Scalable Industrial Synthesis of Resveratrol Intermediates for Global Pharmaceutical Supply Chains
Scalable Industrial Synthesis of Resveratrol Intermediates for Global Pharmaceutical Supply Chains
The global demand for high-purity polyhydroxystilbene compounds, particularly resveratrol, has surged due to its extensive applications in pharmaceuticals, nutraceuticals, and cosmetic formulations. Patent CN1300077C introduces a groundbreaking preparation method that addresses the critical bottlenecks of traditional extraction and synthesis techniques. This innovative approach utilizes a condensation reaction between 3,5-dimethoxybenzaldehyde and p-methoxyphenylacetonitrile, catalyzed by sodium alkoxide, to construct the fundamental diphenylethylene skeleton. Unlike previous methods that relied on complex organometallic couplings, this pathway offers a streamlined route to both cis- and trans-resveratrol isomers through subsequent hydrolysis, decarboxylation, and demethylation steps. The strategic design of this synthesis ensures excellent atom economy and minimizes environmental impact, positioning it as a superior choice for industrial-scale manufacturing. By leveraging readily available raw materials and avoiding hazardous reagents, this technology provides a robust foundation for reliable resveratrol supplier networks aiming to meet stringent quality standards.
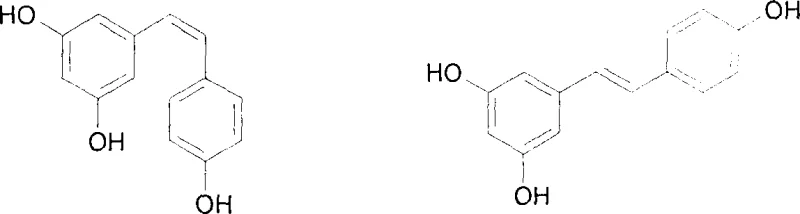
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of resveratrol and its analogues has been plagued by significant technical and economic hurdles that hinder large-scale commercial viability. Traditional methodologies such as the Wittig reaction, Wittig-Horner reaction, and Heck coupling often necessitate the use of expensive and sensitive reagents, including phosphorus ylides and palladium catalysts, which drastically inflate the cost of goods sold. Furthermore, these conventional routes frequently suffer from poor atom economy, generating substantial quantities of stoichiometric byproducts like triphenylphosphine oxide that are difficult to remove and pose disposal challenges. The purification processes associated with these older methods often require extensive chromatography or recrystallization steps, leading to significant yield losses and extended production cycles. Additionally, the reliance on transition metals introduces the risk of heavy metal contamination in the final active pharmaceutical ingredient, necessitating costly downstream purification to meet regulatory limits. These factors collectively render many legacy synthesis routes unsuitable for the high-volume, cost-sensitive demands of the modern fine chemical industry.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent data presents a paradigm shift by employing a condensation-decarboxylation strategy that circumvents the drawbacks of cross-coupling chemistry. This approach initiates with a base-catalyzed condensation of inexpensive aldehyde and nitrile precursors, forming the carbon-carbon double bond under relatively mild thermal conditions without the need for precious metal catalysts. The subsequent transformation involves hydrolysis of the nitrile group to a carboxylic acid, followed by a copper-catalyzed decarboxylation in quinoline, which effectively removes the auxiliary functionality to reveal the desired stilbene core. This sequence not only simplifies the operational workflow but also enhances the overall yield by minimizing side reactions and facilitating easier product isolation through crystallization. The final demethylation step using boron tribromide is highly selective, ensuring the preservation of the sensitive olefinic bond while efficiently unmasking the phenolic hydroxyl groups. This holistic process design results in a manufacturing protocol that is inherently safer, more economical, and environmentally friendlier than its predecessors.
Mechanistic Insights into Sodium Alkoxide Catalyzed Condensation and Decarboxylation
The core of this synthetic innovation lies in the efficient construction of the stilbene backbone via a Knoevenagel-type condensation mechanism facilitated by sodium alkoxide bases. In this initial phase, the alpha-protons of the p-methoxyphenylacetonitrile are deprotonated by the alkoxide to generate a reactive carbanion species, which subsequently attacks the carbonyl carbon of the 3,5-dimethoxybenzaldehyde. This nucleophilic addition is followed by dehydration to establish the conjugated double bond system, yielding the trimethoxy-substituted nitrile intermediate. The choice of sodium ethoxide or sodium methoxide as the catalyst is critical, as these strong bases provide the necessary kinetic energy for the reaction to proceed at moderate temperatures while maintaining high selectivity for the E/Z isomer mixture. Following the skeleton assembly, the nitrile group undergoes alkaline hydrolysis in a high-boiling solvent like ethylene glycol, converting it into the corresponding cinnamic acid derivative. This hydrolysis step is pivotal as it activates the molecule for the subsequent thermal decarboxylation, where the carboxyl group is expelled as carbon dioxide in the presence of copper powder, finalizing the formation of the trimethoxystilbene framework.
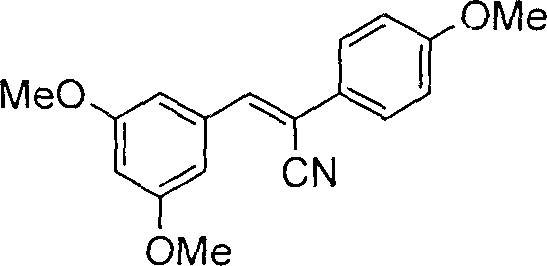
Impurity control within this mechanistic pathway is achieved through the inherent selectivity of the reaction conditions and the physical properties of the intermediates. The use of specific solvent systems, such as ethanol or ethylene glycol, allows for the precipitation of the desired intermediates upon cooling, effectively separating them from soluble byproducts and unreacted starting materials. During the decarboxylation phase, the use of quinoline as a high-boiling solvent ensures that the reaction temperature remains sufficient to drive the elimination of carbon dioxide without degrading the sensitive stilbene double bond. Furthermore, the potential formation of cis/trans isomer mixtures is managed either through precise column chromatography separation or via a catalytic isomerization process using trace amounts of iodine. This ability to interconvert or separate isomers ensures that the final product specification can be tightly controlled to meet the specific requirements for either the cis- or trans-resveratrol forms. The final demethylation with boron tribromide is conducted at low temperatures to prevent polymerization or oxidation of the phenolic rings, guaranteeing a high-purity end product with minimal colored impurities.
How to Synthesize Resveratrol Efficiently
The synthesis of resveratrol via this patented route involves a logical sequence of four distinct chemical transformations that can be executed in standard glass-lined or stainless steel reactors. The process begins with the condensation of aromatic aldehyde and nitrile components, followed by hydrolysis to the acid, thermal decarboxylation to the stilbene, and finally oxidative demethylation. Each step has been optimized to maximize yield and minimize waste, making it an ideal candidate for technology transfer. For detailed operational parameters, safety protocols, and specific workup procedures required to replicate this high-efficiency synthesis, please refer to the standardized technical guide below.
- Condense 3,5-dimethoxybenzaldehyde with p-methoxyphenylacetonitrile using sodium alkoxide catalysis to form the stilbene nitrile skeleton.
- Hydrolyze the nitrile intermediate to the corresponding carboxylic acid using strong base in ethylene glycol solvent.
- Perform decarboxylation using copper powder in quinoline, followed by demethylation with boron tribromide to yield final resveratrol.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthesis route offers profound advantages by fundamentally restructuring the cost drivers associated with resveratrol manufacturing. The substitution of expensive palladium catalysts and phosphorus reagents with commodity chemicals like sodium alkoxide and copper powder results in a drastic reduction in raw material expenditure. Moreover, the simplified purification workflow, which relies heavily on crystallization rather than preparative chromatography, significantly lowers solvent consumption and processing time. This efficiency translates directly into improved margin potential for downstream manufacturers and allows for more competitive pricing in the global marketplace. The robustness of the reaction conditions also means that the process is less susceptible to batch-to-batch variability, ensuring a consistent supply of material that meets rigorous quality specifications without the need for excessive safety stocks.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of inexpensive, bulk-available starting materials such as 3,5-dimethoxybenzaldehyde create a substantially lower cost base for production. By avoiding the complex waste streams associated with phosphine oxides, the facility also saves significantly on waste treatment and disposal costs, further enhancing the overall economic viability of the project. The high atom economy of the condensation and decarboxylation steps ensures that a greater proportion of the input mass is converted into valuable product, reducing the effective cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: The reliance on widely sourced chemical feedstocks mitigates the risk of supply disruptions that often plague specialized reagent markets. Since the key reagents are produced by multiple global suppliers, procurement teams can easily diversify their vendor base to ensure continuity of supply even during market fluctuations. The simplicity of the synthesis also reduces the dependency on highly specialized contract manufacturing organizations, allowing for greater flexibility in sourcing strategies and potentially shortening lead times for order fulfillment.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations such as reflux, filtration, and crystallization that are standard in multi-ton chemical plants. The absence of toxic heavy metals in the primary skeleton construction simplifies the environmental compliance profile, making it easier to obtain necessary permits for expansion. Additionally, the reduced solvent load and the ability to recycle certain reaction media contribute to a smaller environmental footprint, aligning with the increasing corporate sustainability goals of major pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resveratrol synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these details is crucial for R&D directors and process engineers who are assessing the potential for integrating this method into existing production lines.
Q: What are the primary advantages of this resveratrol synthesis route over Wittig reactions?
A: This method avoids expensive phosphorus ylides and generates fewer byproducts, leading to simpler purification and lower raw material costs compared to traditional Wittig or Heck coupling methods.
Q: How is the cis/trans isomer ratio controlled in this process?
A: The process produces a mixture of isomers which can be separated via column chromatography or selectively isomerized to the trans-form using iodine catalysis, ensuring high purity of the desired bioactive isomer.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the use of commodity chemicals like sodium alkoxide and common solvents, combined with robust reaction conditions like reflux and crystallization, makes this route highly adaptable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Resveratrol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the fast-evolving pharmaceutical intermediates market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity resveratrol and related stilbene derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex demethylation and isomerization chemistries allows us to offer customized solutions tailored to the specific isomeric requirements of your final formulation.
We invite you to collaborate with us to leverage this cost-effective and scalable synthesis route for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis specific to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can optimize your supply chain and reduce your overall cost of goods.
