Scalable Synthesis of Chiral Tetrahydroquinoline Derivatives for Advanced Cardiovascular Therapeutics
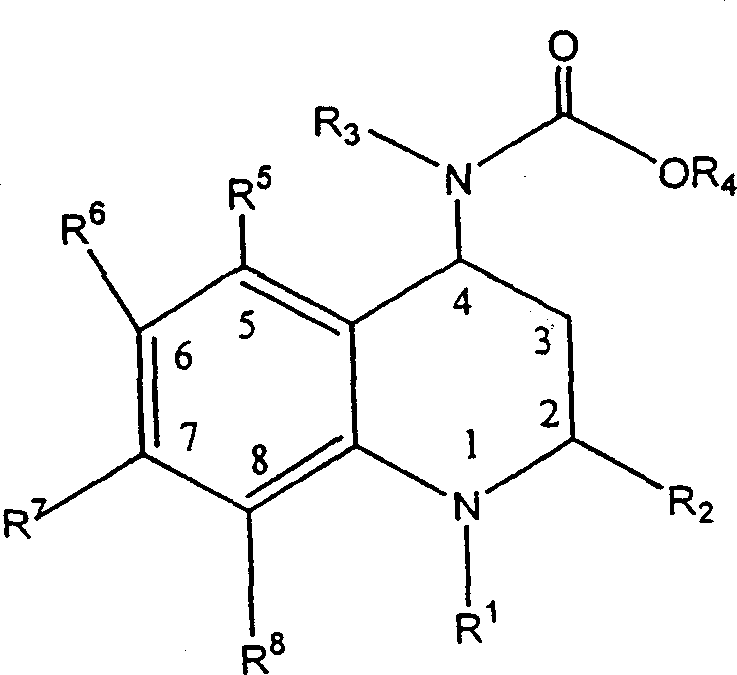
The pharmaceutical industry's relentless pursuit of effective treatments for atherosclerosis and coronary artery disease has placed Cholesterol Ester Transfer Protein (CETP) inhibitors at the forefront of cardiovascular research. Patent CN1173953C discloses a sophisticated methodology for preparing 4-carboxyamino-2-substituted-1,2,3,4-tetrahydroquinoline derivatives, specifically targeting the potent inhibition of CETP to modulate lipoprotein profiles. This technology addresses a critical gap in therapeutic options by offering a robust synthetic route to complex chiral molecules that were previously difficult to manufacture with high stereochemical fidelity. The core innovation lies not merely in the final molecule but in the strategic manipulation of intermediates to ensure scalability and purity, essential factors for any reliable pharmaceutical intermediate supplier aiming to support global drug development pipelines. By leveraging specific diastereomeric resolution techniques, this process circumvents the bottlenecks typically associated with chiral separations in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active tetrahydroquinoline derivatives has been plagued by significant challenges regarding stereocontrol and purification efficiency. Traditional approaches often relied heavily on chiral chromatography for the separation of enantiomers, a technique that, while effective on a laboratory scale, presents severe economic and logistical barriers when transitioning to commercial production. The reliance on chiral stationary phases introduces high operational costs, limits batch sizes, and often results in substantial material loss during the recycling of solents and columns. Furthermore, conventional routes frequently suffered from the formation of difficult-to-remove impurities due to non-selective cyclization steps, necessitating multiple recrystallization cycles that eroded overall yield. For procurement teams, these inefficiencies translate into volatile supply chains and unpredictable cost reduction in API manufacturing initiatives, as the complexity of the synthesis directly correlates with the risk of production delays and quality failures.
The Novel Approach
The methodology outlined in CN1173953C represents a paradigm shift by integrating classical resolution strategies with modern catalytic transformations to achieve superior process control. Instead of attempting to separate enantiomers at the final stage, the process introduces chirality earlier through the formation of diastereomeric salts using resolving agents like (-)-dibenzoyl-L-tartrate or (-)-di-p-toluoyl-L-tartrate. This approach capitalizes on the distinct physical properties of diastereomers, allowing for purification via simple crystallization rather than expensive chromatography. The patent explicitly highlights that intermediates II, V, VI, and VII are easily isolated in crystalline form, which serves as a built-in purification mechanism at multiple stages of the synthesis. This stepwise purification strategy ensures that the final high-purity CETP inhibitor intermediate is obtained with exceptional optical purity, significantly mitigating the risk of toxicological issues associated with the wrong enantiomer while streamlining the manufacturing workflow for industrial applications.
Mechanistic Insights into Diastereomeric Resolution and Reductive Amination
The chemical architecture of this synthesis is built upon a foundation of precise mechanistic controls, beginning with the condensation of benzotriazole, 4-(trifluoromethyl)aniline, and propionaldehyde. This initial step generates a key amine intermediate that serves as the nucleophile for the subsequent cyclization. The cyclization itself is elegantly executed using vinyl carbamates in the presence of an acid catalyst, forming the tetrahydroquinoline core with high regioselectivity. Following the construction of the heterocyclic ring, the removal of the benzyloxycarbonyl protecting group via catalytic transfer hydrogenation using ammonium formate and palladium on carbon reveals the free amine functionality. This specific deprotection method is advantageous as it avoids the use of high-pressure hydrogen gas, enhancing safety profiles for commercial scale-up of complex tetrahydroquinolines. The revealed amine is then subjected to the critical resolution step, where the interaction with the chiral tartrate derivative creates a lattice energy difference sufficient to precipitate the desired (2R, 4S) isomer selectively.
Following resolution, the synthesis proceeds through a reductive amination with 3,5-bis(trifluoromethyl)benzaldehyde, utilizing sodium triacetoxy borohydride as the reducing agent. This reagent is chosen for its mildness and selectivity, preventing the over-reduction of other sensitive functional groups within the molecule. The final transformation involves the carbamylation of the secondary amine using methyl chloroformate in the presence of a base such as sodium carbonate. Throughout this sequence, the patent emphasizes the importance of maintaining specific temperature ranges, such as 20°C to 25°C for the final carbamylation, to prevent racemization or decomposition. The ability to isolate stable crystalline salts at intermediate stages acts as a 'chemical checkpoint,' ensuring that impurities do not carry forward to the final step. This rigorous control over the reaction pathway is what enables the production of materials meeting the stringent purity specifications required for clinical-grade pharmaceutical ingredients.
How to Synthesize Chiral Tetrahydroquinoline Derivatives Efficiently
The synthesis of these complex molecules requires a disciplined approach to reaction conditions and workup procedures to maximize yield and optical purity. The process begins with the preparation of the benzotriazole adduct, followed by cyclization and deprotection to yield the racemic amine. The pivotal step involves the formation of the diastereomeric tartrate salt, which must be carefully controlled to ensure maximum enrichment of the desired stereoisomer. Once the chiral amine is secured, reductive amination and final carbamylation complete the sequence. For detailed operational parameters, stoichiometry, and specific workup instructions, please refer to the standardized guide below.
- Condense benzotriazole with 4-(trifluoromethyl)aniline and propionaldehyde to form the key amine intermediate.
- Perform cyclization using vinyl carbamates followed by catalytic hydrogenation to generate the tetrahydroquinoline core.
- Execute chiral resolution using (-)-dibenzoyl-L-tartrate to isolate the desired (2R, 4S) stereoisomer before final functionalization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages for organizations seeking to optimize their supply chain resilience and cost structures. The primary value driver is the elimination of chiral chromatography, which is notoriously capital-intensive and difficult to scale. By replacing this with crystallization-based resolution, the process becomes compatible with standard stainless steel reactors and filtration equipment found in most multipurpose chemical plants. This compatibility drastically reduces the barrier to entry for manufacturing, allowing for faster technology transfer and more competitive pricing models. Furthermore, the ability to isolate and purify intermediates at multiple stages reduces the burden on the final purification step, leading to higher overall throughput and reduced waste generation. These factors collectively contribute to a more robust and predictable supply chain, essential for maintaining continuity in the production of life-saving cardiovascular medications.
- Cost Reduction in Manufacturing: The substitution of chiral HPLC with diastereomeric crystallization represents a significant structural cost saving. Chromatographic separations require expensive resins, large volumes of high-grade solvents, and specialized equipment, all of which drive up the cost of goods sold. In contrast, crystallization utilizes standard solvents and equipment, significantly lowering both capital expenditure and operating expenses. Additionally, the high recovery rates associated with crystallization minimize material loss, further enhancing the economic viability of the process. This efficiency allows suppliers to offer more competitive pricing without compromising on the quality or purity of the final intermediate, aligning perfectly with procurement goals for sustainable cost management.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as benzotriazole, vinyl carbamates, and common chiral acids like dibenzoyl-L-tartrate ensures a stable supply of raw materials. Unlike proprietary catalysts that may have single-source dependencies, these reagents are commodity chemicals with established global supply networks. This diversity in sourcing options mitigates the risk of supply disruptions caused by geopolitical issues or manufacturer-specific problems. Moreover, the robustness of the crystallization steps means that the process is less sensitive to minor variations in reaction conditions, resulting in consistent batch-to-batch quality. This reliability is crucial for reducing lead time for high-purity intermediates and ensuring that downstream drug formulation schedules are met without delay.
- Scalability and Environmental Compliance: The process design inherently supports scalability, moving seamlessly from kilogram-scale development to multi-ton commercial production. The use of sodium triacetoxy borohydride, while a specialized reagent, is manageable in large batches with appropriate quenching protocols, and the overall solvent profile (toluene, ethanol, ethyl acetate) is conducive to efficient recovery and recycling. This focus on standard solvents simplifies waste treatment and aligns with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. The ability to recycle solvents and the reduction in hazardous waste associated with chromatographic media disposal further enhance the environmental footprint of the manufacturing process, supporting corporate sustainability initiatives and regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these tetrahydroquinoline derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for decision-makers evaluating this technology for their portfolios.
Q: How does the diastereomeric salt formation improve purity compared to chiral chromatography?
A: The patent describes using classical diastereomeric salt formation with (-)-dibenzoyl-L-tartrate, which allows for crystallization-based purification. This is significantly more scalable and cost-effective for industrial production than chiral HPLC, which is often limited to analytical or small preparative scales.
Q: What are the critical intermediates in this tetrahydroquinoline synthesis?
A: The process relies on four key isolable intermediates: the benzotriazole adduct, the cyclic carbamate, the free amine salt, and the reductively aminated product. Each can be purified by crystallization, ensuring high overall purity.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the methodology avoids exotic catalysts and relies on standard unit operations like filtration, crystallization, and standard reductive amination, making it highly amenable to commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroquinoline Supplier
The technological potential of CN1173953C is immense, offering a clear pathway to high-value cardiovascular therapeutics, but realizing this potential requires a manufacturing partner with deep technical expertise and proven scale-up capabilities. NINGBO INNO PHARMCHEM stands as a premier CDMO partner, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art reactor trains capable of handling the specific thermal and mixing requirements of reductive aminations and cryogenic resolutions. We understand that in the pharmaceutical sector, consistency is key; therefore, our operations are governed by stringent purity specifications and supported by rigorous QC labs that utilize advanced analytical techniques to verify optical purity and impurity profiles at every stage of synthesis.
We invite R&D directors and procurement leaders to engage with us to explore how this optimized synthetic route can be integrated into your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and our demonstrated capability to deliver complex intermediates with speed and precision.
