Scalable Photocatalytic Synthesis of Alpha Alpha Difluoro Gamma Hydroxyacetamide Derivatives for Pharma
Scalable Photocatalytic Synthesis of Alpha Alpha Difluoro Gamma Hydroxyacetamide Derivatives for Pharma
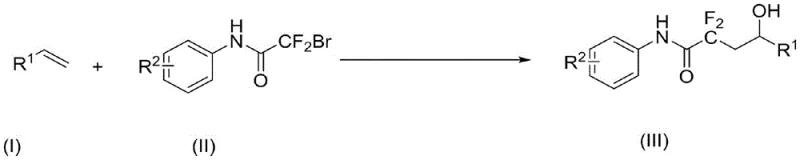
The pharmaceutical industry continuously seeks innovative synthetic methodologies to access complex fluorinated scaffolds, which are critical for enhancing the metabolic stability and bioavailability of modern drug candidates. Patent CN112574056B introduces a groundbreaking approach for the synthesis of alpha,alpha-difluoro-gamma-hydroxyacetamide derivatives, leveraging visible light photocatalysis to achieve efficient olefin difunctionalization. This technology represents a significant paradigm shift from traditional thermal methods, offering a greener, safer, and more economically viable pathway for producing high-value pharmaceutical intermediates. By utilizing bromodifluoroacetanilide compounds and simple olefin derivatives under mild conditions, this process enables the simultaneous introduction of difluoro and hydroxyl groups with exceptional regioselectivity. The strategic application of this patent allows manufacturers to bypass harsh reaction conditions, thereby minimizing side reactions and simplifying downstream purification processes.
For R&D directors and process chemists, the ability to functionalize diverse olefin substrates, ranging from simple aliphatic chains to complex aromatic systems, opens new avenues for library synthesis and lead optimization. The methodology described in CN112574056B not only expands the chemical space accessible for drug discovery but also provides a robust platform for the sustainable manufacturing of fluorinated building blocks. As regulatory pressures mount for greener chemical processes, adopting such photochemical technologies positions forward-thinking organizations at the forefront of sustainable pharmaceutical innovation, ensuring compliance with evolving environmental standards while maintaining high productivity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of difluoroalkyl alcohol compounds and related derivatives has relied heavily on multi-step sequences involving harsh reagents and extreme reaction conditions. Traditional approaches often necessitate the use of strong oxidants, cryogenic temperatures, or expensive transition metal catalysts that pose significant safety hazards and environmental burdens. Furthermore, existing literature predominantly focuses on activated olefins such as styrenes, severely limiting the substrate scope and excluding many valuable aliphatic olefins commonly found in drug scaffolds. These conventional methods frequently suffer from poor atom economy, generating substantial amounts of toxic waste that require complex disposal protocols. The reliance on thermal activation also increases energy consumption and the risk of thermal runaway, making scale-up challenging and costly for industrial applications. Consequently, there is an urgent need for milder, more versatile synthetic strategies that can accommodate a broader range of substrates without compromising safety or efficiency.
The Novel Approach
The novel photocatalytic method disclosed in patent CN112574056B addresses these critical limitations by employing visible light as the sole energy source to drive the reaction at ambient temperature. This approach utilizes inexpensive organic photocatalysts like Rhodamine 6G or metal complexes such as fac-Ir(ppy)3 to generate reactive radical species under mild conditions, eliminating the need for external heating or cooling systems. By dissolving bromodifluoroacetanilide and an organic base directly in the olefin derivative, the process achieves a highly convergent synthesis that minimizes solvent usage and operational steps. The reaction proceeds efficiently under an air atmosphere, demonstrating remarkable tolerance to oxygen and moisture, which simplifies reactor setup and reduces the need for inert gas purging. This methodology successfully extends the substrate scope to include unactivated aliphatic olefins and various substituted aryl groups, providing a versatile tool for constructing complex fluorinated architectures. The result is a streamlined, cost-effective process that aligns perfectly with the principles of green chemistry and modern manufacturing requirements.
Mechanistic Insights into Visible Light Photocatalytic Difunctionalization
The core of this transformative synthesis lies in the intricate photoredox catalytic cycle initiated by the absorption of visible light by the photocatalyst. Upon irradiation, the photocatalyst enters an excited state, facilitating a single-electron transfer (SET) process with the bromodifluoroacetanilide substrate. This interaction generates a crucial carbon-centered radical species by cleaving the carbon-bromine bond, which then adds selectively across the double bond of the olefin derivative. The resulting radical intermediate undergoes further oxidation and subsequent nucleophilic attack by water or hydroxide sources present in the system, leading to the formation of the gamma-hydroxy functionality. This radical-mediated pathway ensures high regioselectivity, placing the difluoroacetamide group at the terminal position and the hydroxyl group at the internal position relative to the original alkene. The use of organic bases like PMDETA plays a pivotal role in stabilizing intermediates and facilitating the proton transfer steps necessary for product formation. Understanding this mechanism allows chemists to fine-tune reaction parameters, such as light wavelength and catalyst loading, to maximize yield and minimize byproduct formation.
From an impurity control perspective, the mild nature of the photocatalytic conditions significantly suppresses common side reactions associated with thermal degradation or over-oxidation. Unlike traditional methods that might produce polymeric byproducts or rearranged isomers due to high energy inputs, this light-driven process maintains a controlled energy profile that favors the desired transformation. The specific choice of photocatalyst and base creates a selective environment where the radical addition occurs rapidly and irreversibly, preventing competing pathways that could lead to structural impurities. Additionally, the reaction's tolerance to air suggests that oxidative side products are minimized, likely due to the rapid consumption of radical intermediates by the intended reaction pathway. This inherent selectivity translates to a cleaner crude reaction mixture, reducing the burden on purification steps such as column chromatography. For quality assurance teams, this means a more consistent impurity profile and easier validation of the manufacturing process, ensuring that the final pharmaceutical intermediates meet stringent purity specifications required for clinical applications.
How to Synthesize Alpha,alpha-difluoro-gamma-hydroxyacetamide Efficiently
Implementing this photocatalytic protocol requires careful attention to reagent stoichiometry and light source selection to ensure optimal conversion rates. The standard procedure involves dissolving the bromodifluoroacetanilide compound, a catalytic amount of Rhodamine 6G, and an organic base such as PMDETA directly into the olefin derivative, which serves as both reactant and solvent. The reaction mixture is then subjected to irradiation using a standard white or blue LED light source at room temperature, typically requiring between 5 to 24 hours to reach completion depending on the substrate's electronic properties. Following the reaction, a straightforward workup involving aqueous extraction and drying yields the crude product, which can be purified via column chromatography using petroleum ether and ethyl acetate mixtures. This simplified workflow eliminates the need for specialized high-pressure equipment or cryogenic setups, making it accessible for most synthetic laboratories. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results accurately.
- Dissolve bromodifluoroacetanilide, photocatalyst (Rhodamine 6G), and organic base (PMDETA) in the olefin derivative solvent.
- Irradiate the reaction mixture with visible light (white or blue) at room temperature for 5 to 24 hours under air atmosphere.
- Perform post-treatment including aqueous workup, extraction with dichloromethane, drying, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this photocatalytic technology offers substantial strategic benefits that extend beyond mere chemical efficiency. The shift from thermal to photochemical activation fundamentally alters the cost structure of manufacturing by drastically reducing energy consumption associated with heating and cooling large-scale reactors. Since the reaction proceeds at room temperature, facilities can avoid the capital expenditure and maintenance costs linked to complex temperature control systems, leading to significant operational savings over the lifecycle of the product. Furthermore, the use of visible light as a renewable energy input aligns with corporate sustainability goals, potentially qualifying the manufacturing process for green incentives or carbon credits. The simplicity of the reaction setup also reduces the demand for specialized labor and training, allowing existing personnel to manage production with greater ease and flexibility. These factors collectively contribute to a more resilient and cost-competitive supply chain capable of responding swiftly to market demands.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of readily available organic photocatalysts like Rhodamine 6G significantly lower the raw material costs per kilogram of product. By avoiding harsh reagents and extreme conditions, the process reduces the wear and tear on reactor vessels and ancillary equipment, extending their operational lifespan and minimizing maintenance downtime. The high atom economy of the difunctionalization reaction ensures that a larger proportion of starting materials are converted into the desired product, reducing waste disposal fees and maximizing resource utilization. Additionally, the simplified purification process lowers the consumption of solvents and stationary phases, further driving down the overall cost of goods sold. These cumulative savings enhance profit margins and allow for more competitive pricing strategies in the global pharmaceutical market.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as bromodifluoroacetanilides and common olefins, mitigates the risk of supply disruptions caused by scarce or regulated reagents. The robustness of the reaction under air atmosphere removes the dependency on high-purity inert gases, simplifying logistics and reducing the complexity of inventory management. Because the process is scalable and does not require specialized infrastructure, it can be easily transferred between different manufacturing sites or contracted manufacturers without significant requalification efforts. This flexibility ensures continuous supply continuity even in the face of regional disruptions or facility maintenance schedules. Moreover, the shorter reaction times compared to multi-step thermal sequences accelerate the production cycle, enabling faster turnaround times for customer orders and improving overall service levels.
- Scalability and Environmental Compliance: The inherent safety of operating at room temperature and atmospheric pressure makes this process highly scalable from laboratory benchtop to industrial tonnage production without encountering the thermal hazards typical of exothermic reactions. The green chemistry credentials of using visible light and generating minimal hazardous waste facilitate easier regulatory approval and compliance with increasingly strict environmental protection laws. Reduced solvent usage and the potential for solvent recycling further diminish the environmental footprint, supporting corporate initiatives for sustainable development. The ability to handle diverse substrates within the same operational framework allows for flexible production lines that can switch between different product variants with minimal changeover time. This adaptability is crucial for meeting the dynamic needs of the pharmaceutical industry while maintaining a commitment to environmental stewardship and operational excellence.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These insights are derived directly from the experimental data and beneficial effects outlined in patent CN112574056B, providing clarity on process feasibility and performance. Understanding these aspects is essential for stakeholders evaluating the technology for integration into their existing manufacturing portfolios. The answers reflect the practical realities of scaling photochemical processes and the specific advantages offered by this novel approach.
Q: What are the key advantages of this photocatalytic method over traditional thermal methods?
A: This method utilizes visible light as a clean energy source, operating at room temperature which significantly enhances safety and reduces energy consumption compared to high-temperature thermal processes. It also demonstrates superior substrate tolerance, accommodating both aliphatic and aromatic olefins effectively.
Q: Which photocatalysts are most effective for this transformation?
A: The patent identifies Rhodamine 6G and fac-Ir(ppy)3 as highly effective photocatalysts, with Rhodamine 6G being particularly preferred due to its cost-effectiveness and high efficiency in generating the necessary radical species under visible light irradiation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability, utilizing readily available raw materials and mild reaction conditions that simplify reactor requirements. The absence of extreme temperatures or pressures makes it highly adaptable for commercial scale-up in pharmaceutical manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,alpha-difluoro-gamma-hydroxyacetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photocatalytic synthesis method described in CN112574056B for producing high-quality fluorinated pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with advanced photochemical reactors capable of delivering uniform light distribution and precise process control, guaranteeing consistent product quality and batch-to-batch reproducibility. We adhere to stringent purity specifications and operate rigorous QC labs to verify that every shipment meets the highest international standards for pharmaceutical ingredients. Our team of expert chemists is ready to optimize this green synthesis route specifically for your target molecules, leveraging our deep understanding of photoredox catalysis to maximize yield and efficiency.
We invite you to collaborate with us to unlock the full commercial potential of this innovative technology for your drug development programs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that details how implementing this photocatalytic route can reduce your overall manufacturing expenses. We encourage you to contact our technical procurement team today to request specific COA data for relevant intermediates and discuss detailed route feasibility assessments tailored to your unique requirements. Let us help you build a more sustainable, efficient, and reliable supply chain for your next-generation fluorinated therapeutics, ensuring your success in the competitive global marketplace.
