes ethanol and water byproducts in real-time via a rectifying tower, shifting the chemical equilibrium towards product formation and preventing the reverse reaction that limits conversion in traditional batch systems.
Q: What are the optimal reaction conditions for this continuous synthesis route?
A: According to the patent data, the reaction operates efficiently at temperatures between 50-80°C with a molar ratio of phenylhydrazine to ethyl acetoacetate ranging from 1:1 to 1:12, utilizing a variable reflux ratio to optimize byproduct removal.
Q: Can unreacted raw materials be recovered in this process?
A: Yes, the process includes a critical recycling step where the filtrate containing unreacted phenylhydrazine and ethyl acetoacetate is fed back into the reaction kettle, significantly reducing raw material waste and improving overall atom economy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Edaravone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced continuous manufacturing technologies like the one described in CN110922360B is key to securing a competitive edge in the pharmaceutical intermediate market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full industrial output is seamless and compliant. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical methods to verify that every batch of Edaravone meets the highest international standards. Our infrastructure is designed to support complex continuous processes, allowing us to deliver high-purity intermediates with the consistency and reliability that global supply chains demand.
We invite you to collaborate with us to leverage these technological advancements for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our continuous manufacturing capabilities can optimize your supply chain and reduce your overall procurement costs.
