Advanced Synthesis of Chiral Pyrrolidone Intermediates for Commercial Herbicide Production
Advanced Synthesis of Chiral Pyrrolidone Intermediates for Commercial Herbicide Production
The global agrochemical industry continuously demands more efficient and stereoselective pathways for producing high-performance herbicides. Patent CN110691516A introduces a robust methodology for synthesizing specific pyrrolidone derivatives, which serve as critical intermediates in the manufacture of next-generation crop protection agents. This technical disclosure outlines a series of novel chemical transformations that significantly enhance the control over stereochemistry while utilizing readily available starting materials. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for securing a reliable agrochemical intermediate supplier capable of delivering high-purity compounds at scale. The described processes leverage asymmetric catalysis and strategic functional group manipulations to construct the complex pyrrolidone core with exceptional precision.
Furthermore, the intellectual property detailed in this document addresses long-standing challenges in the synthesis of 3-substituted pyrrolidin-2-ones, particularly regarding the establishment of multiple chiral centers. By integrating steps such as nucleophilic substitution with dialkyl malonates followed by reductive cyclization, the inventors have created a versatile platform. This platform not only improves overall yields but also simplifies the purification protocols required to meet stringent regulatory standards for agricultural chemicals. As we delve deeper into the technical specifics, it becomes evident that this approach offers substantial advantages over conventional racemic syntheses, positioning it as a preferred route for commercial scale-up of complex agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing substituted pyrrolidone scaffolds often rely on non-stereoselective cyclizations that produce racemic mixtures, necessitating costly and inefficient resolution steps later in the process. These conventional methods frequently employ harsh reaction conditions or expensive chiral auxiliaries that are difficult to recover and recycle, leading to increased waste generation and higher production costs. Moreover, achieving high diastereoselectivity in the formation of the 3,4-disubstituted pyrrolidone ring has historically been a significant bottleneck, limiting the ability to produce the biologically active isomer in sufficient quantities. The reliance on multi-step sequences with poor atom economy further exacerbates the environmental footprint and reduces the overall feasibility of these older methodologies for large-scale manufacturing.
The Novel Approach
In contrast, the methodology disclosed in CN110691516A utilizes a convergent strategy that builds complexity early in the synthesis through the use of chiral epoxides or asymmetric reduction of ketones. This novel approach allows for the precise installation of stereocenters before the ring-closing step, ensuring that the final pyrrolidone product possesses the desired configuration with high enantiomeric excess. By employing catalytic hydrogenation for the cyclization step, the process eliminates the need for stoichiometric amounts of reducing agents, thereby drastically simplifying the workup procedure. This shift from resolution-based strategies to asymmetric synthesis represents a paradigm shift in cost reduction in herbicide manufacturing, offering a cleaner, more direct path to the target molecules while maintaining rigorous control over impurity profiles.
Mechanistic Insights into Reductive Cyclization and Malonate Substitution
The core innovation of this patent lies in the strategic combination of nucleophilic substitution and catalytic hydrogenation to form the pyrrolidone ring. Specifically, the reaction of a chloro-amine derivative (Formula V) with a dialkyl malonate creates a linear precursor (Formula IV) with high fidelity. This substitution reaction is typically conducted in polar aprotic solvents such as acetonitrile or DMF, using mild inorganic bases like potassium carbonate to facilitate the displacement of the chloride ion without causing elimination side reactions. The resulting malonate intermediate retains the stereochemical integrity established in previous steps, serving as a crucial linchpin for the subsequent ring closure.
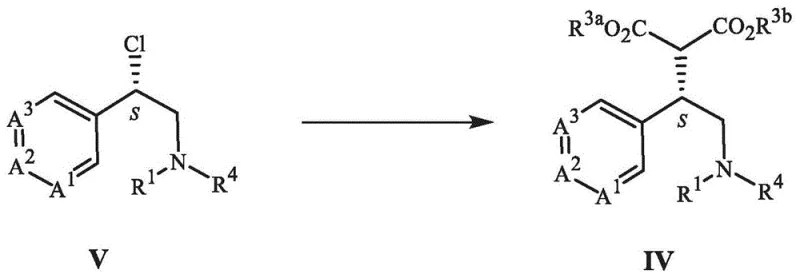
Following the formation of the malonate intermediate, the process undergoes a remarkable reductive cyclization. In this step, the benzyl protecting group is removed via catalytic hydrogenation, simultaneously triggering an intramolecular amidation that closes the five-membered ring. The use of palladium on carbon as a heterogeneous catalyst ensures that the reaction proceeds smoothly under moderate hydrogen pressure. This dual-function transformation—deprotection and cyclization—exemplifies process efficiency, as it combines two distinct chemical events into a single operational unit. The mechanism involves the initial reduction of the benzyl amine to a secondary amine, which then attacks the adjacent ester carbonyl, expelling the alkoxide to form the stable lactam structure found in Formula II-B.
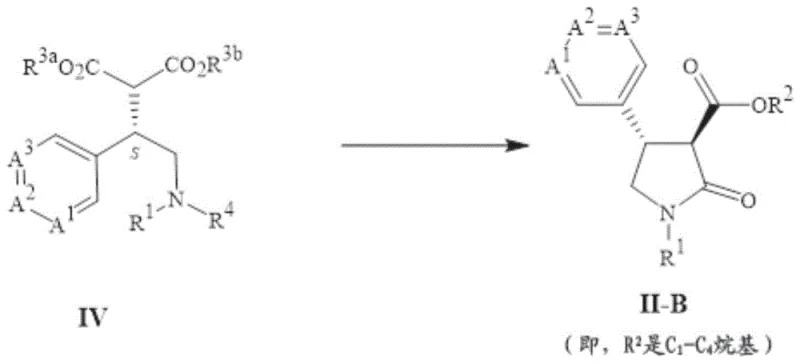
Controlling impurities during these mechanistic steps is paramount for meeting the quality specifications required by agrochemical regulators. The patent describes specific crystallization protocols, such as using methanol-water mixtures or hexane slurries, to purify the intermediates effectively. By optimizing the stoichiometry of the malonate reagent and the loading of the palladium catalyst, manufacturers can minimize the formation of over-reduced byproducts or open-chain hydrolysis products. This level of mechanistic understanding allows process chemists to fine-tune reaction parameters, ensuring that the final active pharmaceutical or agrochemical ingredient meets the necessary purity thresholds without requiring extensive chromatographic purification, which is often impractical on a multi-ton scale.
How to Synthesize Chiral Pyrrolidone Intermediates Efficiently
Implementing this synthesis route requires careful attention to reaction conditions and reagent quality to maximize yield and stereochemical purity. The process begins with the preparation of the chiral amino-alcohol or epoxide precursors, which set the absolute configuration for the entire molecule. Operators must maintain strict temperature control during the malonate substitution phase to prevent racemization, followed by a controlled hydrogenation environment to ensure complete cyclization. The detailed standardized synthesis steps provided in the patent documentation offer a clear roadmap for translating laboratory-scale success into commercial production, highlighting critical parameters such as solvent choice, catalyst loading, and workup procedures.
- React formula V chloro-compounds with dialkyl malonates in the presence of a base to form formula IV intermediates.
- Perform catalytic hydrogenation on formula IV compounds using palladium on carbon to induce reductive cyclization into formula II-B esters.
- Couple the resulting acid or ester intermediates with substituted anilines to finalize the herbicide active ingredient precursors.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers significant strategic benefits beyond mere chemical elegance. The reliance on commodity chemicals such as dimethyl malonate, benzyl amines, and standard hydrogenation catalysts ensures a stable and resilient supply chain, reducing the risk of disruptions associated with specialized or proprietary reagents. Furthermore, the elimination of resolution steps and the use of catalytic rather than stoichiometric chiral sources lead to substantial cost savings in herbicide manufacturing. By improving the overall atom economy and reducing the number of isolation steps, the process minimizes solvent consumption and waste disposal costs, aligning with modern sustainability goals and environmental compliance regulations.
- Cost Reduction in Manufacturing: The integration of deprotection and cyclization into a single catalytic step significantly lowers operational expenses by reducing reactor time and utility consumption. Additionally, the avoidance of expensive chiral resolving agents or enzymatic resolutions removes a major cost driver from the bill of materials. This streamlined approach allows for a more competitive pricing structure for the final intermediates, providing a distinct economic advantage in the global agrochemical market where margin pressure is constant.
- Enhanced Supply Chain Reliability: Since the key reagents involved in this synthesis are widely produced bulk chemicals, sourcing is straightforward and less susceptible to geopolitical or logistical bottlenecks. The robustness of the hydrogenation step, which uses standard industrial equipment, means that production can be easily scaled across multiple manufacturing sites without requiring specialized infrastructure. This flexibility ensures consistent supply continuity for downstream formulation partners, mitigating the risk of stockouts during peak agricultural seasons.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing reaction conditions that are safe and manageable in large-volume reactors. The reduction in solvent usage and the generation of less hazardous waste streams simplify the environmental permitting process and lower the cost of waste treatment. This alignment with green chemistry principles not only enhances the corporate sustainability profile but also future-proofs the manufacturing process against increasingly stringent environmental regulations governing agrochemical production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent, providing clarity on the practical aspects of producing these high-value intermediates. Understanding these details helps stakeholders make informed decisions about technology transfer and vendor qualification.
Q: How does this patent address stereochemical purity in pyrrolidone synthesis?
A: The patent utilizes asymmetric reduction techniques and specific chiral starting materials to achieve high enantiomeric excess, often exceeding 90% e.e., which is critical for biological activity in herbicides.
Q: What are the scalability advantages of the reductive cyclization step?
A: The process employs standard catalytic hydrogenation conditions using palladium on carbon, a widely available and scalable technology that avoids the need for expensive stoichiometric chiral reagents.
Q: Can this synthesis route be adapted for different substituted aryl groups?
A: Yes, the methodology supports various substitutions on the aryl ring (A1, A2, A3 variables), allowing for the production of a diverse library of analogues for structure-activity relationship studies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolidone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to bring complex synthetic routes like those described in CN110691516A to life. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We understand the critical nature of stereochemical purity in agrochemical actives and operate stringent purity specifications enforced by our rigorous QC labs to guarantee that every batch meets your exact requirements. Our commitment to quality assurance means that you can rely on us as a trusted partner for your most demanding synthesis projects.
We invite you to contact our technical procurement team to discuss how we can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how optimizing this synthesis route can improve your bottom line. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-quality pyrrolidone intermediates efficiently. Let us collaborate to drive innovation and efficiency in your agrochemical supply chain.
