Advanced Stereoselective Synthesis of Perindopril Intermediates for Commercial Scale-Up
Introduction to Advanced Perindopril Intermediate Manufacturing
The pharmaceutical industry continuously seeks robust, scalable, and economically viable pathways for the production of critical cardiovascular medications. Patent CN1176903C discloses a groundbreaking stereoselective method for the industrial synthesis of N-[(S)-1-carboxybutyl]-S-alanine ester derivatives, which serve as pivotal intermediates in the manufacture of Perindopril. This innovation addresses long-standing challenges in process chemistry by replacing hazardous reagents with a catalytic hydrogenation protocol that operates efficiently in an aqueous medium. The strategic shift towards using natural amino acids as chiral sources not only enhances the stereochemical integrity of the final product but also aligns with modern green chemistry principles. For global procurement teams and R&D directors, understanding this technological leap is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials consistently.
![Chemical structure of Formula I, the N-[(S)-1-carboxybutyl]-S-alanine ester intermediate](/insights/img/perindopril-intermediate-synthesis-catalytic-supplier-20260307214558-01.png)
The significance of this patent extends beyond mere chemical transformation; it represents a fundamental optimization of the supply chain for ACE inhibitors. By streamlining the synthesis of the key intermediate shown in Formula I, manufacturers can drastically reduce lead times and mitigate the risks associated with handling toxic reducing agents. The method ensures that the resulting compound possesses the requisite optical purity needed for downstream coupling reactions to form Perindopril, as depicted in Formula II. This level of process control is vital for maintaining regulatory compliance and ensuring the therapeutic efficacy of the final drug product.
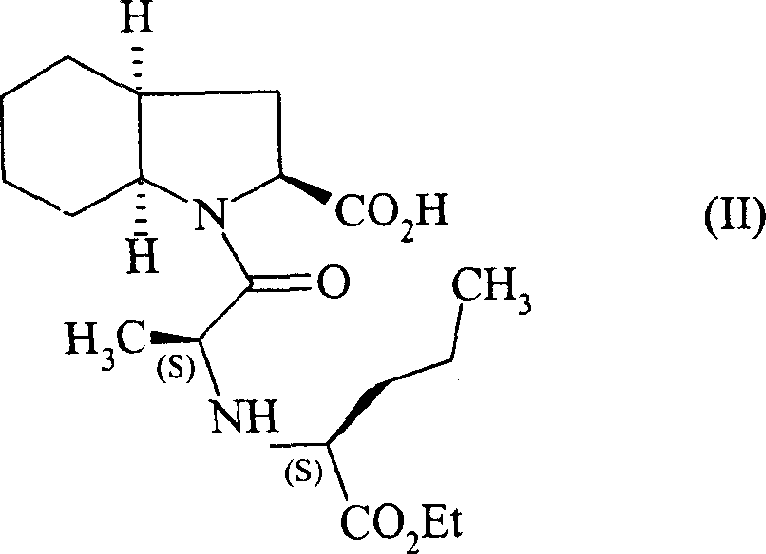
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of these critical intermediates has been plagued by significant operational and safety hurdles that hinder efficient cost reduction in API manufacturing. Early methodologies, such as those described in Tetrahedron Letters (1982), relied heavily on sodium cyanoborohydride as a reducing agent. While chemically effective on a small scale, this reagent is notoriously toxic, highly hygroscopic, and extremely difficult to handle safely in large industrial reactors, posing severe occupational health risks. Furthermore, alternative routes utilizing pyruvic acid, as seen in European patents EP0 308 340 and EP0 308 341, introduced substantial impurity profiles due to the inherent instability of the keto-acid starting material. These conventional processes often necessitated the use of hydrochloride salts of amino acids, requiring massive quantities of sodium hydroxide for neutralization, which inevitably generated excessive amounts of sodium chloride waste that complicated downstream purification.
The Novel Approach
In stark contrast, the novel approach detailed in CN1176903C revolutionizes the production landscape by employing a direct condensation followed by catalytic hydrogenation. This method utilizes ethyl 2-oxopentanoate (Formula IV) reacting with L-alanine in water, eliminating the need for hazardous cyanide-based reductants entirely. The process operates under mild conditions, typically between 10-60°C and at low hydrogen pressures of 1-5 bar, utilizing a standard 5% palladium on carbon catalyst. By minimizing the use of base (sodium hydroxide) to merely 0.1-0.2 moles per mole of substrate, the formation of inorganic salt byproducts is drastically curtailed. This innovation allows for a simplified workup procedure where the product precipitates upon acidification, avoiding the energy-intensive and time-consuming step of evaporating large volumes of water, thereby enhancing overall process throughput.
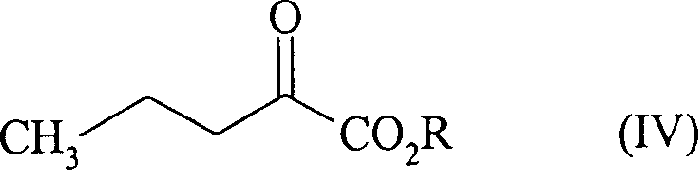
Mechanistic Insights into Pd/C-Catalyzed Reductive Amination
The core of this technological advancement lies in the precise execution of a stereoselective reductive amination mechanism. The reaction initiates with the condensation of the amino group of L-alanine with the ketone functionality of ethyl 2-oxopentanoate to form an imine or iminium intermediate in situ. The presence of the chiral center in the alanine moiety exerts a powerful diastereofacial influence during the subsequent hydrogenation step. When exposed to the palladium catalyst surface under hydrogen pressure, the imine is reduced preferentially to form the (S,S) diastereomer. This intrinsic stereocontrol is crucial because it bypasses the need for complex resolution steps or chiral chromatography that are often required in non-selective syntheses. The aqueous environment plays a dual role here: it acts as a benign solvent that facilitates the solubility of the zwitterionic amino acid while also helping to stabilize the transition states involved in the catalytic cycle.
From an impurity control perspective, this mechanism offers superior cleanliness compared to prior art. The avoidance of pyruvic acid eliminates the risk of decarboxylation byproducts and self-condensation impurities that typically contaminate the crude reaction mass. Furthermore, the use of catalytic hydrogenation ensures that the reduction is clean and specific to the C=N bond, leaving the ester functionalities intact. The final purification step involves a simple acidification to pH 3-3.5, which protonates the amine and induces precipitation of the product while leaving most organic impurities in the mother liquor. Subsequent recrystallization from acetonitrile further upgrades the optical purity, ensuring that the material meets the stringent specifications required for a high-purity pharmaceutical intermediate destined for human consumption.
How to Synthesize N-[(S)-1-carboxybutyl]-S-alanine ester Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and stereochemical fidelity. The process begins by dissolving L-alanine in water with a stoichiometric amount of sodium hydroxide, followed by the addition of the keto-ester. Once the condensation equilibrium is established, the mixture is transferred to a hydrogenation vessel containing the palladium catalyst. The reaction is monitored for hydrogen uptake, and upon completion, the catalyst is removed via filtration.
- Condense L-alanine with ethyl 2-oxopentanoate (Formula IV) in an aqueous medium with minimal sodium hydroxide.
- Perform catalytic hydrogenation using 5% palladium on carbon at 1-5 bar pressure and 10-40°C.
- Acidify the reaction mixture to pH 3-3.5, filter the precipitate, and recrystallize from acetonitrile to obtain the pure (S,S) isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that go beyond simple chemical yields. The elimination of toxic sodium cyanoborohydride removes a major regulatory and safety bottleneck, significantly reducing the costs associated with hazardous waste disposal and specialized handling equipment. Moreover, the shift to an aqueous-based system reduces reliance on volatile organic solvents, which are subject to fluctuating market prices and strict environmental regulations. This process stability ensures a more predictable production schedule, allowing suppliers to offer more competitive pricing structures without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the simplification of the unit operations. By avoiding the energy-intensive evaporation of water and the extensive washing required to remove excess salts, the utility consumption per kilogram of product is significantly lowered. Additionally, the use of inexpensive, naturally sourced L-alanine as a chiral building block reduces the raw material cost basis compared to synthetic chiral auxiliaries. The high selectivity of the reaction minimizes the loss of valuable materials to side products, effectively increasing the overall mass balance efficiency and driving down the cost of goods sold.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. Operating at low pressures (1-5 bar) and moderate temperatures means that the process can be executed in standard glass-lined or stainless steel reactors available in most multipurpose chemical plants, reducing the need for specialized high-pressure infrastructure. The simplicity of the workup—filtration followed by recrystallization—shortens the batch cycle time considerably. This agility allows manufacturers to respond more rapidly to demand spikes, ensuring a continuous flow of materials for downstream API synthesis and reducing the risk of stockouts.
- Scalability and Environmental Compliance: From an environmental standpoint, this method represents a significant leap towards sustainable manufacturing. The drastic reduction in sodium chloride waste alleviates the burden on wastewater treatment facilities and minimizes the environmental footprint of the production site. The absence of cyanide reagents eliminates the risk of catastrophic contamination events, aligning the production process with increasingly rigorous global safety standards. This eco-friendly profile not only facilitates easier permitting for capacity expansion but also enhances the brand reputation of the supply chain partners involved in the production of cardiovascular therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is critical for stakeholders evaluating the feasibility of integrating this intermediate into their existing supply networks.
Q: What are the primary advantages of this synthesis method over prior art?
A: This method eliminates the use of toxic sodium cyanoborohydride and unstable pyruvic acid. It significantly reduces salt waste (NaCl) formation and avoids energy-intensive water evaporation steps, leading to a cleaner and more cost-effective process.
Q: How is stereochemical purity controlled in this process?
A: The process utilizes natural L-alanine as a chiral source, ensuring the correct configuration at one center. The subsequent reductive amination under controlled catalytic conditions favors the formation of the desired (S,S) diastereomer, which is further purified via recrystallization.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It uses water as the primary solvent, operates at moderate pressures (1-5 bar), and employs a simple filtration workup instead of complex distillation, making it highly robust for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perindopril Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving cardiovascular medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated catalytic hydrogenation techniques described in CN1176903C are executed with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-[(S)-1-carboxybutyl]-S-alanine ester meets the exacting standards required for GMP API synthesis. Our commitment to process excellence ensures that our clients receive materials that facilitate smooth downstream processing and final drug registration.
We invite global pharmaceutical partners to collaborate with us to optimize their supply chains for Perindopril production. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical foundation available in the market.
