Advanced Heck Coupling Strategy for High-Purity Resveratrol and Piceatannol Manufacturing
Advanced Heck Coupling Strategy for High-Purity Resveratrol and Piceatannol Manufacturing
The pharmaceutical and nutraceutical industries continuously seek robust, scalable synthetic routes for bioactive stilbene derivatives, particularly trans-resveratrol and piceatannol. Patent CN101117317B introduces a significant technological advancement in this domain by detailing a refined Heck reaction methodology that streamlines the production of these high-value compounds. This intellectual property outlines a comprehensive process that not only enhances the stereochemical purity of the final (E)-isomers but also optimizes the synthetic pathway through the use of novel protected intermediates. By leveraging palladium-catalyzed cross-coupling strategies, this method addresses long-standing challenges regarding yield efficiency and operational simplicity that have plagued earlier synthetic attempts. For R&D directors and procurement specialists, understanding the nuances of this patent is critical for evaluating next-generation supply chains capable of delivering consistent, high-quality active pharmaceutical ingredients (APIs) and fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of resveratrol and its analogues has relied heavily on classical condensation reactions such as the Wittig, Horner-Wadsworth-Emmons, or Perkin reactions. These traditional pathways often suffer from inherent inefficiencies, including the requirement for multiple synthetic steps, the generation of significant stoichiometric waste, and difficulties in controlling the E/Z isomeric ratio. For instance, prior art references such as WO 01/60774 describe multi-step sequences starting from dihydroxybenzaldehydes that, while functional, often cap out at moderate yields around 70% and involve cumbersome purification protocols to separate geometric isomers. Furthermore, the handling of sensitive aldehyde intermediates and the use of strong bases or unstable ylides in Wittig-type reactions introduce safety hazards and scalability bottlenecks that are undesirable for modern GMP manufacturing environments. These legacy methods frequently result in complex impurity profiles that necessitate expensive chromatographic separations, thereby inflating the cost of goods and extending lead times for commercial batches.
The Novel Approach
In stark contrast, the methodology disclosed in CN101117317B utilizes a direct Heck coupling between a protected vinylbenzene derivative and a protected aryl halide, offering a more convergent and atom-economical route. This innovative approach simplifies the molecular construction by directly forging the carbon-carbon double bond under mild catalytic conditions, significantly reducing the step count compared to classical condensations. The process employs stable, protected starting materials that minimize side reactions and facilitate easier handling on a large scale. By optimizing the reaction parameters, including solvent choice and base selection, this novel technique achieves superior conversion rates and stereoselectivity, predominantly favoring the biologically active trans-isomer. The integration of this streamlined Heck reaction into the manufacturing workflow represents a paradigm shift, enabling producers to bypass the limitations of older chemistries and achieve a more reliable, cost-effective production model for stilbene derivatives.
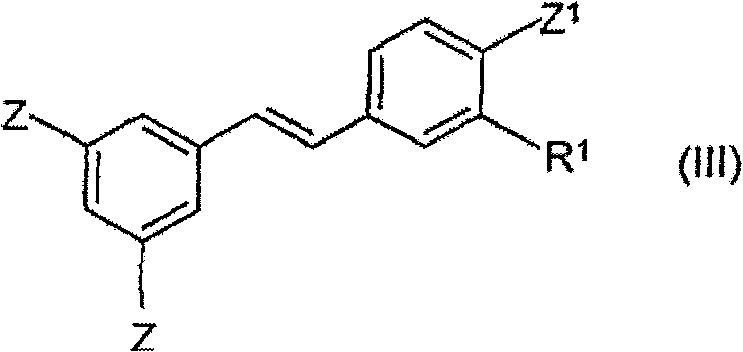
Mechanistic Insights into Pd-Catalyzed Heck Coupling
The core of this synthetic breakthrough lies in the precise orchestration of the palladium-catalyzed Heck reaction mechanism, which proceeds through a well-defined catalytic cycle involving oxidative addition, migratory insertion, and beta-hydride elimination. In this specific application, the reaction initiates with the oxidative addition of the aryl halide (Formula II) to the active Pd(0) species, forming an aryl-palladium(II) complex. This intermediate then undergoes migratory insertion with the protected vinylbenzene (Formula I), establishing the new carbon-carbon bond with high regioselectivity. The subsequent beta-hydride elimination step releases the coupled stilbene product (Formula III) and regenerates the Pd(II) species, which is reduced back to Pd(0) by the base to close the catalytic loop. The patent highlights the efficacy of various palladium sources, including PdCl2, Pd(dba)2, and specialized palladacycles, demonstrating that the choice of ligand and catalyst precursor can be tuned to maximize turnover numbers and minimize metal residue in the final product.
Beyond the coupling event, the patent details a sophisticated strategy for precursor synthesis that further enhances overall process efficiency. A key innovation involves the preparation of the vinylbenzene starting material via the reduction of a protected acetophenone derivative (Formula IV) followed by dehydration. 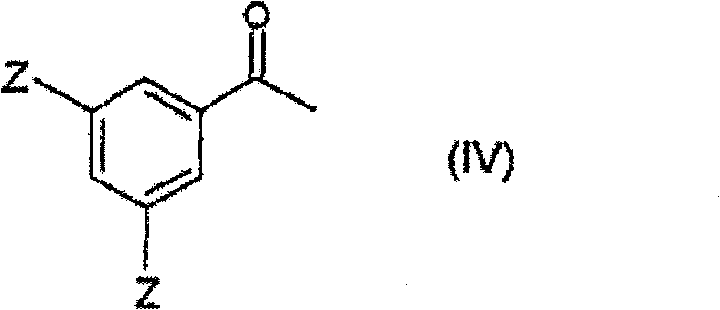 This sequence allows for the generation of the olefinic coupling partner from readily available ketones, avoiding the need for unstable aldehyde precursors often required in other routes. The reduction step, typically performed using hydrogen and a noble metal catalyst like Pd/C or Raney Nickel, proceeds with high fidelity to yield the corresponding alcohol, which is then converted to a leaving group (such as a halide or sulfonate) and eliminated to form the vinyl group. This modular approach to building block synthesis ensures a consistent supply of high-purity reactants, which is essential for maintaining the integrity of the downstream Heck coupling and minimizing the formation of difficult-to-remove impurities.
This sequence allows for the generation of the olefinic coupling partner from readily available ketones, avoiding the need for unstable aldehyde precursors often required in other routes. The reduction step, typically performed using hydrogen and a noble metal catalyst like Pd/C or Raney Nickel, proceeds with high fidelity to yield the corresponding alcohol, which is then converted to a leaving group (such as a halide or sulfonate) and eliminated to form the vinyl group. This modular approach to building block synthesis ensures a consistent supply of high-purity reactants, which is essential for maintaining the integrity of the downstream Heck coupling and minimizing the formation of difficult-to-remove impurities.
How to Synthesize Stilbene Derivatives Efficiently
Implementing this synthesis requires careful attention to reaction conditions, particularly regarding the protection of phenolic hydroxyl groups and the selection of appropriate bases and solvents. The patent emphasizes the use of electron-withdrawing protecting groups such as acetyl, benzoyl, or carbonate moieties, which stabilize the aromatic ring during the coupling process and can be easily removed in the final step via hydrolysis. The reaction is typically conducted in polar aprotic solvents like DMF or NMP, or alternatively in hydrocarbons like toluene, under an inert atmosphere to prevent catalyst deactivation. Bases such as sodium bicarbonate, potassium phosphate, or organic amines are employed to neutralize the acid byproduct and facilitate the catalytic cycle. While the general principles are straightforward, the exact optimization of temperature, pressure, and catalyst loading is critical for achieving the reported high yields and purity levels. The detailed standardized synthesis steps见下方的指南.
- Prepare protected vinylbenzene precursors via reduction of acetophenone derivatives followed by dehydration or elimination.
- Execute the Heck coupling reaction between the protected vinylbenzene and protected aryl halide using a palladium catalyst and base.
- Remove protecting groups via hydrolysis to yield the final trans-resveratrol or piceatannol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN101117317B offers tangible benefits that extend beyond mere chemical elegance. The transition to this Heck-based methodology fundamentally alters the cost structure and risk profile of producing resveratrol and piceatannol intermediates. By reducing the number of unit operations and eliminating the need for hazardous reagents associated with older condensation methods, manufacturers can realize significant operational expenditure savings. The robustness of the palladium catalysts and the stability of the protected intermediates contribute to a more predictable manufacturing timeline, reducing the likelihood of batch failures that can disrupt supply continuity. Furthermore, the ability to source starting materials from a broader range of commodity chemical suppliers enhances supply chain resilience, mitigating the risks associated with single-source dependencies for exotic reagents.
- Cost Reduction in Manufacturing: The streamlined nature of this synthetic route directly translates to lower production costs by minimizing solvent consumption, energy usage, and waste disposal fees. The elimination of complex purification steps required for isomer separation in traditional methods results in substantial savings in both time and materials. Additionally, the use of recyclable palladium catalysts and the potential for catalyst recovery further drive down the variable costs per kilogram of product. This economic efficiency allows suppliers to offer more competitive pricing structures without compromising on quality, providing a distinct advantage in price-sensitive markets such as nutraceuticals and generic pharmaceuticals.
- Enhanced Supply Chain Reliability: The reliance on stable, protected intermediates and common industrial solvents ensures that the supply chain is less vulnerable to fluctuations in the availability of specialized reagents. The process conditions are compatible with standard stainless steel reactors and filtration equipment, meaning that production can be easily shifted between different manufacturing sites if necessary. This flexibility is crucial for maintaining continuous supply in the face of global logistical challenges or regional disruptions. By adopting a process that is less sensitive to environmental variables and reagent quality, manufacturers can guarantee more consistent delivery schedules and reduce the lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method aligns well with green chemistry principles by reducing the generation of stoichiometric waste and avoiding the use of toxic heavy metals other than the catalytic palladium. The simplified workup procedures, often involving straightforward crystallization or extraction, minimize the volume of organic waste streams requiring treatment. This compliance with stringent environmental standards facilitates smoother regulatory approvals and reduces the long-term liability associated with waste management. Moreover, the process has been demonstrated to scale effectively from laboratory to pilot and commercial scales, ensuring that capacity can be ramped up rapidly to meet surging market demand without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented Heck reaction process. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the technology's capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios or sourcing strategies. The answers reflect the consensus on best practices for maximizing yield and purity while adhering to safety and quality standards.
Q: What are the key advantages of the Heck reaction method for resveratrol synthesis?
A: The Heck reaction method described in CN101117317B offers fewer reaction steps compared to traditional Wittig or Perkin condensations, utilizes readily available starting materials, and achieves higher overall yields with improved stereoselectivity for the (E)-isomer.
Q: How does the patent address impurity control during synthesis?
A: Impurity control is managed through the strategic use of protecting groups (such as acetyl or benzoyl) on the phenolic hydroxyls, which prevent side reactions during the coupling phase and allow for clean deprotection in the final step.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes standard industrial solvents like DMF, toluene, and alcohols, along with robust palladium catalysts, making it highly adaptable for scale-up from kilogram to multi-ton production without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Stilbene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic strategies outlined in CN101117317B for the production of high-value stilbene derivatives. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these patented methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and quality of every batch, guaranteeing that our resveratrol and piceatannol intermediates meet the highest industry standards.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize your supply chain and accelerate your time to market. Partner with NINGBO INNO PHARMCHEM to secure a sustainable, high-quality source of critical pharmaceutical intermediates.
