Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Iron Catalysis for Commercial API Production
Introduction to Advanced Quinazolinone Synthesis
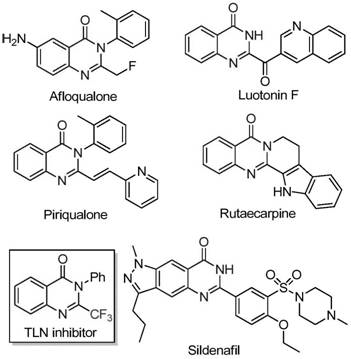
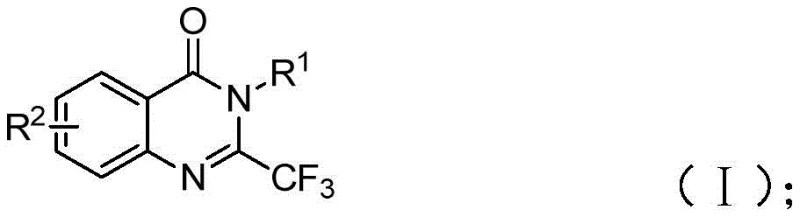
The field of medicinal chemistry continuously seeks efficient pathways to access nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Quinazolinones, in particular, represent a privileged scaffold found in numerous pharmaceutical agents exhibiting anti-cancer, anticonvulsant, and anti-inflammatory properties. The strategic introduction of a trifluoromethyl group into these scaffolds further enhances their metabolic stability and lipophilicity, making them highly desirable targets for drug discovery programs. A significant breakthrough in this domain is documented in patent CN111675662B, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. This technology leverages an iron-catalyzed cyclization strategy that circumvents the limitations of traditional synthetic routes, offering a robust platform for generating high-purity pharmaceutical intermediates.
The significance of this innovation lies in its ability to utilize readily available starting materials while employing an earth-abundant metal catalyst. Unlike conventional methods that often rely on expensive reagents or harsh conditions, this approach utilizes ferric chloride and sodium hydride to drive the formation of the quinazolinone core. For R&D directors and procurement specialists alike, this represents a pivotal shift towards more sustainable and cost-effective manufacturing processes. The versatility of the method allows for the synthesis of a wide array of derivatives, providing a reliable foundation for the development of next-generation therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted quinazolinones has been fraught with challenges that hinder efficient large-scale production. Traditional literature methods typically rely on the cyclization of synthons bearing trifluoromethyl groups, such as trifluoroacetic anhydride or ethyl trifluoroacetate, with substrates like anthranilamide or isatoic anhydride. While chemically feasible, these routes are often plagued by severe reaction conditions that require stringent temperature control and specialized equipment. Furthermore, the substrates employed in these classical approaches can be prohibitively expensive or difficult to source in bulk quantities, creating bottlenecks in the supply chain.
Another critical drawback of conventional methodologies is the narrow substrate scope and generally low yields observed across diverse derivatives. The sensitivity of these reactions to functional groups often necessitates complex protection and deprotection strategies, adding unnecessary steps and reducing overall atom economy. For a procurement manager, these inefficiencies translate directly into higher costs of goods sold (COGS) and extended lead times. The reliance on specific, high-cost trifluoromethylating agents also introduces volatility into the raw material pricing structure, making long-term budget forecasting difficult for manufacturing teams.
The Novel Approach
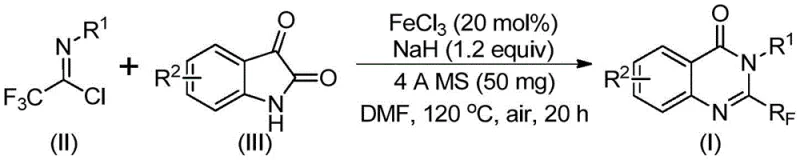
In stark contrast to these legacy methods, the technology described in patent CN111675662B introduces a streamlined and economically viable alternative. By utilizing trifluoroethylimidoyl chloride and isatin as the primary building blocks, the process accesses the quinazolinone core through a direct and efficient pathway. The use of ferric chloride as a catalyst is a game-changer; it is not only significantly cheaper than noble metals like palladium or rhodium but also exhibits remarkable catalytic activity under relatively mild conditions. This novel approach effectively bypasses the need for expensive activated esters or anhydrides, drastically simplifying the raw material portfolio required for production.
The operational simplicity of this new method cannot be overstated. The reaction proceeds in common organic solvents like DMF and tolerates exposure to air, eliminating the need for rigorous inert atmosphere techniques that often complicate scale-up. The protocol involves a two-stage heating profile, initially at 40°C followed by heating to 120°C, which ensures high conversion rates while minimizing side reactions. This robustness allows for the synthesis of a broad range of derivatives with varying electronic properties, demonstrating excellent functional group tolerance that was previously difficult to achieve. Consequently, this method stands out as a superior choice for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process further. The reaction is believed to proceed through an initial alkali-promoted formation of carbon-nitrogen bonds between the trifluoroethylimidoyl chloride and the isatin substrate. This step generates a trifluoroacetamidine intermediate, which serves as the precursor for the subsequent cyclization event. The presence of sodium hydride is critical here, acting as a strong base to deprotonate the isatin nitrogen, thereby facilitating the nucleophilic attack on the imidoyl chloride. This initial coupling is efficient and sets the stage for the ring-closing step that defines the quinazolinone architecture.
Following the formation of the amidine intermediate, the iron catalyst plays a pivotal role in driving the decarbonylation and cyclization sequence. The ferric chloride likely coordinates with the carbonyl oxygen of the isatin moiety, activating it towards nucleophilic attack or facilitating the loss of carbon monoxide. This iron-catalyzed decarbonylation is the key differentiator of this method, allowing for the construction of the fused heterocyclic system without the need for pre-functionalized leaving groups. The result is an isomerization that yields the final 2-trifluoromethyl-substituted quinazolinone with high regioselectivity. This mechanistic pathway not only explains the high yields observed but also highlights the unique reactivity profile enabled by the iron catalyst.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural parameters to ensure optimal results. The patent outlines a clear protocol that balances reagent stoichiometry with thermal management to maximize yield and purity. The process is designed to be user-friendly, utilizing standard glassware and heating equipment found in most synthetic chemistry facilities. By following the established guidelines, chemists can reliably produce target compounds with minimal optimization effort, making it an ideal candidate for rapid library generation or process development.
- Charge a reaction vessel with ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in DMF solvent.
- Stir the mixture at 40°C for 8-10 hours to initiate the reaction, then heat to 120°C and maintain for 18-20 hours under air atmosphere.
- Upon completion, filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iron-catalyzed methodology offers profound benefits for supply chain stability and cost management. The shift away from precious metal catalysts and exotic reagents towards commodity chemicals fundamentally alters the economic landscape of producing these valuable intermediates. For procurement managers, this translates to a more resilient supply chain that is less susceptible to geopolitical fluctuations affecting rare metal prices. The simplicity of the workup procedure, involving basic filtration and chromatography, further reduces the operational overhead associated with purification, leading to faster throughput and lower labor costs.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with ferric chloride represents a direct and significant reduction in raw material costs. Ferric chloride is an abundant, industrial-grade chemical that costs a fraction of palladium or platinum complexes. Additionally, the use of isatin and trifluoroethylimidoyl chloride as starting materials leverages widely available feedstock, avoiding the premium pricing associated with specialized trifluoromethylating agents. This combination of cheap catalyst and accessible substrates drives down the overall cost of goods, enabling more competitive pricing for the final API or intermediate.
- Enhanced Supply Chain Reliability: The reliance on commercially available reagents ensures a consistent and reliable supply of inputs for production. Unlike proprietary reagents that may have single-source suppliers, the key components of this reaction—such as DMF, sodium hydride, and iron salts—are produced by multiple global vendors. This diversification mitigates the risk of supply disruptions and allows for flexible sourcing strategies. Furthermore, the reaction's tolerance to air and moisture simplifies logistics, as strict anhydrous or inert conditions are not required for every step, reducing the complexity of storage and handling.
- Scalability and Environmental Compliance: The environmental profile of this process is markedly improved compared to traditional methods. The use of iron, a non-toxic metal, eliminates the need for extensive heavy metal scavenging steps, which are often required to meet strict regulatory limits for residual metals in pharmaceuticals. This simplification of the purification train reduces solvent consumption and waste generation. Moreover, the high atom economy and efficient conversion rates minimize the formation of byproducts, aligning with green chemistry initiatives and reducing the burden on waste treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 2-trifluoromethyl quinazolinones. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation. They serve to clarify the practical aspects of implementing this technology in an industrial setting.
Q: What are the key advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) offers a substantial cost advantage over precious metal catalysts often used in heterocycle synthesis. Furthermore, iron is environmentally benign, reducing the burden of heavy metal removal and waste disposal, which aligns with green chemistry principles in modern API manufacturing.
Q: Can this synthesis method tolerate diverse functional groups on the substrate?
A: Yes, the protocol demonstrates excellent functional group tolerance. The patent data confirms successful synthesis with substrates containing electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens (F, Cl, Br, I) and nitro groups, yielding products with high purity.
Q: Is this process suitable for large-scale industrial production?
A: The method is highly scalable due to the use of inexpensive, commercially available raw materials like isatin and trifluoroacetimidoyl chlorides. The reaction conditions (using air atmosphere and standard solvents like DMF) and simple post-treatment (filtration and chromatography) facilitate easy scale-up from gram to kilogram levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed synthesis for the pharmaceutical industry. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this patented methodology into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from the bench to the plant. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to collaborate with us to leverage this cost-effective and scalable technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing excellence can accelerate your path to market while optimizing your production costs.
