Advanced DCC-Free Synthesis Strategy for High Purity Perindopril Intermediates and Commercial Scalability
Advanced DCC-Free Synthesis Strategy for High Purity Perindopril Intermediates and Commercial Scalability
The pharmaceutical industry constantly seeks robust synthetic pathways that guarantee the highest levels of purity for active pharmaceutical ingredients (APIs), particularly for chronic medications like ACE inhibitors. Patent CN1622936A introduces a groundbreaking methodology for the preparation of high-purity perindopril and its tert-butylamine salt, specifically addressing the critical issue of contamination derived from dicyclohexylcarbodiimide (DCC). This intellectual property outlines a novel route that bypasses traditional coupling agents known to generate stubborn urea byproducts, thereby offering a significant leap forward in process chemistry. For R&D directors and procurement specialists, this technology represents a viable solution to long-standing purification bottlenecks, ensuring that the final drug substance meets stringent regulatory standards without the need for excessive downstream processing. The core innovation lies in the strategic use of thionyl chloride for activating N-alkoxycarbonyl amino acids, a approach that fundamentally alters the impurity profile of the synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial-scale synthesis of perindopril has relied heavily on peptide coupling strategies involving dicyclohexylcarbodiimide (DCC) and 1-hydroxybenzotriazole (HOBt). While effective for forming amide bonds, this conventional approach suffers from a severe drawback: the formation of dicyclohexylurea (DCU) and related structural analogs that act as persistent contaminants. As detailed in the background art, the reaction of formula V compounds with formula II compounds in the presence of DCC inevitably leads to the generation of benzyl ester contaminants designated as formulas VII and VIII. These impurities are chemically similar to the desired product and undergo debenzylation to form formulas VII' and VIII', which are notoriously difficult to separate from the final API. The presence of these DCC-derived pollutants necessitates complex and costly purification sequences, often involving multiple recrystallizations or chromatographic steps, which drastically reduce overall yield and increase manufacturing costs. Furthermore, the removal of DCU itself can be mechanically challenging on a large scale, creating operational inefficiencies that hinder supply chain reliability.
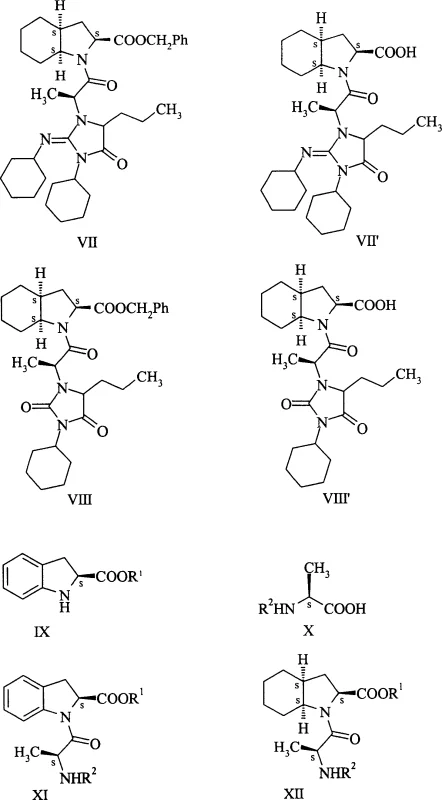
The Novel Approach
In stark contrast to the legacy methods, the process described in CN1622936A eliminates the use of carbodiimides entirely, replacing them with a more efficient activation strategy using carbonic acid derivatives and thionyl chloride. This novel approach begins with the acylation of the starting amino acid using agents like ethyl chloroformate or di-tert-butyl dicarbonate to form stable N-protected intermediates (Formula III). By shifting the activation mechanism to thionyl chloride, the process avoids the formation of non-volatile urea byproducts altogether. The resulting activated species reacts cleanly with perhydroindole-2-carboxylic acid (Formula IV) to form the peptide bond in a single step. This methodological shift not only simplifies the reaction workflow but also ensures that the final perindopril product is inherently free from the specific contaminants associated with DCC chemistry. For a reliable pharmaceutical intermediate supplier, adopting this route means delivering a product with a superior impurity profile right from the crude stage, significantly reducing the burden on quality control laboratories.
Mechanistic Insights into Thionyl Chloride-Mediated Peptide Bond Formation
The mechanistic elegance of this invention lies in the successful application of thionyl chloride to activate N-alkoxycarbonyl amino acids, a transformation that was previously considered risky due to the potential for over-reaction or racemization. In this specific catalytic cycle, the carboxylic acid group of the N-protected intermediate (Formula III) reacts with thionyl chloride to form a highly reactive acid chloride species in situ. Unlike traditional Schotten-Baumann conditions which require aqueous alkaline environments, this reaction proceeds efficiently in inert organic solvents such as dichloromethane or tetrahydrofuran at temperatures ranging from 0°C to 30°C. The activation step generates hydrogen chloride and sulfur dioxide as byproducts, both of which are gases that readily escape the reaction mixture or are easily removed under vacuum. This volatility is a crucial mechanistic advantage, as it drives the equilibrium forward and leaves no trace residues in the reaction vessel. Subsequently, the activated acid chloride couples with the nucleophilic amine of the perhydroindole derivative to form the target amide bond. Surprisingly, despite the high reactivity of acid chlorides, the process maintains stereochemical integrity and does not lead to the degradation of the sensitive alkoxy-carbonyl protecting groups during the peptide formation step.
From an impurity control perspective, this mechanism offers a distinct advantage over carbodiimide-mediated couplings. In DCC reactions, the O-acylisourea intermediate can rearrange to form unreactive N-acylureas, or react with the added HOBt to form esters that may lead to racemization. By utilizing the thionyl chloride pathway, the reaction bypasses these complex equilibrium states. The direct conversion to the acid chloride ensures a clean electrophilic attack on the amine partner. Furthermore, the absence of bulky urea molecules means that the crystallization behavior of the final product is not disrupted by lattice inclusion of impurities. This results in a cleaner crystal structure for the perindopril tert-butylamine salt, which is essential for meeting the rigorous pharmacopeial standards required for cardiovascular medications. The ability to perform this coupling without the need for additional coupling additives like HOBt further streamlines the material input, reducing the complexity of the bill of materials and minimizing the risk of introducing foreign particulate matter into the batch.
How to Synthesize Perindopril Intermediates Efficiently
The implementation of this synthetic route requires precise control over reaction parameters to maximize yield and purity while maintaining safety standards. The process is divided into three critical stages: the initial protection of the amino acid, the activation of the carboxyl group, and the final coupling and salt formation. Operators must ensure that the acylation step is conducted at controlled low temperatures (0-5°C) to prevent side reactions, followed by a careful workup to isolate the N-protected intermediate. The subsequent activation with thionyl chloride must be managed to ensure complete conversion while safely handling the evolved gases. Finally, the coupling reaction with perhydroindole-2-carboxylic acid is performed under reflux to drive the reaction to completion, followed by a straightforward salt formation with tert-butylamine. Detailed standardized operating procedures for these steps are essential for technology transfer and scale-up.
- Acylation of the starting amino acid with a suitable carbonic acid derivative (such as ethyl chloroformate) in the presence of a base to form the N-alkoxycarbonyl protected intermediate.
- Activation of the resulting N-protected acid using thionyl chloride in an inert organic solvent like dichloromethane to generate the reactive acid chloride species.
- Coupling the activated acid chloride with perhydroindole-2-carboxylic acid under reflux conditions, followed by salt formation with tert-butylamine to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DCC-free synthesis route offers transformative economic and logistical benefits that extend far beyond simple yield improvements. The primary value driver is the drastic simplification of the purification train. By eliminating the formation of tenacious urea contaminants, manufacturers can significantly reduce the number of purification cycles, solvent consumption, and processing time required to bring the API to market specifications. This reduction in processing intensity translates directly into lower utility costs and higher throughput capacity for existing manufacturing facilities. Moreover, the reagents utilized in this process, such as thionyl chloride and chloroformates, are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages that often plague specialized coupling reagents. The robustness of the method also enhances supply continuity, as the process is less sensitive to minor variations in reaction conditions compared to the delicate balance required for carbodiimide couplings.
- Cost Reduction in Manufacturing: The elimination of dicyclohexylcarbodiimide removes the need for expensive filtration steps to remove dicyclohexylurea precipitates, which often clog filters and slow down production lines. Additionally, the volatile nature of the byproducts (HCl and SO2) means that waste disposal costs are minimized compared to the handling of solid urea waste. The overall cost of goods sold (COGS) is optimized through higher effective yields and reduced solvent usage for recrystallization, providing a competitive pricing structure for high-purity perindopril intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on a synthesis path that avoids niche coupling agents mitigates the risk of supply disruptions. Thionyl chloride and basic carbonates are produced at massive scales globally, ensuring consistent availability and price stability. This reliability allows for better long-term planning and inventory management for pharmaceutical companies. Furthermore, the simplified workflow reduces the dependency on highly specialized operators, making it easier to scale production across different manufacturing sites without extensive retraining, thus securing the supply chain against labor fluctuations.
- Scalability and Environmental Compliance: The process is inherently scalable because the exothermic nature of the thionyl chloride reaction can be easily managed in large reactors using standard cooling systems. The removal of gaseous byproducts simplifies the reactor headspace management and reduces the volume of liquid waste generated. From an environmental standpoint, avoiding the persistence of urea derivatives in wastewater streams simplifies effluent treatment protocols. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports corporate sustainability goals, making the manufacturing process more resilient to tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel perindopril synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their current production portfolios. The answers highlight the specific advantages of the thionyl chloride method over traditional approaches, focusing on purity, safety, and operational efficiency.
Q: Why is the elimination of dicyclohexylcarbodiimide (DCC) critical for perindopril purity?
A: Conventional methods using DCC lead to the formation of persistent urea byproducts (specifically formulas VII and VIII) that are structurally similar to the target molecule and extremely difficult to remove, compromising pharmaceutical grade purity.
Q: What are the primary advantages of using thionyl chloride for activation in this process?
A: Thionyl chloride activation generates gaseous byproducts (HCl and SO2) that are easily removed from the reaction mixture, simplifying the workup process and avoiding the introduction of non-volatile contaminants associated with carbodiimide coupling agents.
Q: Can this synthetic route be scaled for industrial manufacturing of ACE inhibitors?
A: Yes, the process utilizes common industrial solvents like dichloromethane and ethyl acetate, and the reaction conditions (0°C to ambient temperature) are manageable on a large scale, making it highly suitable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perindopril Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the one described in CN1622936A requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with state-of-the-art rigorous QC labs capable of detecting trace impurities at ppm levels, guaranteeing that every batch of perindopril intermediate meets stringent purity specifications. We understand the critical nature of cardiovascular APIs and are committed to maintaining the highest standards of quality assurance throughout the entire manufacturing lifecycle, from raw material sourcing to final packaging.
We invite you to collaborate with us to leverage this innovative DCC-free technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your bottom line. Please contact us to request specific COA data and route feasibility assessments for high-purity perindopril intermediates. Let us help you secure a sustainable and cost-effective supply chain for your essential medications.
