Advanced Ruthenium-Catalyzed Hydrogenation for Scalable Tetrahydroquinoline Manufacturing
The pharmaceutical and fine chemical industries continuously seek robust synthetic routes for privileged scaffolds like the 1,2,3,4-tetrahydroquinoline core, which is ubiquitous in bioactive natural products and therapeutic agents. Patent CN112250628B introduces a transformative method for preparing these compounds via catalytic hydrogenation using a ruthenium catalyst system. This technology addresses critical bottlenecks in traditional synthesis by employing p-cymene ruthenium chloride dimer as a precatalyst and molecular hydrogen as the reducing agent. The significance of this innovation lies in its ability to convert various quinoline derivatives into their saturated counterparts with exceptional efficiency and selectivity. By leveraging commercially available reagents and operating under ambient conditions, this process represents a paradigm shift towards greener and more sustainable manufacturing practices for high-value chemical intermediates used in drug discovery and development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,3,4-tetrahydroquinoline compounds has relied heavily on the Aza-Diels-Alder reaction or reduction methods utilizing stoichiometric amounts of silanes. The Aza-Diels-Alder approach often necessitates the use of strong acids as catalysts, which poses severe challenges regarding equipment corrosion and ecological impact due to acidic waste streams. Furthermore, alternative reduction strategies employing silver triflate (AgOTf) combined with silane reducing agents suffer from poor atom economy and generate substantial quantities of silicon-containing byproducts that are difficult to dispose of responsibly. These conventional pathways not only inflate production costs through the consumption of expensive reagents but also complicate downstream purification processes, thereby limiting their viability for large-scale industrial applications where environmental compliance and cost-efficiency are paramount concerns for supply chain managers.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a highly efficient ruthenium catalytic system that operates under remarkably mild conditions. By employing p-cymene ruthenium chloride dimer in conjunction with phosphine ligands, the reaction proceeds smoothly at room temperature and normal pressure, eliminating the need for energy-intensive heating or high-pressure equipment. The use of molecular hydrogen as the sole reducing agent ensures that the only byproduct is potentially trace amounts of unreacted gas, leading to a synthesis process with near-perfect atom economy. This approach drastically simplifies the operational workflow, as the catalyst system is stable to air and moisture, allowing for easier handling and setup compared to sensitive organometallic alternatives. The versatility of this method is further demonstrated by its compatibility with a wide range of substrates, including those with methyl, phenyl, and halogen substituents.
Mechanistic Insights into Ruthenium-Catalyzed Hydrogenation
The core of this technological advancement lies in the specific interaction between the ruthenium center and the quinoline substrate facilitated by the phosphine ligand environment. The p-cymene ruthenium chloride dimer serves as a robust precatalyst that, upon activation with phosphine ligands such as triphenylphosphine or tricyclohexylphosphine, forms an active species capable of coordinating with the nitrogen-containing heterocycle. The mechanism likely involves the oxidative addition of hydrogen to the metal center followed by migratory insertion into the carbon-nitrogen or carbon-carbon double bonds of the quinoline ring. This catalytic cycle is highly tuned to favor the complete saturation of the heterocyclic ring while preserving sensitive functional groups attached to the aromatic system. The choice of alcohol solvents like methanol or ethanol plays a crucial role in stabilizing the transition states and facilitating proton transfer steps essential for the reduction process, ensuring high turnover numbers and minimal catalyst deactivation.
Impurity control is a critical aspect of this synthesis, particularly for pharmaceutical intermediates where regulatory standards are stringent. The high selectivity observed in this ruthenium-catalyzed system minimizes the formation of partially reduced intermediates or over-reduced byproducts that often plague less specific reduction methods. For instance, the method successfully tolerates halogen substituents, as evidenced by the conversion of 5-bromoquinoline and 6-chloroquinoline without undergoing dehalogenation, a common side reaction in catalytic hydrogenation. This chemoselectivity is vital for maintaining the structural integrity of complex molecules intended for further functionalization. The ability to achieve yields exceeding 90% with high purity reduces the burden on purification steps, directly impacting the overall cost of goods and timeline for process development teams aiming to scale these materials.
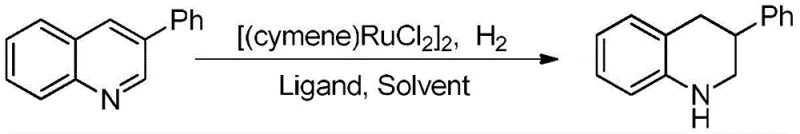
How to Synthesize Tetrahydroquinoline Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves a straightforward mixing of the ruthenium precatalyst, the chosen phosphine ligand, and the quinoline substrate in a suitable alcohol solvent. Following the establishment of a hydrogen atmosphere, the reaction proceeds autonomously at ambient temperature, requiring minimal monitoring or intervention. This simplicity allows technical teams to focus on optimization rather than troubleshooting complex reaction parameters. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which outlines the precise steps validated by the patent examples.
- Mix p-cymene ruthenium chloride dimer, a phosphine ligand (such as triphenylphosphine), and the quinoline substrate in an alcohol solvent like methanol or ethanol.
- Purge the reaction vessel with hydrogen gas to create an inert atmosphere, then maintain the reaction at room temperature and normal pressure for 8 to 12 hours.
- Upon completion, concentrate the reaction mixture via rotary evaporation and purify the crude product using column chromatography with a petroleum ether and dichloromethane eluent.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this ruthenium-catalyzed hydrogenation method offers substantial strategic benefits for procurement and supply chain operations. The shift away from expensive and hazardous silane reducing agents to inexpensive hydrogen gas fundamentally alters the cost structure of the manufacturing process. Additionally, the use of commercially available, shelf-stable catalysts reduces the risk of supply disruptions associated with custom-synthesized reagents. The mild reaction conditions translate to lower energy consumption and reduced wear on reactor vessels, contributing to long-term operational expenditure savings. These factors collectively enhance the economic feasibility of producing tetrahydroquinoline derivatives at scale, making it an attractive option for companies seeking to optimize their supply chains for key pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of costly silane reagents and the use of catalytic amounts of ruthenium significantly lower the raw material costs per kilogram of product. Furthermore, the high atom economy means less waste is generated, reducing disposal fees and environmental compliance costs. The simplified workup procedure, which avoids complex quenching steps required for reactive hydrides, streamlines the production line and reduces labor hours associated with batch processing.
- Enhanced Supply Chain Reliability: Since all key reagents, including the ruthenium dimer and phosphine ligands, are commercially available from multiple global suppliers, the risk of single-source dependency is minimized. The stability of the catalyst to air and moisture simplifies logistics and storage requirements, allowing for safer and more flexible inventory management. This reliability ensures consistent production schedules and reduces the likelihood of delays caused by reagent degradation or availability issues.
- Scalability and Environmental Compliance: The operation at normal pressure and room temperature removes the need for specialized high-pressure autoclaves, allowing the process to be scaled up using standard glass-lined or stainless steel reactors found in most multipurpose facilities. The green nature of the process, generating minimal hazardous waste, aligns with increasingly strict environmental regulations, facilitating smoother permitting and audit processes for manufacturing sites aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ruthenium-catalyzed hydrogenation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is essential for technical teams evaluating the adoption of this method for their specific intermediate synthesis needs.
Q: What are the primary advantages of this ruthenium method over traditional silane reduction?
A: Unlike traditional methods using expensive silanes which generate significant silicon-containing waste, this ruthenium-catalyzed hydrogenation uses hydrogen gas as a clean reducing agent. This results in superior atom economy, eliminates heavy metal waste issues associated with other catalysts, and utilizes commercially available, stable reagents.
Q: Does this catalytic system tolerate functional groups like halogens?
A: Yes, the method demonstrates excellent functional group tolerance. The patent data confirms successful hydrogenation of substrates containing bromine and chlorine substituents, such as 5-bromoquinoline and 6-chloroquinoline, without dehalogenation side reactions, ensuring high selectivity for complex intermediates.
Q: What are the typical reaction conditions required for this synthesis?
A: The process operates under exceptionally mild conditions, specifically at room temperature and normal atmospheric pressure using a hydrogen balloon. This eliminates the need for high-pressure autoclaves or elevated temperatures, significantly simplifying the equipment requirements and enhancing operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in the timely delivery of high-quality pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like this ruthenium-catalyzed hydrogenation can be seamlessly transferred from the bench to the plant. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of tetrahydroquinoline derivative meets the exacting standards required for downstream drug synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages tailored to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring a secure and efficient supply chain for your critical chemical building blocks.
