Advanced Photocatalytic Synthesis of Alpha Alpha Difluoro Gamma Hydroxyacetamide Derivatives for Pharmaceutical Applications
Introduction to Next-Generation Fluorinated Intermediate Synthesis
The integration of fluorine atoms into organic scaffolds represents a cornerstone strategy in modern medicinal chemistry, profoundly influencing the lipophilicity, metabolic stability, and bioavailability of drug candidates. Patent CN112574056A discloses a groundbreaking synthetic methodology for producing alpha,alpha-difluoro-gamma-hydroxyacetamide derivatives, a class of compounds with significant potential as key intermediates in the development of protease inhibitors and nucleotide analogs. This innovation leverages visible-light photocatalysis to achieve the simultaneous introduction of difluoro and hydroxyl groups across olefinic bonds, marking a substantial departure from traditional thermal or stoichiometric oxidant-dependent protocols. By utilizing light as the primary energy source, this process not only enhances safety profiles by eliminating hazardous reagents but also aligns with the growing industry demand for sustainable and green manufacturing practices. For R&D directors and procurement specialists alike, understanding the nuances of this technology is critical for securing reliable supply chains of high-value fluorinated building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of difluoroalkylated alcohols and amides has relied heavily on harsh reaction conditions that pose significant challenges for large-scale industrial application. Traditional routes often necessitate the use of strong oxidants, cryogenic temperatures, or expensive transition metal catalysts that require rigorous removal steps to meet pharmaceutical purity standards. Furthermore, many existing methods exhibit limited substrate tolerance, frequently failing when applied to unactivated aliphatic olefins or sterically hindered aromatic systems, thereby restricting the chemical space available for drug discovery. The reliance on stoichiometric amounts of hazardous reagents also generates substantial waste streams, complicating environmental compliance and driving up disposal costs for manufacturing facilities. These inherent inefficiencies create bottlenecks in the supply chain, leading to extended lead times and volatile pricing for critical fluorinated intermediates needed in the production of active pharmaceutical ingredients.
The Novel Approach
In stark contrast to these legacy techniques, the method described in CN112574056A utilizes a mild, visible-light-driven protocol that operates efficiently at room temperature under an air atmosphere. This approach employs readily available organic photocatalysts such as Rhodamine 6G or fac-Ir(ppy)3 in conjunction with simple organic bases like PMDETA to facilitate the radical difluoroalkylation of diverse olefin derivatives. The reaction proceeds with remarkable selectivity, tolerating a wide array of functional groups including halogens, ethers, and alkyl chains without the need for protective group strategies. 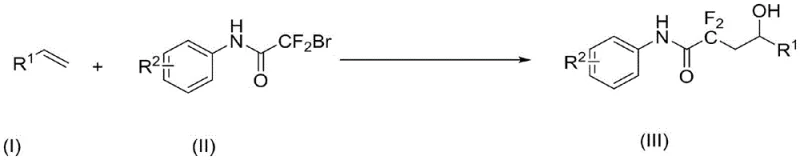 By dissolving the bromodifluoroacetanilide precursor directly in the olefin substrate which acts as both reactant and solvent, the process simplifies the reaction setup and minimizes solvent waste. This streamlined workflow not only accelerates the timeline from bench to pilot scale but also significantly reduces the operational complexity associated with handling sensitive organometallic reagents, offering a robust alternative for the commercial production of complex fluorinated motifs.
By dissolving the bromodifluoroacetanilide precursor directly in the olefin substrate which acts as both reactant and solvent, the process simplifies the reaction setup and minimizes solvent waste. This streamlined workflow not only accelerates the timeline from bench to pilot scale but also significantly reduces the operational complexity associated with handling sensitive organometallic reagents, offering a robust alternative for the commercial production of complex fluorinated motifs.
Mechanistic Insights into Visible-Light Mediated Difluoroalkylation
The core of this transformative synthesis lies in the photoredox catalytic cycle initiated by the absorption of visible light by the photocatalyst, which generates a highly reactive radical species capable of abstracting the bromine atom from the bromodifluoroacetanilide precursor. This homolytic cleavage produces an alpha-amido alkyl radical that subsequently adds across the carbon-carbon double bond of the olefin substrate with high regioselectivity, forming a new carbon-carbon bond and a stabilized benzylic or alkyl radical intermediate. The presence of molecular oxygen from the air atmosphere plays a crucial role in the subsequent oxidation steps, facilitating the conversion of the carbon-centered radical into a cationic or peroxyl species that is eventually trapped by water or hydroxide sources to install the gamma-hydroxyl functionality. 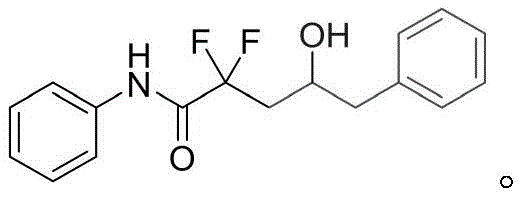 This elegant cascade avoids the use of external oxidants and leverages ambient conditions to drive the reaction forward, demonstrating exceptional atom economy. The mechanistic pathway ensures that the difluoro methylene group is precisely positioned adjacent to the carbonyl, preserving the electronic properties essential for the biological activity of the final drug molecule while minimizing the formation of undesired side products or isomers.
This elegant cascade avoids the use of external oxidants and leverages ambient conditions to drive the reaction forward, demonstrating exceptional atom economy. The mechanistic pathway ensures that the difluoro methylene group is precisely positioned adjacent to the carbonyl, preserving the electronic properties essential for the biological activity of the final drug molecule while minimizing the formation of undesired side products or isomers.
From an impurity control perspective, the mildness of the reaction conditions inherently suppresses common degradation pathways such as polymerization of the olefin or over-oxidation of the alcohol moiety. The use of organic photocatalysts eliminates the risk of heavy metal contamination, a critical quality attribute for pharmaceutical intermediates that often requires costly scavenging resins or recrystallization steps to address. Furthermore, the high chemoselectivity observed across various substrates, including those with electron-withdrawing or electron-donating substituents on the aromatic rings, indicates a robust tolerance that simplifies downstream purification. The reaction system's ability to proceed in the olefin itself as a solvent further reduces the complexity of the mixture, allowing for straightforward isolation of the target alpha,alpha-difluoro-gamma-hydroxyacetamide derivatives through standard extraction and chromatographic techniques. This level of control over the reaction trajectory provides R&D teams with the confidence to scale the process without fearing unpredictable impurity profiles that could derail regulatory filings.
How to Synthesize Alpha Alpha Difluoro Gamma Hydroxyacetamide Efficiently
Implementing this photocatalytic protocol in a laboratory or pilot plant setting requires careful attention to light source intensity and reagent ratios to maximize yield and throughput. The patent details a standardized procedure where the bromodifluoroacetanilide compound is combined with a catalytic amount of Rhodamine 6G and an organic base in the presence of the olefin derivative, followed by irradiation with white or blue LED lights for a duration ranging from 5 to 24 hours depending on the specific substrate reactivity. Detailed standardized synthesis steps see the guide below.
- Dissolve bromodifluoroacetanilide, photocatalyst (e.g., Rhodamine 6G), and organic base (e.g., PMDETA) in the olefin derivative solvent.
- Irradiate the reaction mixture with visible light (white or blue) at room temperature under an air atmosphere for 5 to 24 hours.
- Perform post-treatment by extracting with dichloromethane, drying over sodium sulfate, and purifying via column chromatography to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers tangible benefits that extend beyond mere chemical novelty, directly impacting the bottom line through operational efficiency and risk mitigation. The elimination of expensive transition metal catalysts and hazardous stoichiometric oxidants translates into a significant reduction in raw material costs and waste disposal expenses, creating a more economically viable manufacturing model. Moreover, the ability to run reactions at room temperature under air atmosphere removes the need for specialized high-pressure equipment or cryogenic cooling infrastructure, thereby lowering capital expenditure requirements for facility upgrades. This accessibility allows for greater flexibility in sourcing production capacity, as the process can be easily adapted to existing multipurpose reactors equipped with simple lighting arrays, ensuring continuity of supply even during market fluctuations.
- Cost Reduction in Manufacturing: The shift from thermal or metal-catalyzed processes to visible-light photocatalysis fundamentally alters the cost structure of producing fluorinated intermediates by removing the dependency on precious metals like palladium or iridium in stoichiometric quantities. Since the photocatalyst is used in minute catalytic loads and can potentially be recovered or replaced with inexpensive organic dyes like Rhodamine 6G, the direct material cost per kilogram of product is drastically lowered. Additionally, the simplified workup procedure involving basic extraction and chromatography reduces the consumption of solvents and consumables, further enhancing the overall cost efficiency of the manufacturing campaign. These cumulative savings allow suppliers to offer more competitive pricing for high-purity pharmaceutical intermediates without compromising on quality or margin.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against variations in substrate structure ensures a stable supply of diverse intermediates required for parallel drug development programs. Because the reagents involved, such as bromodifluoroacetanilides and common olefins, are commercially available and do not require complex custom synthesis, the risk of raw material shortages is minimized. The mild reaction conditions also mean that the process is less prone to runaway exotherms or safety incidents, reducing the likelihood of unplanned production shutdowns due to safety audits or accidents. This reliability is crucial for maintaining just-in-time inventory levels and meeting the tight deadlines imposed by clinical trial timelines in the pharmaceutical sector.
- Scalability and Environmental Compliance: Scaling photocatalytic reactions has historically been challenging due to light penetration issues, but advancements in flow chemistry and reactor design have made it feasible to translate these batch processes to continuous manufacturing environments. The green nature of using light as a reagent and air as an oxidant aligns perfectly with increasingly stringent environmental regulations regarding volatile organic compound emissions and heavy metal discharge. By adopting this method, manufacturers can demonstrate a commitment to sustainability, which is becoming a key differentiator in supplier selection criteria for major multinational corporations. The ease of scaling from gram to kilogram quantities without re-optimizing reaction parameters ensures a smooth transition from process development to commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this photocatalytic synthesis method, providing clarity for technical teams evaluating its feasibility for their specific projects. These answers are derived directly from the experimental data and embodiments presented in the patent documentation, ensuring accuracy and relevance to practical applications. Understanding these details is essential for making informed decisions about process adoption and resource allocation.
Q: What are the optimal photocatalysts for this difluoroalkylation reaction?
A: The patent specifies Rhodamine 6G and fac-Ir(ppy)3 as effective photocatalysts, with Rhodamine 6G being preferred for its cost-effectiveness and high yield performance under white light irradiation.
Q: Can this method accommodate diverse olefin substrates?
A: Yes, the method demonstrates broad substrate scope, successfully reacting with styrene derivatives, aliphatic olefins like 1-octene, and various substituted allylbenzenes to produce corresponding hydroxyacetamides.
Q: How is the product purified after the photocatalytic reaction?
A: Post-reaction processing involves aqueous workup followed by extraction with dichloromethane or ethyl acetate, drying, and final purification using column chromatography with a petroleum ether and ethyl acetate gradient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha Alpha Difluoro Gamma Hydroxyacetamide Supplier
As the pharmaceutical industry continues to demand more sophisticated fluorinated building blocks, NINGBO INNO PHARMCHEM stands ready to support your development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle photocatalytic processes safely and efficiently, ensuring that we can meet your volume requirements while adhering to stringent purity specifications and rigorous QC labs. We understand the critical nature of supply chain continuity and are committed to delivering high-quality intermediates that enable your team to focus on innovation rather than sourcing challenges. By leveraging our technical expertise in green chemistry and process optimization, we help you navigate the complexities of bringing new fluorinated drugs to market faster and more cost-effectively.
We invite you to contact our technical procurement team to discuss your specific requirements and request a Customized Cost-Saving Analysis tailored to your project needs. Our experts are available to provide specific COA data and route feasibility assessments to ensure that our manufacturing capabilities align perfectly with your quality and timeline expectations. Partnering with us means gaining access to a reliable supply of advanced intermediates backed by a commitment to excellence and continuous improvement in chemical manufacturing.
