Advanced Mycophenolic Acid Purification: Ensuring High Purity for Pharmaceutical Manufacturing
The pharmaceutical industry continuously demands higher purity standards for critical immunosuppressant intermediates, particularly for Mycophenolic Acid (MPA), which serves as the foundational active ingredient for widely prescribed transplant rejection medications. Patent CN115838363A introduces a transformative purification methodology that addresses the persistent challenge of removing the structurally similar Z-isomer impurity, often referred to as Impurity B in European Pharmacopoeia standards. This innovative approach leverages a synergistic combination of pH regulation, specific salt additives, and controlled crystallization conditions to achieve impurity levels significantly below the stringent 0.10% threshold required for clinical-grade materials. For R&D directors and procurement specialists, understanding the technical nuances of this patent is essential for securing a reliable mycophenolic acid supplier capable of delivering consistent quality at scale. The method described herein not only enhances the chemical profile of the final product but also streamlines the downstream processing workflow, offering substantial advantages for commercial-scale manufacturing operations seeking to optimize their supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional purification techniques for mycophenolic acid have historically struggled to effectively discriminate between the target molecule and its Z-isomer counterpart due to their nearly identical physicochemical properties. Conventional methods often rely on simple recrystallization from ethanol or acetone mixtures, sometimes supplemented with activated carbon decolorization, which frequently fails to reduce the Z-isomer content below the critical 0.10% limit mandated by international regulatory bodies. As highlighted in prior art such as CN109020933B and CN103880798A, these older processes often result in residual impurity levels exceeding 0.40%, necessitating additional, costly reprocessing steps that erode profit margins and extend production lead times. Furthermore, the reliance on harsh acidic conditions or non-selective solvents in legacy processes can lead to product degradation or the formation of secondary impurities, complicating the impurity profile and jeopardizing batch consistency. For supply chain heads, these inefficiencies translate into unpredictable yields and potential delays in fulfilling large-volume orders for complex polymer additives or pharmaceutical intermediates, creating significant operational risk for downstream drug manufacturers who depend on uninterrupted material flow.
The Novel Approach
The novel approach detailed in patent CN115838363A overcomes these historical limitations through a meticulously engineered four-step sequence that integrates specific chemical additives to alter the solubility dynamics of the impurity relative to the product. By introducing sodium dihydrogen phosphate during the initial extraction phase and utilizing a sodium bicarbonate wash followed by triethylamine-assisted crystallization, the process creates a chemical environment that selectively favors the precipitation of pure mycophenolic acid while keeping the Z-isomer in solution. This method has been demonstrated to consistently achieve Z-isomer levels as low as 0.02%, representing a dramatic improvement over the 0.43% observed in comparative conventional examples. The strategic use of mild pH adjustments between 4.0 and 5.0 ensures that the product remains stable while maximizing the partition coefficient difference between the target compound and the impurity during the liquid-liquid extraction phase. For procurement managers focused on cost reduction in pharmaceutical intermediate manufacturing, this enhanced selectivity means fewer batches are rejected, raw material utilization is optimized, and the overall cost of goods sold is significantly reduced without compromising on the stringent quality specifications required for global market access.
Mechanistic Insights into Triethylamine-Assisted Crystallization
The core of this purification breakthrough lies in the sophisticated manipulation of molecular interactions during the crystallization phase, where the addition of triethylamine plays a pivotal role in lattice formation and impurity exclusion. The Z-isomer, characterized by a specific geometric configuration around the double bond in the side chain, exhibits slightly different polarity and hydrogen bonding capabilities compared to the E-isomer form of mycophenolic acid, which this process exploits to achieve separation. 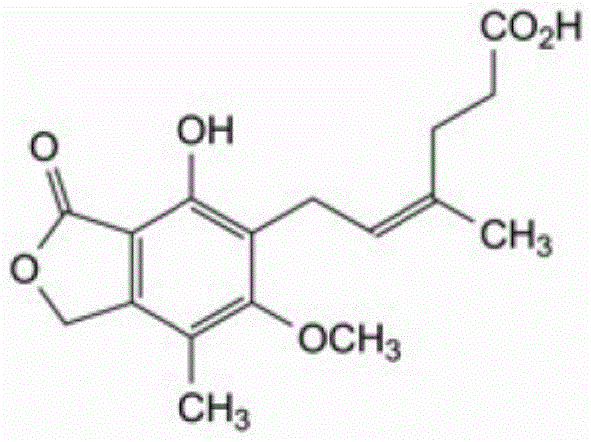 By carefully controlling the concentration of triethylamine between 6% and 10% of the concentrated solution volume, the process modifies the solvent system's basicity, thereby influencing the ionization state of the carboxylic acid group and altering the nucleation kinetics. This precise control prevents the co-crystallization of the Z-isomer, which remains solvated in the mother liquor due to the unfavorable energetic conditions for its incorporation into the growing crystal lattice of the pure product. For R&D directors evaluating the feasibility of this工艺 structure, understanding this mechanism is crucial as it highlights the importance of reagent grade and addition rates in maintaining the robustness of the purification protocol across different batch sizes and reactor configurations.
By carefully controlling the concentration of triethylamine between 6% and 10% of the concentrated solution volume, the process modifies the solvent system's basicity, thereby influencing the ionization state of the carboxylic acid group and altering the nucleation kinetics. This precise control prevents the co-crystallization of the Z-isomer, which remains solvated in the mother liquor due to the unfavorable energetic conditions for its incorporation into the growing crystal lattice of the pure product. For R&D directors evaluating the feasibility of this工艺 structure, understanding this mechanism is crucial as it highlights the importance of reagent grade and addition rates in maintaining the robustness of the purification protocol across different batch sizes and reactor configurations.
Furthermore, the preliminary treatment steps involving sodium dihydrogen phosphate and sodium bicarbonate washing serve as critical pre-conditioning stages that remove bulk fermentation residues and acidic byproducts before the final crystallization occurs. The addition of sodium dihydrogen phosphate at 0.8% to 1.5% of the product mass helps to buffer the aqueous phase during extraction, ensuring that the pH remains within the optimal 4.0 to 5.0 window where mycophenolic acid is most efficiently transferred into the organic phase while polar impurities remain behind. Subsequent washing with a dilute sodium bicarbonate solution further scrubs the organic layer of residual acidic contaminants that could otherwise interfere with the crystallization dynamics or catalyze degradation pathways during the concentration step. This multi-stage purification logic ensures that the feed material entering the crystallizer is of exceptionally high quality, thereby reducing the burden on the final separation step and enhancing the overall purity profile of the resulting crystals. Such rigorous impurity control mechanisms are essential for producing high-purity OLED material or pharmaceutical intermediates where trace contaminants can have outsized effects on downstream reaction performance and final drug safety.
How to Synthesize Mycophenolic Acid Efficiently
Implementing this purification protocol requires strict adherence to the specified operational parameters to ensure the reproducible removal of the Z-isomer and the attainment of pharmaceutical-grade quality standards. The process begins with the filtration of the fermentation broth to remove biomass, followed by the precise addition of buffering agents and extraction solvents that must be managed with high accuracy to maintain the delicate phase equilibrium required for effective separation. Detailed standardized synthesis steps see the guide below, which outlines the critical control points for temperature, pH, and reagent stoichiometry that define the success of this manufacturing route. For technical teams looking to scale this process, it is imperative to validate each unit operation from the initial plate and frame filtration through to the final vacuum drying stage to ensure that the laboratory-scale efficacy translates seamlessly to commercial production volumes.
- Filter fermentation broth, add sodium dihydrogen phosphate, adjust pH to 4.0-5.0, and extract with organic solvent.
- Filter the extract with diatomaceous earth and wash with sodium bicarbonate solution to remove acidic impurities.
- Concentrate under reduced pressure, add triethylamine, and cool to 5-15°C for crystallization.
- Filter the crystals and dry under reduced pressure at 40-55°C until loss on drying is less than 1%.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this advanced purification technology offers profound benefits for procurement managers and supply chain leaders who are tasked with balancing cost efficiency against rigorous quality compliance requirements. The elimination of complex, multi-step recrystallization cycles found in older methods drastically simplifies the manufacturing workflow, leading to reduced energy consumption, lower solvent usage, and decreased labor hours per kilogram of finished product. This streamlining of the production process directly contributes to substantial cost savings in manufacturing, allowing suppliers to offer more competitive pricing structures without sacrificing margin, which is a critical factor for buyers managing tight budgets in the generic pharmaceutical sector. Moreover, the robustness of the method against variations in fermentation broth composition enhances supply chain reliability, ensuring that delivery schedules are met consistently even when upstream biological processes exhibit natural variability.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive chromatographic purification steps or excessive solvent exchanges that characterize less efficient legacy methods, thereby lowering the variable cost per unit significantly. By utilizing recyclable extraction agents like ethyl acetate or n-butyl acetate and minimizing the loss of product to mother liquors through high-yield crystallization, the overall material efficiency is greatly improved. This economic efficiency allows for better resource allocation and investment in other areas of quality assurance, ultimately strengthening the financial health of the supply partnership and providing long-term stability for procurement contracts.
- Enhanced Supply Chain Reliability: The simplicity and controllability of the four-step process reduce the likelihood of batch failures or out-of-specification results, which are common causes of supply disruption in the fine chemical industry. With a method that consistently delivers Z-isomer levels well below the regulatory limit, manufacturers can maintain higher safety stocks and shorter lead times, responding more agilely to fluctuations in market demand for immunosuppressant drugs. This reliability is paramount for supply chain heads who must guarantee the continuity of raw material flow to formulation plants, avoiding costly production stoppages that can ripple through the entire pharmaceutical value chain.
- Scalability and Environmental Compliance: Designed with industrial application in mind, the purification method avoids the use of hazardous reagents or extreme conditions that would complicate waste treatment and environmental permitting. The ability to recycle solvents and the use of common inorganic salts for pH adjustment align with modern green chemistry principles, reducing the environmental footprint of the manufacturing site. This compliance facilitates easier scaling from pilot plant to full commercial production, ensuring that capacity expansions can be executed rapidly to meet growing global demand for high-quality mycophenolic acid without encountering regulatory bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this purification technology, derived directly from the patent specifications and comparative data. Understanding these details helps stakeholders assess the fit of this material within their specific development pipelines and supply strategies.
Q: Why is the Z-isomer difficult to remove from Mycophenolic Acid?
A: The Z-isomer possesses a chemical structure highly similar to mycophenolic acid, making standard separation techniques like simple crystallization ineffective without specific pH and additive modifications.
Q: What is the achieved purity level regarding Z-isomer content?
A: The patented purification method efficiently reduces the Z-isomer content to below 0.10%, meeting stringent pharmacopoeia standards for immunosuppressant intermediates.
Q: Does this process utilize environmentally friendly solvents?
A: Yes, the process allows for the recycling of extraction agents and utilizes common, manageable reagents like ethyl acetate and sodium bicarbonate, supporting green manufacturing goals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mycophenolic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patented laboratory methods to robust commercial production requires deep technical expertise and a commitment to uncompromising quality standards. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the precise conditions required for Z-isomer removal are maintained regardless of batch size. We operate stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every shipment of mycophenolic acid meets the <0.10% Z-isomer criterion, providing our partners with the confidence needed for regulatory filings and clinical trials. Our dedication to process optimization means we continuously refine our manufacturing protocols to enhance yield and consistency, solidifying our position as a trusted partner for global pharmaceutical companies.
We invite you to engage with our technical procurement team to discuss how our advanced purification capabilities can support your specific project requirements and cost objectives. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our efficient manufacturing process translates into tangible economic value for your organization. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to evaluate the suitability of our high-purity mycophenolic acid for your next generation of immunosuppressant formulations with complete transparency and scientific rigor.
