Revolutionizing Prenol Production: A Deep Dive into Dual-Enzyme Biocatalysis for Commercial Scale-Up
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the need for greener, more selective, and cost-effective synthetic routes. A pivotal advancement in this domain is documented in patent CN110643556A, which discloses a sophisticated recombinant genetically engineered bacterium capable of co-expressing alkenal reductase and glucose dehydrogenase. This innovation addresses a longstanding challenge in the synthesis of alpha,beta-unsaturated alcohols, specifically the production of prenol from prenaldehyde. Traditional chemical methods often struggle with thermodynamic and kinetic barriers that favor the reduction of carbon-carbon double bonds over carbonyl groups, leading to undesirable saturated by-products. The technology outlined in this patent offers a compelling solution by leveraging the exquisite regioselectivity of biological catalysts, ensuring that the reduction occurs exclusively at the C=O bond. For R&D directors and procurement specialists seeking a reliable prenol supplier, this biocatalytic approach represents a paradigm shift towards higher purity and operational efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of prenol, a critical intermediate for pyrethroid insecticides and fragrance compounds like citral, has relied heavily on chemical hydrogenation or reduction processes. These conventional methodologies are plagued by inherent selectivity issues rooted in fundamental chemical principles. Thermodynamically, the activation energy required to reduce a carbon-carbon double bond (C=C) is often lower than that for a carbonyl group (C=O), while kinetically, the C=C bond is frequently more reactive under standard catalytic conditions. Consequently, when general chemical catalysts are employed to reduce alpha,beta-unsaturated aldehydes like prenaldehyde, the major products are often saturated aldehydes or alcohols rather than the desired unsaturated alcohol. This lack of chemoselectivity necessitates complex and costly downstream purification steps to remove impurities, significantly driving up the overall production cost and reducing the final yield of the high-value target molecule. Furthermore, many chemical processes require harsh reaction conditions, including high pressures and temperatures, which pose safety risks and increase energy consumption.
The Novel Approach
In stark contrast to these traditional limitations, the novel approach described in the patent utilizes a recombinant Escherichia coli strain engineered to co-express two specific enzymes: an alkenal reductase derived from Yokenella sp. and a D-glucose dehydrogenase from Exiguobacterium. This dual-enzyme system creates a highly efficient biocatalytic cascade that operates under mild physiological conditions, typically around neutral pH and moderate temperatures. The alkenal reductase exhibits exceptional specificity for the carbonyl group of prenaldehyde, effectively bypassing the thermodynamic preference for C=C reduction observed in chemical catalysis. This results in a reaction profile where the substrate is converted entirely into the desired prenol without the detection of saturated alcohol by-products. Moreover, the integration of glucose dehydrogenase facilitates an in-situ cofactor regeneration cycle, allowing the reaction to proceed continuously without the depletion of essential reducing equivalents. This biological strategy not only enhances product purity but also simplifies the process workflow, making it an attractive option for cost reduction in fine chemical manufacturing.
Mechanistic Insights into Dual-Enzyme Coupled Catalysis
The core of this technological breakthrough lies in the synergistic interaction between the two expressed enzymes within the recombinant host cell. The alkenal reductase acts as the primary catalyst, binding to the prenaldehyde substrate and facilitating the hydride transfer from the reduced cofactor NADPH to the carbonyl carbon. This specific interaction is governed by the enzyme's active site architecture, which sterically and electronically favors the C=O bond over the conjugated C=C bond. Simultaneously, the oxidation of NADPH to NADP+ would normally halt the reaction if not for the presence of the second enzyme, glucose dehydrogenase. This auxiliary enzyme oxidizes the co-substrate, D-glucose, to gluconolactone, concurrently reducing NADP+ back to NADPH. This closed-loop regeneration system ensures a constant supply of the reducing power necessary for the primary reduction reaction, effectively decoupling the process from the need for stoichiometric amounts of expensive external cofactors.
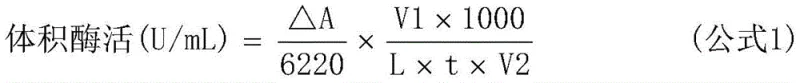
To achieve this dual functionality, the genetic construct was meticulously designed using the pACYCDuet-1 vector, which allows for the independent expression of two target genes under separate promoters. The alkenal reductase gene and the D-glucose dehydrogenase gene were inserted into distinct multiple cloning sites within the plasmid, ensuring balanced expression levels in the E. coli BL21(DE3) host. This genetic stability is crucial for maintaining consistent catalytic performance across large-scale fermentation batches. The successful expression of both proteins was confirmed through SDS-PAGE analysis, demonstrating clear bands corresponding to the molecular weights of both enzymes post-induction. The structural integrity of this plasmid design is fundamental to the robustness of the biocatalyst, enabling it to withstand the stresses of industrial bioprocessing while maintaining high specific activity.

How to Synthesize Prenol Efficiently
Implementing this biocatalytic route requires precise control over fermentation and reaction parameters to maximize yield and productivity. The process begins with the cultivation of the recombinant strain followed by induction with IPTG to trigger enzyme production. Once the biomass is harvested and prepared as either wet cells or lyophilized powder, it serves as the whole-cell biocatalyst for the conversion of prenaldehyde. The reaction is conducted in a buffered aqueous system where glucose is supplied as the sacrificial co-substrate to drive the cofactor cycle. Optimization studies indicate that maintaining a specific molar ratio between glucose and prenaldehyde is critical for sustaining the reaction rate, particularly at higher substrate loadings. The detailed standardized synthesis steps for implementing this protocol are provided in the guide below.
- Construct the recombinant vector by inserting alkenal reductase and D-glucose dehydrogenase genes into the pACYCDuet-1 plasmid and transform into E. coli BL21(DE3).
- Induce protein expression in the host bacteria using IPTG at optimized temperatures (20-22°C) to maximize enzyme activity and stability.
- Perform the biocatalytic reaction using wet cells or lyophilized powder with prenaldehyde and glucose as a co-substrate at pH 7.5 and 45°C to achieve complete conversion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from chemical to this enzymatic synthesis offers profound strategic advantages beyond mere technical feasibility. The elimination of harsh chemical reagents and the reliance on renewable biological feedstocks align perfectly with modern sustainability goals and regulatory compliance standards. By removing the need for precious metal catalysts or high-pressure hydrogenation equipment, the capital expenditure and operational complexity associated with the manufacturing facility are drastically simplified. This reduction in process intensity translates directly into a more resilient supply chain, less susceptible to fluctuations in the availability of specialized chemical reagents or energy costs. Furthermore, the high selectivity of the enzyme minimizes waste generation, reducing the burden on wastewater treatment systems and lowering the environmental footprint of the production site.
- Cost Reduction in Manufacturing: One of the most significant economic drivers of this technology is the self-sufficient cofactor regeneration system. In traditional biocatalysis, the cost of adding stoichiometric amounts of NADPH can be prohibitive for large-scale applications. However, this coupled enzyme system recycles the cofactor continuously using inexpensive glucose, effectively removing a major cost barrier. Additionally, the high regioselectivity means that downstream purification is streamlined, as there are no saturated alcohol impurities to separate, leading to substantial savings in solvent usage and distillation energy. The ability to use whole cells or simple lyophilized powders further reduces the cost associated with enzyme purification and immobilization.
- Enhanced Supply Chain Reliability: The robustness of the recombinant E. coli strain ensures consistent production quality, which is vital for maintaining long-term contracts with pharmaceutical and agrochemical clients. The fermentation process utilizes standard media components and operates under ambient pressure, reducing the risk of equipment failure or safety incidents that could disrupt supply. Moreover, the high substrate tolerance demonstrated by the system, capable of handling concentrations up to 500 mM, allows for higher volumetric productivity. This efficiency means that smaller reactor volumes can produce the same amount of product, optimizing facility utilization and reducing lead time for high-purity prenol deliveries.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is often challenging due to oxygen transfer limitations or shear stress, but this whole-cell system has shown remarkable stability under optimized stirring conditions. The mild reaction conditions (45°C, pH 7.5) are easily manageable in standard stainless steel reactors, facilitating a smooth transition from laboratory to commercial scale-up of complex fine chemicals. From an environmental perspective, the process generates benign by-products like gluconolactone, which are easier to treat than heavy metal residues or organic solvents typical of chemical synthesis. This aligns with increasingly stringent global environmental regulations, future-proofing the supply chain against potential regulatory tightening.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recombinant biocatalytic technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on performance metrics and operational requirements. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing production lines.
Q: How does this biocatalytic method improve selectivity compared to chemical hydrogenation?
A: Unlike chemical catalysts which often reduce the C=C bond due to lower activation energy, this recombinant alkenal reductase specifically targets the C=O bond, ensuring 100% conversion to the valuable alpha,beta-unsaturated alcohol without saturated by-products.
Q: Is the addition of expensive cofactors like NADPH required for this process?
A: No, the process utilizes a coupled enzyme system where glucose dehydrogenase continuously regenerates NADPH from NADP+ using glucose, eliminating the need for costly external cofactor supplementation.
Q: What are the substrate tolerance limits for this enzymatic system?
A: The system demonstrates high robustness, capable of completely converting substrate concentrations up to 500 mM within a short reaction timeframe, indicating strong potential for high-density industrial fermentation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prenol Supplier
The technological potential of the dual-enzyme system described in patent CN110643556A underscores the importance of partnering with a CDMO expert who understands the nuances of biocatalytic scale-up. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale optimization to industrial manufacturing is seamless. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of prenol meets the exacting standards required for pharmaceutical and fragrance applications. We leverage advanced fermentation technologies to maximize the expression of recombinant enzymes, delivering cost-effective solutions without compromising on quality or consistency.
We invite you to collaborate with us to optimize your supply chain for alpha,beta-unsaturated alcohols. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our biocatalytic expertise can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
