Advanced One-Pot Synthesis of Vortioxetine Hydrobromide for Commercial Scale-Up
Introduction to Patent CN113563285A: A Breakthrough in Antidepressant Synthesis
The pharmaceutical landscape for treating Major Depressive Disorder (MDD) has been significantly advanced by the development of Vortioxetine, a multimodal antidepressant with a unique pharmacological profile. However, the commercial viability of such complex molecules often hinges on the efficiency and scalability of their synthetic routes. Patent CN113563285A introduces a transformative preparation method that addresses critical bottlenecks in the existing manufacturing processes for Vortioxetine and its hydrobromide salt. This innovation centers on a novel one-pot synthesis strategy that utilizes 2,4-dimethylthiophenol, 2-bromoiodobenzene, and piperazine as key starting materials, catalyzed by cuprous iodide (CuI) in the presence of specific organic bases. By shifting away from traditional palladium-catalyzed systems and heterogeneous inorganic base conditions, this technology enables a homogeneous reaction environment that drastically reduces processing time while enhancing product quality. For industry stakeholders, this represents a pivotal opportunity to optimize the supply chain for high-purity pharmaceutical intermediates, ensuring both economic efficiency and robust production capabilities.
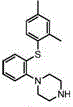
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Vortioxetine has relied heavily on palladium-catalyzed cross-coupling reactions, which present substantial challenges for large-scale industrial application. Traditional methods often employ expensive palladium complexes coupled with phosphine ligands, which not only inflate the raw material costs but also introduce significant toxicity concerns due to the ligands themselves. A major drawback of these prior art methods is the difficulty in controlling residual metal levels; strict regulatory limits on palladium content in Active Pharmaceutical Ingredients (APIs) necessitate complex and costly purification steps to ensure patient safety. Furthermore, earlier protocols utilizing inorganic bases like potassium phosphate frequently result in heterogeneous reaction mixtures. This lack of homogeneity severely impedes mass transfer, leading to sluggish reaction kinetics that can extend processing times to over 40 hours. Such inefficiencies result in lower yields, typically hovering around 70-76%, and generate higher levels of impurities, complicating downstream isolation and crystallization processes.
The Novel Approach
In stark contrast, the methodology disclosed in CN113563285A leverages a copper-catalyzed system that fundamentally alters the reaction dynamics. By substituting expensive palladium catalysts with economical cuprous iodide and replacing inorganic bases with soluble organic amines such as triethylamine or N,N-diisopropylethylamine (DIPEA), the process achieves a fully homogeneous reaction phase. This transition to a homogeneous system is the key driver behind the dramatic reduction in reaction time, compressing the synthesis window from days down to merely 4 to 5 hours. The use of organic bases ensures that all reagents are uniformly distributed in the solvent (typically isopropanol), facilitating rapid interaction between the thiol, the aryl halide, and the amine. Consequently, this approach not only boosts the isolated yield to impressive levels ranging from 89% to 96% but also delivers exceptional purity profiles, with single impurity levels consistently maintained below 0.1%. This streamlined one-pot procedure eliminates the need for intermediate isolation steps, thereby reducing solvent consumption and operational complexity.
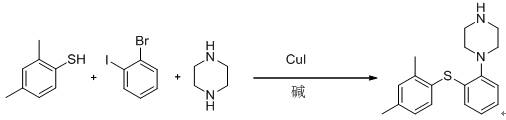
Mechanistic Insights into CuI-Catalyzed Homogeneous Coupling
The core mechanistic advantage of this novel process lies in the synergistic effect between the copper catalyst and the organic base within a protic solvent system. In the presence of organic amines like triethylamine, the cuprous iodide catalyst effectively mediates the formation of carbon-sulfur and carbon-nitrogen bonds in a sequential or concurrent manner, depending on the specific addition protocol. The organic base serves a dual function: it acts as a proton scavenger to drive the equilibrium forward and, crucially, it solubilizes the reaction components to prevent the formation of insoluble salt precipitates that characterize inorganic base systems. This homogeneous environment allows for precise thermal control and uniform heating, which minimizes localized hot spots that often lead to decomposition or side reactions. The patent data indicates that the reaction proceeds efficiently at reflux temperatures in isopropanol, suggesting a robust activation energy profile that is accessible without extreme conditions. Furthermore, the compatibility of CuI with the thiol and halide functionalities ensures high chemoselectivity, preventing the reduction of the aryl halide or the oxidation of the thiol, which are common pitfalls in less optimized systems.
Impurity control is another critical aspect where this mechanism excels, particularly through the optimization of reagent addition sequences. The patent highlights that adding piperazine simultaneously with the starting materials can lead to competitive side reactions, whereas a staged addition protocol significantly mitigates this risk. By allowing the initial coupling between the thiol and the aryl halide to proceed before introducing the piperazine nucleophile, the process minimizes the formation of bis-arylated byproducts or other oligomeric impurities. This strategic temporal separation of reagents ensures that the highly reactive aryl-copper intermediate is primarily consumed by the desired sulfur nucleophile first, establishing the correct scaffold before the nitrogen nucleophile attacks. The result is a cleaner crude reaction mixture that requires less aggressive purification, preserving the overall yield and ensuring that the final hydrobromide salt meets stringent pharmacopeial standards for related substances.
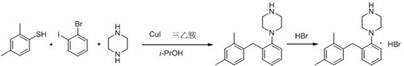
How to Synthesize Vortioxetine Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing pilot and commercial manufacturing facilities. The protocol utilizes readily available solvents like isopropanol and common organic bases, eliminating the need for specialized anhydrous conditions or exotic reagents that often complicate scale-up. The process begins with the charging of the thiol and aryl halide substrates along with the copper catalyst and organic base under an inert atmosphere, followed by heating to reflux. The simplicity of the work-up procedure, involving filtration, concentration, and recrystallization, further underscores its suitability for high-throughput production environments. For detailed operational parameters, stoichiometry, and specific temperature profiles required to replicate the high yields reported in the patent, please refer to the standardized synthesis guide below.
- Combine 2,4-dimethylthiophenol, 2-bromoiodobenzene, an organic base (such as triethylamine or DIPEA), and cuprous iodide catalyst in isopropanol under nitrogen protection.
- Heat the mixture to reflux for 2-5 hours to facilitate the coupling reaction, optionally adding piperazine in a staged manner to minimize side reactions.
- Cool the reaction, filter, concentrate the filtrate, recrystallize the residue, and finally treat with hydrobromic acid to obtain the pure hydrobromide salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement and supply chain perspective, the adoption of this CuI-catalyzed organic base methodology offers profound advantages over legacy palladium-based routes. The shift to a non-precious metal catalyst immediately decouples the production cost from the volatile market prices of palladium and phosphine ligands, providing a more stable and predictable cost structure for long-term supply agreements. Additionally, the elimination of heavy metal removal steps simplifies the manufacturing workflow, reducing the consumption of specialized scavenging resins and the associated waste disposal costs. This efficiency gain translates directly into a more competitive pricing model for the final API intermediate, making it an attractive option for generic drug manufacturers seeking to optimize their bill of materials. The robustness of the homogeneous reaction system also implies a lower risk of batch failures due to mixing issues, thereby enhancing the reliability of supply deliveries.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts and phosphine ligands with inexpensive cuprous iodide results in a drastic reduction in raw material expenditures. Furthermore, the simplified purification process, which no longer requires rigorous testing and removal of trace palladium residues, lowers the operational overhead associated with quality control and waste management. The ability to achieve high yields in a shorter timeframe also maximizes the throughput of existing reactor assets, effectively lowering the fixed cost per kilogram of product manufactured.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as triethylamine, DIPEA, and isopropanol ensures that the supply chain is resilient against shortages of specialized reagents. Unlike complex ligand systems that may have limited suppliers and long lead times, the materials required for this process are widely available from multiple global sources. This diversification of the supply base mitigates the risk of production stoppages and ensures consistent availability of the intermediate for downstream API synthesis, supporting uninterrupted medication supply for patients.
- Scalability and Environmental Compliance: The homogeneous nature of the reaction facilitates straightforward scale-up from laboratory to multi-ton production scales without the engineering challenges associated with handling heterogeneous slurries. The use of isopropanol, a greener solvent compared to many chlorinated alternatives often used in palladium chemistry, aligns with modern environmental, health, and safety (EHS) standards. Reduced reaction times and higher atom economy contribute to a smaller environmental footprint, helping manufacturers meet increasingly stringent sustainability goals and regulatory requirements for green chemistry practices.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the operational advantages and chemical rationale behind the process improvements. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and process validation.
Q: Why is an organic base preferred over inorganic bases like potassium phosphate in this synthesis?
A: Organic bases such as triethylamine or DIPEA create a homogeneous reaction phase, which significantly accelerates the reaction rate compared to the heterogeneous systems formed by inorganic bases. This homogeneity reduces reaction time from over 40 hours to just 4-5 hours and improves overall yield and purity.
Q: What are the advantages of using Cuprous Iodide (CuI) instead of Palladium catalysts?
A: Cuprous Iodide is significantly more cost-effective than palladium complexes and eliminates the need for expensive phosphine ligands. Furthermore, it avoids the stringent requirement for removing trace heavy metal residues (palladium) from the final API, simplifying the purification process and ensuring regulatory compliance.
Q: How does the staged addition of piperazine affect the product quality?
A: Adding piperazine after the initial coupling step optimizes the feeding sequence, which effectively suppresses the formation of byproducts. This strategic timing leads to a substantial improvement in product purity, often exceeding 99%, and maximizes the isolated yield of the target compound.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vortioxetine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive pharmaceutical market. Our team of expert chemists has thoroughly analyzed the technological advancements presented in CN113563285A and is fully equipped to translate this intellectual property into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and compliant. Our state-of-the-art facilities are designed to handle complex coupling reactions with precision, supported by rigorous QC labs that guarantee stringent purity specifications for every batch of Vortioxetine intermediate we produce.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific volume requirements and cost targets. By leveraging our expertise in process chemistry and supply chain management, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this copper-catalyzed method. Please contact us today to request specific COA data, route feasibility assessments, and sample availability, and let us demonstrate how NINGBO INNO PHARMCHEM can become your trusted partner in delivering high-quality antidepressant intermediates to the global market.
