Advanced Synthesis of Substituted Phenoxyalkylamine Triazole Antifungal Intermediates for Commercial Scale
Advanced Synthesis of Substituted Phenoxyalkylamine Triazole Antifungal Intermediates for Commercial Scale
The pharmaceutical industry continuously seeks novel chemical entities to combat the rising threat of resistant fungal infections, particularly in immunocompromised patient populations. Patent CN101402613B introduces a significant advancement in this domain by disclosing a series of substituted phenoxyalkylamine triazole alcohol compounds. These molecules represent a strategic evolution in azole chemistry, designed to overcome the limitations of current clinical antifungals. The core structure, 1-(1H-1,2,4-triazol-1-yl)-2-(2,4-difluorophenyl)-3-(N-substituted phenoxyalkylamino)-2-propanol, offers a robust scaffold for developing next-generation therapeutics. For R&D directors and procurement specialists, understanding the synthetic accessibility and structural versatility of this patent is crucial for securing a reliable antifungal intermediate supplier. The following analysis details the chemical innovation and its commercial implications for the global supply chain.
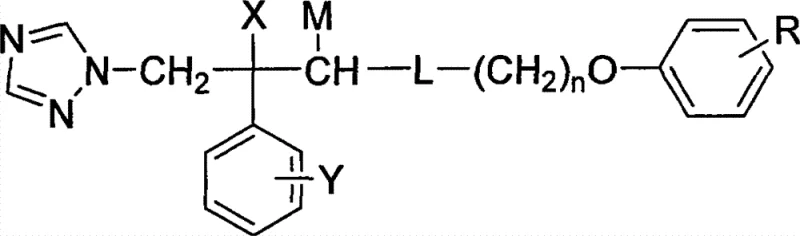
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional antifungal therapies, primarily based on early-generation azoles and allylamines, face increasing challenges regarding efficacy and safety profiles. Many existing drugs exhibit narrow antifungal spectra, rendering them ineffective against emerging resistant strains of Candida and Aspergillus species. Furthermore, the metabolic stability of conventional azoles often leads to significant drug-drug interactions and hepatotoxicity, complicating long-term treatment regimens for chronic infections. From a manufacturing perspective, older synthetic routes frequently rely on harsh reaction conditions or expensive chiral catalysts that drive up the cost of goods. These factors collectively create a bottleneck in the production of high-purity pharmaceutical intermediates, limiting the ability of supply chain heads to ensure consistent availability. The need for a chemical scaffold that balances potent biological activity with synthetic feasibility has never been more critical for cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
The methodology outlined in the patent data presents a divergent synthetic strategy that addresses these historical pain points through structural modification. By introducing a specific N-substituted phenoxyalkylamine side chain to the triazole core, the new compounds demonstrate enhanced lipophilicity and membrane permeability, which are key drivers for improved bioavailability. The synthetic route avoids complex enzymatic resolutions in the initial stages, favoring robust chemical transformations that are easier to control on a large scale. This approach not only simplifies the purification process but also significantly reduces the generation of hazardous waste, aligning with modern environmental compliance standards. For procurement managers, this translates to a more stable supply chain with reduced risk of production delays caused by complex processing steps. The ability to generate various salts further enhances the commercial viability, allowing formulators to select the optimal physicochemical properties for the final drug product.
Mechanistic Insights into Triazole-Catalyzed Antifungal Activity
The biological efficacy of these compounds is rooted in their ability to inhibit lanosterol 14α-demethylase, a critical enzyme in fungal ergosterol biosynthesis. The 2,4-difluorophenyl moiety plays a pivotal role in binding affinity, creating strong hydrophobic interactions within the enzyme's active site. The triazole ring coordinates with the heme iron of the cytochrome P450 enzyme, effectively blocking the conversion of lanosterol to ergosterol. This mechanism disrupts fungal cell membrane integrity, leading to cell death. The introduction of the phenoxyalkylamine tail allows for fine-tuning of the molecule's electronic properties, potentially reducing affinity for human cytochrome P450 enzymes and thereby lowering toxicity. For R&D teams, this structural insight provides a clear path for structure-activity relationship (SAR) studies to optimize potency further. Understanding these mechanistic details is essential for predicting metabolic stability and ensuring the development of safe, effective antifungal agents.
Impurity control is another critical aspect of the mechanistic profile, particularly concerning the epoxidation and ring-opening steps. The patent describes specific conditions for the formation of the epoxide intermediate using trimethylsulfur iodide, which minimizes side reactions that could lead to difficult-to-remove byproducts. The subsequent nucleophilic attack by the amine is conducted under controlled basic conditions to ensure regioselectivity, favoring the formation of the desired secondary alcohol over potential elimination products. This level of control is vital for maintaining high-purity triazole derivatives, as impurities can significantly impact the safety profile of the final API. By optimizing reaction parameters such as temperature and stoichiometry, manufacturers can achieve consistent quality batches. This rigorous approach to impurity management ensures that the commercial scale-up of complex antifungal agents meets stringent regulatory requirements without compromising yield.
How to Synthesize Substituted Phenoxyalkylamine Triazole Alcohol Efficiently
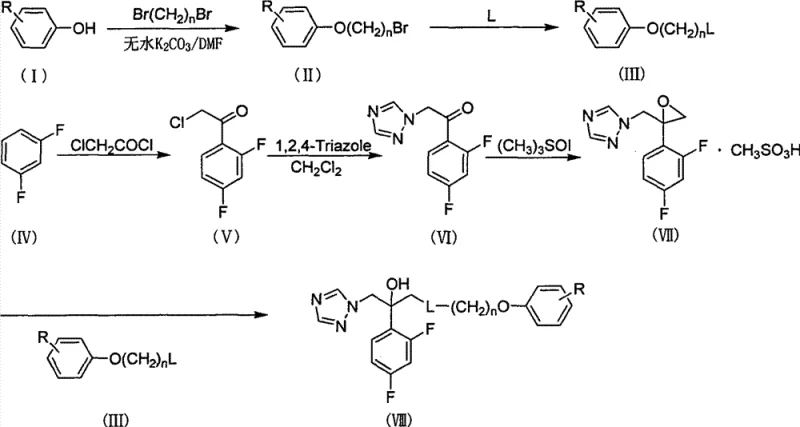
The synthesis of these high-value intermediates follows a logical sequence of organic transformations that are well-suited for industrial application. The process begins with the preparation of the phenoxyalkylamine fragment, followed by the construction of the triazole-containing ketone, and concludes with the coupling of these two key intermediates. Each step has been optimized to maximize yield and minimize purification burden, making the route attractive for large-scale production. The use of common solvents like ethanol and dichloromethane facilitates solvent recovery and recycling, contributing to overall process sustainability. Detailed knowledge of these steps allows technical teams to assess feasibility and identify potential scale-up risks early in the development phase. The following guide outlines the standardized synthesis steps derived from the patent documentation.
- Preparation of bromoalkyl substituted phenol ether via reaction of substituted phenol with dibromoalkane in the presence of anhydrous potassium carbonate.
- Synthesis of 2-chloro-2',4'-difluoroacetophenone through Friedel-Crafts reaction of m-difluorobenzene with chloroacetyl chloride using anhydrous aluminum trichloride.
- Final coupling of the epoxide intermediate with N-substituted phenoxyalkylamine in ethanol under reflux conditions to yield the target triazole alcohol compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible benefits beyond mere chemical novelty. The streamlined process reduces the number of isolation steps required, which directly correlates to lower operational costs and shorter manufacturing cycles. By utilizing readily available starting materials such as substituted phenols and difluorobenzene, the supply chain becomes less vulnerable to raw material shortages. This resilience is crucial for maintaining continuous production schedules and meeting market demand without significant lead time fluctuations. Furthermore, the robustness of the chemistry allows for flexible manufacturing across different facilities, enhancing supply security. These factors collectively contribute to substantial cost savings and improved reliability for partners seeking a reliable antifungal intermediate supplier.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metal catalysts often required in cross-coupling reactions, replacing them with cost-effective reagents like potassium carbonate and triethylamine. This substitution drastically simplifies the downstream processing, as there is no need for specialized heavy metal scavenging steps to meet regulatory limits. The reduction in purification complexity leads to higher overall throughput and lower utility consumption per kilogram of product. Consequently, this efficiency drives down the cost of goods sold, allowing for more competitive pricing strategies in the global market. The economic advantage is further amplified by the high yields reported in the patent examples, ensuring minimal material waste during production.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals for the synthesis ensures that raw material sourcing is not a bottleneck. Unlike routes dependent on specialized chiral building blocks that may have limited suppliers, this method uses bulk chemicals available from multiple vendors worldwide. This diversification of the supply base mitigates the risk of disruption due to geopolitical issues or single-source failures. Additionally, the stability of the intermediates allows for stockpiling if necessary, providing a buffer against demand spikes. For supply chain heads, this means reducing lead time for high-purity pharmaceutical intermediates and ensuring a steady flow of materials to downstream API manufacturers without unexpected delays.
- Scalability and Environmental Compliance: The reaction conditions described, such as reflux in ethanol or stirring at room temperature, are easily transferable from laboratory glassware to industrial reactors. There are no extreme pressure or cryogenic temperature requirements that would necessitate specialized equipment investments. Moreover, the solvent systems used are amenable to standard waste treatment protocols, reducing the environmental footprint of the manufacturing process. This alignment with green chemistry principles facilitates regulatory approval and community acceptance of manufacturing sites. The ability to scale from 100 kgs to 100 MT/annual commercial production without significant process re-engineering makes this technology a low-risk investment for long-term supply partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these triazole intermediates. The answers are derived directly from the patent specifications and general chemical engineering principles applicable to this class of compounds. Understanding these details helps stakeholders make informed decisions about integrating this technology into their development pipelines. It clarifies the scope of the intellectual property and the practical aspects of manufacturing.
Q: What distinguishes this triazole intermediate from existing antifungal drugs?
A: This novel compound class features a unique substituted phenoxyalkylamine side chain combined with a 2,4-difluorophenyl triazole core. Patent data indicates broader antifungal spectra and potentially lower toxicity profiles compared to standard azoles like fluconazole, addressing resistance issues in deep fungal infections.
Q: Is the synthesis route scalable for industrial production?
A: Yes, the described methodology utilizes standard organic transformations such as Friedel-Crafts acylation and nucleophilic substitution. The reagents are commercially available, and the reaction conditions, including temperature and solvent systems, are compatible with large-scale manufacturing equipment without requiring exotic catalysts.
Q: What forms of the compound are available for development?
A: The technology covers the free base forms of the triazole alcohol compounds as well as various pharmaceutically acceptable salts, including hydrochloride, nitrate, hydrobromide, and methanesulfonate salts, offering flexibility for formulation development and stability optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Phenoxyalkylamine Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of quality and consistency in the production of pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of triazole intermediate meets the highest international standards. Our commitment to excellence extends beyond mere synthesis; we provide comprehensive support to ensure your supply chain remains robust and uninterrupted. Partnering with us means gaining access to a wealth of chemical expertise dedicated to your success.
We invite you to contact our technical procurement team to discuss your specific requirements for antifungal intermediates. We are prepared to provide a Customized Cost-Saving Analysis tailored to your project's volume and timeline. Please reach out to request specific COA data and route feasibility assessments to see how our capabilities align with your strategic goals. Let us help you accelerate your development timeline with our reliable supply and technical support.
