Advanced Catalytic Bromination for High-Purity 2-Amino-3-Bromo-6-Chloropyrazine Manufacturing
The pharmaceutical industry's relentless pursuit of potent oncology therapeutics has placed significant demand on the supply chain for specialized heterocyclic intermediates, particularly those serving as core scaffolds for SHP2 inhibitors. Patent CN112724089B, published in late August 2022, introduces a robust and economically viable synthesis process for 2-amino-3-bromo-6-chloropyrazine, a critical building block for next-generation anti-tumor agents such as TNO155 and SHP099. This technical disclosure represents a substantial leap forward in process chemistry, addressing long-standing inefficiencies in yield and purification that have historically plagued the manufacturing of this specific pyrazine derivative. By shifting from traditional, low-efficiency bromination protocols to a catalytic system utilizing liquid bromine and hydrobromic acid, the inventors have established a pathway that not only enhances chemical output but also streamlines the operational complexity for commercial manufacturers. For R&D directors and procurement strategists alike, understanding the nuances of this patented methodology is essential for securing a stable, cost-effective supply of high-purity intermediates necessary for clinical and commercial drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in this patent, the synthesis of 2-amino-3-bromo-6-chloropyrazine was characterized by significant technical hurdles that negatively impacted both cost and timeline. Existing literature, including various international patent filings, described direct bromination methods that suffered from abysmally low yields, typically ranging from merely 11% to 31%. Such poor efficiency is unacceptable in modern pharmaceutical manufacturing, where material throughput directly correlates to the final cost of goods. Furthermore, these conventional routes were plagued by severe selectivity issues, frequently generating substantial quantities of the regioisomer 2-bromo-3-chloro-5-aminopyrazine as a major byproduct. The structural similarity of this impurity to the desired product made purification exceptionally difficult, often requiring extensive and wasteful chromatographic separations that further eroded overall yield. Alternative approaches, such as those starting from 3-aminopyrazine-2-carboxylic acid, introduced excessive synthetic steps, increasing the cumulative risk of yield loss and operational failure at every stage of the sequence.
The Novel Approach
The methodology disclosed in CN112724089B fundamentally reimagines this transformation by optimizing the electrophilic aromatic substitution conditions to favor the desired 3-bromo substitution pattern with high fidelity. By employing 2-amino-6-chloropyrazine as the starting material and introducing a specific catalytic system comprising HBr in acetic acid or water, the reaction achieves a dramatic improvement in conversion efficiency. The use of liquid bromine as the brominating agent, carefully dosed into a cooled solution of the substrate in chloroform, allows for precise control over the reaction kinetics. This controlled environment minimizes poly-bromination and suppresses the formation of the troublesome regioisomers that plagued earlier attempts. The result is a streamlined process that delivers the target yellow solid compound with significantly improved purity profiles and yields approaching 50%, effectively doubling the output compared to the best available prior art. 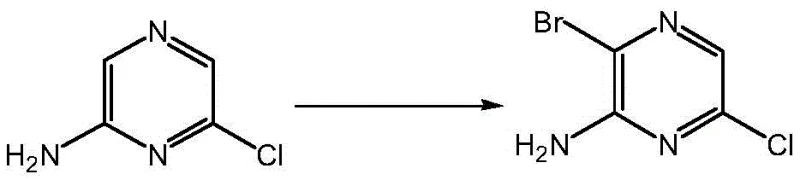
Mechanistic Insights into HBr-Catalyzed Electrophilic Bromination
The success of this novel synthesis lies in the sophisticated manipulation of the electronic properties of the pyrazine ring through acid catalysis. The pyrazine nucleus is inherently electron-deficient due to the presence of two electronegative nitrogen atoms, which generally deactivates the ring towards electrophilic attack. However, the introduction of an amino group at the 2-position activates the adjacent positions, yet achieving selective bromination at the 3-position without affecting the 5-position or the amino group requires careful tuning. The addition of HBr serves a dual purpose: it protonates the ring nitrogens to modulate electron density and potentially generates a more reactive bromonium species in situ. This catalytic activation lowers the energy barrier for the electrophilic attack specifically at the C3 position, which is ortho to the activating amino group and para to the chlorine substituent. The low temperature range of -5°C to 5°C is critical in this mechanism, as it kinetically favors the formation of the mono-brominated product while thermodynamically disfavoring side reactions or over-bromination that would occur at higher temperatures.
From an impurity control perspective, the mechanism inherently limits the generation of the 2-bromo-3-chloro-5-aminopyrazine byproduct by ensuring that the bromine source is consumed efficiently by the most activated site before alternative pathways can compete. The workup procedure, involving the quenching of excess bromine with sodium sulfite followed by extraction and dual recrystallization, is designed to exploit the solubility differences between the target molecule and any remaining minor impurities. The first recrystallization using petroleum ether and ethyl acetate removes bulk impurities, while the second recrystallization with petroleum ether and tert-butyl methyl ether polishes the crystal lattice to achieve the stringent purity specifications required for pharmaceutical intermediates. This rigorous purification strategy ensures that the final material is free from genotoxic alerts or heavy metal contaminants, a crucial consideration for regulatory compliance in drug substance manufacturing.
How to Synthesize 2-Amino-3-Bromo-6-Chloropyrazine Efficiently
The practical execution of this synthesis is designed for reproducibility and safety, utilizing standard laboratory and plant equipment. The process begins with the dissolution of the starting material in a halogenated solvent, preferably chloroform, followed by the addition of the acid catalyst under cooling. The dropwise addition of liquid bromine is the rate-determining step that requires careful monitoring to maintain the exotherm within safe limits. Following the reaction period, the mixture is treated with a reducing agent to neutralize unreacted halogen, and the product is isolated through a combination of phase separation and crystallization techniques. For a comprehensive, step-by-step guide including exact molar ratios, stirring speeds, and safety protocols, please refer to the standardized operating procedures derived from the patent examples below.
- Dissolve 2-amino-6-chloropyrazine in chloroform, cool the system to between -5°C and 5°C, and add a catalytic amount of HBr/AcOH.
- Dropwise add liquid bromine while maintaining low temperature, then allow the reaction to proceed with heat preservation until TLC indicates completion.
- Quench excess bromine with sodium sulfite, extract with chloroform, dry, concentrate, and purify via silica gel chromatography followed by dual recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates directly into tangible operational benefits and risk mitigation. The shift from low-yielding, multi-step, or impurity-laden processes to this high-efficiency catalytic route fundamentally alters the cost structure of producing 2-amino-3-bromo-6-chloropyrazine. By nearly doubling the yield compared to historical benchmarks, the amount of raw material required per kilogram of finished product is drastically reduced, leading to substantial savings in input costs. Furthermore, the simplification of the purification workflow reduces the consumption of solvents and silica gel, lowering both material expenses and waste disposal costs. This efficiency gain is not merely theoretical; it represents a robust pathway to cost reduction in pharmaceutical intermediate manufacturing that can be immediately leveraged in contract negotiations and budget planning.
- Cost Reduction in Manufacturing: The elimination of complex, multi-step sequences and the avoidance of difficult-to-separate isomers significantly lowers the overall processing time and resource consumption. By utilizing liquid bromine, a commodity chemical with stable pricing, instead of expensive specialized reagents like N-bromosuccinimide (NBS) which was used in comparative examples with poor results, the process ensures a lower bill of materials. The high yield means less starting material is wasted, and the simplified purification reduces the load on downstream processing equipment, collectively driving down the unit cost of production without compromising quality.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials like 2-amino-6-chloropyrazine and common solvents such as chloroform ensures that the supply chain is resilient to disruptions. Unlike processes dependent on exotic catalysts or custom-synthesized reagents, this method utilizes chemicals that are widely stocked by global suppliers, minimizing lead times for raw material acquisition. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry without catastrophic failure, further enhances batch-to-batch consistency, ensuring a steady flow of material to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with parameters that translate easily from gram-scale laboratory synthesis to multi-ton commercial production. The use of chloroform allows for efficient recycling and recovery, aligning with modern green chemistry initiatives to minimize solvent waste. Additionally, the straightforward workup involving sodium sulfite quenching ensures that hazardous bromine vapors are contained and neutralized effectively, meeting strict environmental health and safety standards required for large-scale chemical operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of this critical oncology intermediate. These answers are derived directly from the experimental data and technical disclosures found within the patent documentation, providing a transparent view of the technology's capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of integrating this intermediate into their own development pipelines.
Q: What is the primary advantage of this synthesis method over prior art?
A: The primary advantage is a significant increase in yield (up to 49% compared to 11-31% in previous methods) and simplified purification, avoiding the formation of difficult-to-remove isomers like 2-bromo-3-chloro-5-aminopyrazine.
Q: Why is liquid bromine preferred over NBS in this specific transformation?
A: Liquid bromine, when used with an HBr catalyst, provides superior regioselectivity for the 3-position on the pyrazine ring and avoids the generation of succinimide byproducts associated with NBS, simplifying downstream waste treatment.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes common industrial solvents like chloroform and standard reagents like liquid bromine, with mild temperature requirements (-5 to 5°C), making it highly adaptable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-3-Bromo-6-Chloropyrazine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful development of SHP2 inhibitors depends heavily on the consistent availability of high-quality intermediates like 2-amino-3-bromo-6-chloropyrazine. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your clinical and commercial needs are met without interruption. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee that every batch meets stringent purity specifications, fully compliant with the high standards demanded by the global pharmaceutical industry. We are committed to delivering not just a chemical product, but a reliable partnership that supports your drug development timeline.
We invite you to engage with our technical procurement team to discuss how our optimized manufacturing processes can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our implementation of this patented technology can reduce your overall COGS. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on real-world performance metrics and our proven track record in complex heterocycle synthesis.
