Industrial Scalability and Cost Efficiency in Producing 2-Amino-3-Bromo-6-Chloropyrazine Intermediates
Industrial Scalability and Cost Efficiency in Producing 2-Amino-3-Bromo-6-Chloropyrazine Intermediates
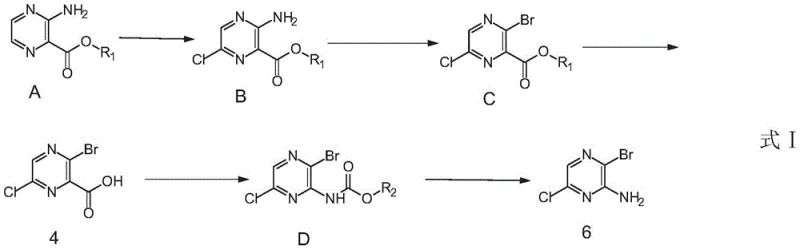
The pharmaceutical industry's relentless pursuit of potent kinase inhibitors has placed significant demand on the supply chain for specialized heterocyclic building blocks. Specifically, 2-amino-3-bromo-6-chloropyrazine serves as a critical scaffold for the synthesis of SHP2 inhibitors, a promising class of anti-tumor medicaments. However, traditional synthetic routes have long been plagued by regioselectivity issues and purification bottlenecks that hinder commercial viability. A breakthrough detailed in patent CN108101857B offers a transformative solution, outlining a scalable, five-step amplifiable process that fundamentally restructures the synthetic logic. By shifting from a direct bromination strategy to a chlorination-first approach followed by a strategic Curtius rearrangement, this methodology achieves high yields and simplifies purification to mere recrystallization. For R&D directors and procurement specialists alike, this represents a pivotal shift towards more robust and cost-effective manufacturing of complex pyrazine derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-amino-3-bromo-6-chloropyrazine relied heavily on the direct bromination of 2-amino-6-chloropyrazine. This conventional pathway is inherently flawed due to the strong activating effect of the amino group on the pyrazine ring, which directs electrophilic substitution primarily to the para position rather than the desired ortho position relative to the nitrogen. Consequently, the reaction generates substantial quantities of unwanted byproducts, specifically 2-bromo-3-chloro-5-aminopyrazine and dibromo impurities like 2-amino-3,5-dibromo-6-chloropyrazine. The isolation of the target molecule from this complex mixture is notoriously difficult, often necessitating labor-intensive column chromatography which yields a meager 25% to 31% recovery. Such low efficiency and high purification costs render this method entirely unsuitable for the multi-kilogram or ton-scale production required by modern drug development pipelines.
The Novel Approach
In stark contrast, the novel process disclosed in the patent data utilizes 3-aminopyrazine-2-carboxylate as the starting material, effectively bypassing the regioselectivity pitfalls of direct electrophilic substitution. The strategy involves an initial chlorination to install the chlorine atom, followed by a diazotization-bromination sequence to replace the original amino group with bromine. Crucially, the final amino group is introduced not by direct substitution, but via a carboxyl rearrangement (Curtius rearrangement) of the ester functionality. This ingenious inversion of synthetic steps ensures that the bromine and chlorine atoms are positioned correctly without competing side reactions. The result is a process where solid products are obtained at every stage, allowing for simple recrystallization instead of chromatography, thereby facilitating seamless industrial scale-up and dramatically lowering the cost of goods sold.
Mechanistic Insights into the Chlorination-Diazotization-Rearrangement Sequence
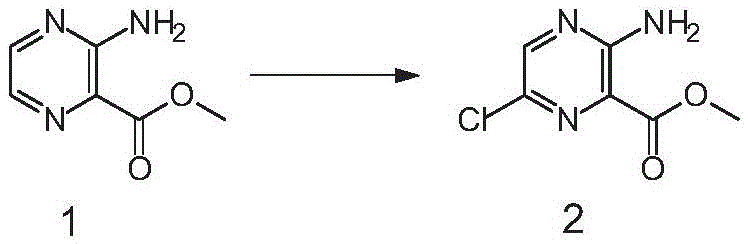
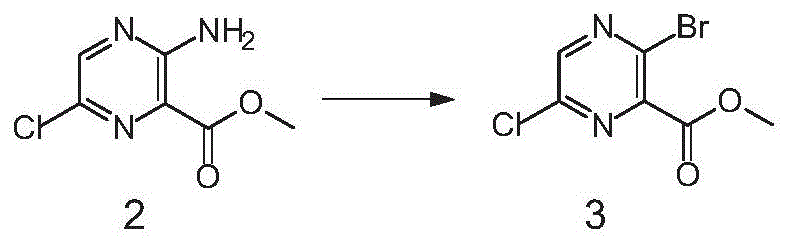
The success of this scalable route hinges on the precise control of three distinct chemical transformations: selective halogenation, diazotization, and the Curtius rearrangement. The process initiates with the chlorination of the pyrazine ester using N-chlorosuccinimide (NCS) in acetonitrile at elevated temperatures ranging from 50°C to 82°C. This step is critical as it installs the chlorine atom at the 6-position with high fidelity, leveraging the electronic properties of the ester and amino groups to direct the electrophile. Following this, the amino group is converted to a diazonium salt using sodium nitrite and hydrobromic acid at low temperatures (-20°C to 15°C), which is immediately displaced by bromide to yield the 3-bromo-6-chloro intermediate. This sequence avoids the formation of poly-halogenated byproducts common in direct bromination methods.
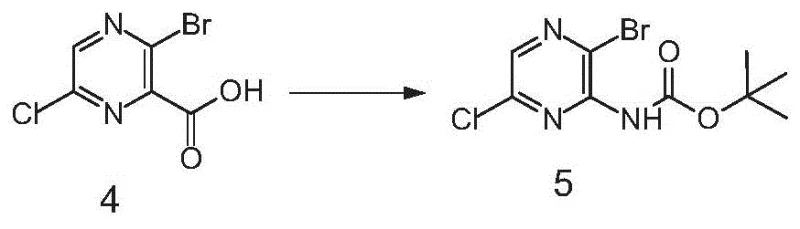
The most sophisticated aspect of this mechanism is the transformation of the ester group into the final amine functionality. After hydrolyzing the ester to the corresponding carboxylic acid using lithium hydroxide, the molecule undergoes a Curtius rearrangement mediated by diphenyl phosphorazidate (DPPA) and triethylamine. This reaction proceeds through an acyl azide intermediate which rearranges to an isocyanate, subsequently trapped by t-butanol to form a Boc-protected amine. Finally, acidic deprotection using trifluoroacetic acid reveals the free amine. This multi-step functional group interconversion guarantees that the amino group ends up exactly where needed, adjacent to the bromine, with exceptional purity profiles that meet stringent pharmaceutical standards without the need for complex downstream processing.
How to Synthesize 2-Amino-3-Bromo-6-Chloropyrazine Efficiently
Implementing this synthesis requires strict adherence to the optimized reaction parameters outlined in the patent to ensure maximum yield and safety. The process is designed to be telescoped where possible, but typically involves isolating stable solid intermediates to guarantee quality control at each stage. Operators must pay close attention to temperature controls during the exothermic diazotization step and the thermal rearrangement phase. The use of standard solvents like acetonitrile, ethyl acetate, and dichloromethane ensures compatibility with existing manufacturing infrastructure. Below is the structural framework for executing this synthesis, detailing the specific reagents and conditions required to transition from raw materials to the final high-purity intermediate.
- Chlorinate 3-aminopyrazine-2-carboxylate using N-chlorosuccinimide (NCS) in acetonitrile at elevated temperatures to introduce the chlorine atom regioselectively.
- Perform diazotization followed by bromination using sodium nitrite and hydrobromic acid at low temperatures to substitute the amino group with bromine.
- Hydrolyze the ester group to a carboxylic acid, then execute a Curtius rearrangement using diphenyl phosphorazidate (DPPA) to migrate the functionality, followed by Boc deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates directly into enhanced operational resilience and significant cost optimization. The elimination of column chromatography is perhaps the most impactful economic driver; by relying solely on crystallization and filtration, manufacturers can drastically reduce solvent consumption, silica gel waste, and labor hours associated with purification. This simplification not only lowers the variable cost per kilogram but also accelerates the batch cycle time, allowing for higher throughput in the same reactor volume. Furthermore, the ability to isolate solids at every step minimizes the risk of carrying impurities forward, reducing the likelihood of batch failures and ensuring a consistent supply of material for downstream API synthesis.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the fundamental simplification of the purification workflow. Traditional methods requiring chromatography are prohibitively expensive at scale due to the high cost of stationary phases and the vast volumes of solvents needed for elution and recovery. By replacing this with recrystallization, the process achieves substantial cost savings in both material and waste disposal. Additionally, the high overall yield, reported to be improved by more than one fold compared to prior art, means that less raw material is required to produce the same amount of final product, further driving down the unit cost of this valuable pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex processes that are sensitive to minor variations in raw material quality or reaction conditions. This new route utilizes robust, commodity-grade reagents such as N-chlorosuccinimide, sodium nitrite, and diphenyl phosphorazidate, which are readily available from multiple global suppliers. The process tolerance is widened by the formation of stable solid intermediates that can be stored or transported if necessary, decoupling the production stages. This flexibility allows manufacturers to build inventory buffers and respond more agilely to fluctuations in demand from drug developers, ensuring a reliable supply of high-purity pharmaceutical intermediates even during market volatility.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often exposes hidden inefficiencies, particularly regarding heat transfer and mixing in heterogeneous systems. This protocol is explicitly designed for amplification, utilizing homogeneous solutions for key steps and generating solids that are easily filtered. The reduction in solvent usage and the avoidance of silica waste align perfectly with modern green chemistry principles and environmental regulations. Facilities can achieve commercial scale-up of complex pyrazine derivatives with a smaller environmental footprint, reducing the burden on wastewater treatment systems and lowering the overall carbon intensity of the manufacturing operation.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into their supply chains. The following questions address common concerns regarding reaction safety, impurity profiles, and scalability. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a transparent view of the process capabilities and limitations for potential partners and technical evaluators.
Q: Why is the new chlorination-first route superior to direct bromination for this pyrazine intermediate?
A: Conventional direct bromination suffers from poor regioselectivity due to amino activation, leading to significant byproducts like 2-bromo-3-chloro-5-aminopyrazine and requiring difficult column chromatography. The new route reverses the order, installing chlorine first and using a Curtius rearrangement to position the amine, which eliminates complex purification and significantly boosts yield.
Q: What are the critical reaction conditions for the Curtius rearrangement step in this process?
A: The rearrangement requires reacting the carboxylic acid intermediate with diphenyl phosphorazidate (DPPA) and triethylamine in t-butanol. The reaction temperature must be carefully controlled between 60-85°C, preferably 77-83°C, to ensure complete conversion to the carbamate intermediate without degradation.
Q: How does this process impact the environmental footprint of large-scale manufacturing?
A: By replacing column chromatography with simple recrystallization and filtration at every step, the process drastically reduces solvent consumption and silica gel waste. Furthermore, the use of standard reagents like NCS and DPPA allows for easier waste stream management compared to heavy metal catalyzed alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-3-Bromo-6-Chloropyrazine Supplier
As the global demand for targeted oncology therapies continues to surge, the need for reliable sources of advanced heterocyclic intermediates has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep technical expertise to translate innovative patent methodologies like CN108101857B into commercial reality. Our facility is equipped with state-of-the-art reactors and rigorous QC labs capable of handling hazardous reagents such as azides and strong acids safely and efficiently. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive material that meets stringent purity specifications regardless of order volume. Our commitment to quality assurance means every batch of 2-amino-3-bromo-6-chloropyrazine is thoroughly characterized to support your regulatory filings.
We invite pharmaceutical companies and contract research organizations to collaborate with us to secure a stable supply of this critical building block. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can accelerate your drug development programs while optimizing your overall budget.
