Advanced Palladium-Catalyzed Synthesis of (Iso)chroman Amides for Scalable Pharmaceutical Manufacturing
Advanced Palladium-Catalyzed Synthesis of (Iso)chroman Amides for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks efficient, atom-economical pathways to construct complex heterocyclic scaffolds, particularly those containing amide linkages which are ubiquitous in bioactive molecules. Patent CN114539198A discloses a groundbreaking preparation method for amide compounds containing (iso)chroman structures, addressing critical bottlenecks in modern organic synthesis. This technology leverages a sophisticated palladium-catalyzed cyclocarbopalladation and aminocarbonylation cascade, utilizing nitroarenes as a sustainable nitrogen source and molybdenum carbonyl as a dual-purpose carbonyl source and reducing agent. By operating under relatively mild thermal conditions of 110 to 130 °C, this process achieves high reaction efficiency and exceptional functional group tolerance. For R&D directors and procurement specialists, this represents a significant leap forward in accessing high-purity pharmaceutical intermediates with reduced operational complexity and enhanced safety profiles compared to traditional high-pressure carbonylation techniques.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of amide bonds relies heavily on the acylation of amines with carboxylic acids or their activated derivatives, such as acid chlorides or anhydrides. While effective for simple systems, these classical methods often suffer from poor atom economy, generating stoichiometric amounts of waste salts and requiring harsh activation reagents that can be incompatible with sensitive functional groups. Furthermore, transition metal-catalyzed carbonylation of halogenated aryl compounds with amines, although more direct, typically necessitates the use of toxic carbon monoxide gas under high pressure, posing significant safety hazards and requiring specialized, expensive reactor infrastructure. The reliance on pre-synthesized amines also adds steps to the supply chain, as amines can be unstable or costly to produce and store. These limitations collectively increase the cost of goods sold (COGS) and extend the lead time for developing new drug candidates, creating a pressing need for more streamlined synthetic strategies in the manufacturing of complex heterocyclic intermediates.
The Novel Approach
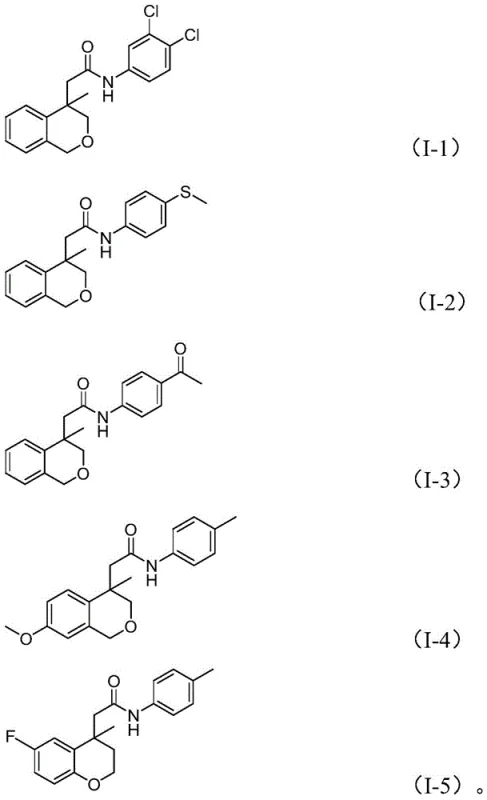
The methodology described in CN114539198A offers a transformative solution by integrating an intramolecular Heck reaction with a subsequent cross-coupling aminocarbonylation in a single operational sequence. This tandem process starts from readily available iodoarene compounds and nitroarenes, bypassing the need for unstable amines or high-pressure CO gas cylinders. Instead, solid molybdenum hexacarbonyl serves as a safe, controllable source of carbon monoxide while simultaneously acting as a reducing agent to convert the nitro group into the requisite amine functionality in situ. The reaction demonstrates remarkable versatility, accommodating a wide range of substituents including halogens, alkoxy groups, and trifluoromethyl groups on the aromatic rings. As illustrated in the specific examples below, this route successfully generates diverse (iso)chroman amide derivatives with high structural fidelity.
Mechanistic Insights into Pd-Catalyzed Cyclocarbopalladation/Aminocarbonylation
The core of this innovation lies in the intricate catalytic cycle driven by the palladium/Xantphos system. The reaction initiates with the oxidative addition of the aryl iodide to the active Pd(0) species, followed by an intramolecular migratory insertion into the pendant alkene moiety. This key step forms a crucial sigma-alkyl-palladium intermediate, effectively constructing the fused (iso)chroman ring system. Subsequently, carbon monoxide, released from the decomposition of molybdenum carbonyl, inserts into the palladium-carbon bond to generate an acyl-palladium complex. Concurrently, the nitroarene undergoes reduction, likely facilitated by the molybdenum species and water present in the system, to form the corresponding aniline or hydroxylamine intermediate which then acts as the nucleophile. The final reductive elimination releases the desired amide product and regenerates the palladium catalyst, closing the loop. This elegant mechanism ensures that the carbon-carbon bond formation and the amide bond construction occur seamlessly in one pot.
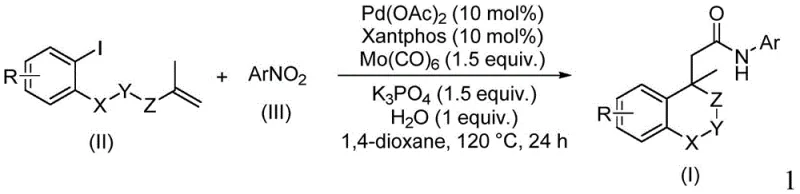
From an impurity control perspective, this mechanism offers distinct advantages over stepwise syntheses. By avoiding the isolation of reactive intermediates, the potential for side reactions such as hydrolysis of acid chlorides or oxidation of sensitive amines is significantly minimized. The use of Xantphos, a bidentate ligand with a large bite angle, stabilizes the palladium center and promotes the reductive elimination step, thereby suppressing beta-hydride elimination pathways that could lead to olefinic byproducts. Furthermore, the specific choice of potassium phosphate as a base and the controlled addition of water are critical for facilitating the reduction of the nitro group without deactivating the catalyst. This precise balance of reagents ensures a clean reaction profile, simplifying downstream purification and resulting in a final product with a superior impurity profile, which is paramount for meeting the stringent quality standards required for reliable pharmaceutical intermediate suppliers.
How to Synthesize (Iso)chroman Amides Efficiently
To implement this robust synthetic route in a laboratory or pilot plant setting, operators must adhere to specific protocol parameters to maximize yield and reproducibility. The process involves charging a reaction vessel with the iodoarene substrate, the nitroarene coupling partner, and the catalytic system comprising palladium acetate and the Xantphos ligand. Crucially, the stoichiometry of molybdenum carbonyl must be maintained at approximately 1.5 equivalents to ensure sufficient CO generation and reducing power throughout the 24-hour reaction window. The detailed standardized synthesis steps, including precise mixing orders and workup procedures, are outlined in the guide below to ensure consistent batch-to-batch quality.
- Combine palladium acetate, Xantphos ligand, molybdenum carbonyl, potassium phosphate, water, iodoarene, and nitroarene in 1,4-dioxane.
- Heat the reaction mixture to 120 °C and maintain stirring for 24 hours to ensure complete conversion.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final amide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits beyond mere chemical novelty. The shift from traditional amine-based coupling to nitroarene-based carbonylation fundamentally alters the cost structure and risk profile of the supply chain. By utilizing nitroaromatics, which are commodity chemicals produced on a massive scale for dyes and agrochemicals, manufacturers can secure a more stable and cost-effective raw material base compared to specialized amines. Moreover, the elimination of high-pressure carbon monoxide gas removes a significant safety liability and reduces the capital expenditure required for specialized high-pressure autoclaves, allowing for production in standard glass-lined reactors. This flexibility enhances supply chain resilience and reduces the lead time for high-purity pharmaceutical intermediates by simplifying the logistical requirements for hazardous gas transport and storage.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the substitution of expensive reagents with commodity chemicals. Nitroarenes are generally significantly cheaper and more shelf-stable than their corresponding amines, which often require cold storage and careful handling to prevent degradation. Additionally, the use of solid molybdenum carbonyl as a CO surrogate eliminates the need for complex gas handling infrastructure and the associated regulatory compliance costs. The high atom economy of the tandem reaction means less waste is generated per kilogram of product, reducing disposal costs and improving the overall green chemistry metrics of the manufacturing process. These factors combine to deliver substantial cost savings in API manufacturing without compromising on the quality or purity of the final intermediate.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the scarcity of specialized building blocks. This method mitigates such risks by relying on widely available starting materials like iodoarenes and nitrobenzenes, which are produced by multiple global vendors. The robustness of the catalytic system, which tolerates a wide range of functional groups including halogens and electron-withdrawing groups, means that a single platform technology can be applied to synthesize a diverse library of analogues. This versatility allows manufacturers to respond rapidly to changing demand patterns or design modifications during drug development. Furthermore, the simplified workup procedure involving filtration and standard chromatography ensures that production bottlenecks associated with complex extractions or crystallizations are avoided, ensuring a steady flow of materials to downstream processing units.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but this protocol is designed with scalability in mind. The reaction operates at moderate temperatures (120 °C) and does not require cryogenic conditions or extreme pressures, making it compatible with existing multipurpose manufacturing facilities. From an environmental standpoint, the avoidance of stoichiometric coupling reagents like thionyl chloride or carbodiimides drastically reduces the generation of hazardous waste streams. The use of water as a co-reagent in the reduction step further aligns with green chemistry principles. Consequently, scaling this process from gram to ton quantities involves straightforward linear expansion rather than complex re-engineering, facilitating the commercial scale-up of complex polymer additives or pharmaceutical intermediates with minimal environmental impact and regulatory friction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the operational feasibility and strategic value of the technology for potential partners and licensees.
Q: What is the nitrogen source in this novel synthesis method?
A: Unlike traditional methods that rely on amines, this process utilizes nitroarenes as the nitrogen source, which are abundant, stable, and cost-effective raw materials.
Q: How does this method improve upon conventional amide synthesis?
A: This approach combines intramolecular Heck cyclization with aminocarbonylation in a single pot, eliminating the need for pre-functionalized carboxylic acids and offering superior atom economy.
Q: Is this process suitable for large-scale production?
A: Yes, the reaction uses commercially available catalysts and simple workup procedures like filtration and chromatography, making it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (Iso)chroman Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has thoroughly evaluated the palladium-catalyzed pathway described in CN114539198A and confirmed its potential for delivering high-value heterocyclic scaffolds. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop optimization to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of (iso)chroman amide intermediate meets the exacting standards required for clinical and commercial applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this route can optimize your budget. We encourage you to contact us today to request specific COA data for similar structures and comprehensive route feasibility assessments. Let us help you secure a competitive advantage through superior chemistry and reliable supply chain execution.
